-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Environmental Sex Determination in the Branchiopod Crustacean : Deep Conservation of a Gene in the Sex-Determining Pathway
Sex-determining mechanisms are diverse among animal lineages and can be broadly divided into two major categories: genetic and environmental. In contrast to genetic sex determination (GSD), little is known about the molecular mechanisms underlying environmental sex determination (ESD). The Doublesex (Dsx) genes play an important role in controlling sexual dimorphism in genetic sex-determining organisms such as nematodes, insects, and vertebrates. Here we report the identification of two Dsx genes from Daphnia magna, a freshwater branchiopod crustacean that parthenogenetically produces males in response to environmental cues. One of these genes, designated DapmaDsx1, is responsible for the male trait development when expressed during environmental sex determination. The domain organization of DapmaDsx1 was similar to that of Dsx from insects, which are thought to be the sister group of branchiopod crustaceans. Intriguingly, the molecular basis for sexually dimorphic expression of DapmaDsx1 is different from that of insects. Rather than being regulated sex-specifically at the level of pre–mRNA splicing in the coding region, DapmaDsx1 exhibits sexually dimorphic differences in the abundance of its transcripts. During embryogenesis, expression of DapmaDsx1 was increased only in males and its transcripts were primarily detected in male-specific structures. Knock-down of DapmaDsx1 in male embryos resulted in the production of female traits including ovarian maturation, whereas ectopic expression of DapmaDsx1 in female embryos resulted in the development of male-like phenotypes. Expression patterns of another D. magna Dsx gene, DapmaDsx2, were similar to those of DapmaDsx1, but silencing and overexpression of this gene did not induce any clear phenotypic changes. These results establish DapmaDsx1 as a key regulator of the male phenotype. Our findings reveal how ESD is implemented by selective expression of a fundamental genetic component that is functionally conserved in animals using GSD. We infer that there is an ancient, previously unidentified link between genetic and environmental sex determination.
Published in the journal: . PLoS Genet 7(3): e32767. doi:10.1371/journal.pgen.1001345
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1001345Summary
Sex-determining mechanisms are diverse among animal lineages and can be broadly divided into two major categories: genetic and environmental. In contrast to genetic sex determination (GSD), little is known about the molecular mechanisms underlying environmental sex determination (ESD). The Doublesex (Dsx) genes play an important role in controlling sexual dimorphism in genetic sex-determining organisms such as nematodes, insects, and vertebrates. Here we report the identification of two Dsx genes from Daphnia magna, a freshwater branchiopod crustacean that parthenogenetically produces males in response to environmental cues. One of these genes, designated DapmaDsx1, is responsible for the male trait development when expressed during environmental sex determination. The domain organization of DapmaDsx1 was similar to that of Dsx from insects, which are thought to be the sister group of branchiopod crustaceans. Intriguingly, the molecular basis for sexually dimorphic expression of DapmaDsx1 is different from that of insects. Rather than being regulated sex-specifically at the level of pre–mRNA splicing in the coding region, DapmaDsx1 exhibits sexually dimorphic differences in the abundance of its transcripts. During embryogenesis, expression of DapmaDsx1 was increased only in males and its transcripts were primarily detected in male-specific structures. Knock-down of DapmaDsx1 in male embryos resulted in the production of female traits including ovarian maturation, whereas ectopic expression of DapmaDsx1 in female embryos resulted in the development of male-like phenotypes. Expression patterns of another D. magna Dsx gene, DapmaDsx2, were similar to those of DapmaDsx1, but silencing and overexpression of this gene did not induce any clear phenotypic changes. These results establish DapmaDsx1 as a key regulator of the male phenotype. Our findings reveal how ESD is implemented by selective expression of a fundamental genetic component that is functionally conserved in animals using GSD. We infer that there is an ancient, previously unidentified link between genetic and environmental sex determination.
Introduction
Sex determination is a fundamental biological process. It affects not only the sexual differentiation of gonads, but also the development of most organs, and leads to sex-specific differences in behavior, physiology and morphology. Organisms have evolved a variety of different sex-determining systems [1], [2] that can be broadly divided phenomenologically into two categories: genetic and environmental [3]. Genetic sex determination (GSD) is attributed to the genetic segregation of genes, often residing on sex chromosomes that initiate alternate sex-determining developmental pathways. Environmental sex determination (ESD) is initiated by environmental cues that presumably trigger alternative genetic signals, which regulate male or female sex-determining genes [4]. Although GSD is a more prevalent system in animals, ESD is also phylogenetically widespread, occurring in such diverse taxa as rotifers, nematodes, crustaceans, insects, fishes, and reptiles [5]. Environmental cues involved in ESD include temperature, photoperiod, nutrition, and population density [5]. Temperature is the most widely studied environmental cue, particularly in the case of reptiles where the temperature at which the egg is incubated determines sex [6]. ESD has arisen repeatedly during evolution [7], which may imply the adaptive significance of this system in environments [8].
It has long been suggested that selection forces drove the transition between GSD and ESD [9], [10]. Previous experiments using a temperature-sensitive mutation created artificially in Drosophila melanogaster and Caenorhabditis elegans also demonstrated how GSD could rapidly evolve into ESD as a consequence of a mutation in a single control gene [11], [12]. In addition, orthologs of some genes involved in GSD have been examined in species that use ESD, especially in temperature-dependent sex-determining reptiles [13]. Some of those are expressed in the gonads during the temperature-sensitive period [13]. These observations have led to the hypothesis that both ESD and GSD should have the same origin and share similar genetic components in their sex-determining pathways [4]. However, a complete functional analysis of temperature dependent ESD has not yet been performed. Therefore, analyzing function of genes involved in ESD and unraveling the sex-determining pathways are crucial to understand the origin and evolution of sex-determining pathway.
The water flea Daphnia magna is a branchiopod crustacean, which is a common inhabitant in fresh water ponds in Europe and Asia. D. magna is known to switch between parthenogenetic and sexual reproduction when environmental quality declines [14]. Normal, healthy populations are entirely female. However, shortened photoperiod, a lack of food and/or increased population density, lead to the clonal production of males that are genetically identical to their sisters and mothers. First instar male juveniles are easily distinguished from the females by their elongated first antennae [15]. During maturation, daphnids undergo morphological sexual differentiation of various somatic tissues including the first thoracic leg that is armed with the copulatory hook in males, which becomes larger in the fifth instar [16]. Gonads develop and finally settle at both sides of the gut during embryogenesis in both males and females [17]. It has been reported that the gonads exhibit morphological sex differences in the first instar juveniles [18], [19]. The appearance of males allows sexual reproduction to occur [20], [21] when females begin producing haploid eggs requiring fertilization.
Recently, we and others have shown that juvenile hormone analogs (JHA) induce male production in cladoceran crustaceans without environmental cues [22], [23]. Interestingly, exposure of D. magna to JHA at the stage corresponding to the environmentally-sensitive period for the other cladoceran Moina sex determination [24], reliably produces exclusively male broods, suggesting that juvenile hormone could be a key molecule for understanding environmental sex determination [22], [25], [26]. Together with the growing genome and transcriptome resources for Daphnia [27]-[30], this system is ideal to study genes responsible for ESD.
Mechanisms underlying genetic sex-determining pathways have been extensively studied in model organisms such as D. melanogaster, C. elegans and mouse and key genes have been identified [31]-[33]. A Doublesex (Dsx) gene was originally identified in D. melanogaster as a critical transcription factor considered to be at the end of sex determination cascades in GSD, that directly targets genes conferring sexually dimorphic traits [34]. Dsx contains two conserved domains: one is the Dsx/Mab-3 (DM) domain at the N-terminus that is evolutionarily conserved even in vertebrates [35] and another is the oligomerization domain at the C-terminus [36]. Genes encoding DM-domain (DM-domain genes) were discovered to play a related role not only in C. elegans [37], [38], but even in vertebrates [39]. In contrast, results from numerous studies have shown that other genetic sex-determining genes are widely diverse among species [1], [2], [40].
To understand the molecular and evolutionary relationships between GSD and ESD, we analyzed the function of two Dsx genes from D. magna using gene manipulations that we have developed [41]. We provide evidence that one of the homologs, termed DapmaDsx1, plays an important role in directing the major sexually dimorphic development of D. magna. Intriguingly, the function of Dsx is significantly conserved between Daphnia and genetic sex-determining insect species, which are thought to be the sister group of branchiopod crustaceans [42]; however, the factors that regulate Dsx gene expression are independently co-opted in each lineage. Our functional demonstration that Dsx controls sexual dimorphism in an environmental sex-determining organism supports the hypothesis that genetic and environmental sex determination are similar at their most fundamental level.
Results
Molecular Cloning of Two Doublesex Genes from Daphnia
In an effort to understand environmental sex determination, we previously identified three DM-domain genes from D. magna and showed that two of the three DM-domain genes have sex dimorphic gene expression pattern in adult gonads [43]. However, none of these DM-domain genes exhibited sexually dimorphic expression patterns during embryonic development, suggesting that they are not involved in sex determination (Figure S1). This result prompted us to search for other DM-domain genes that might be involved in Daphnia sex determination. Two additional DM-domain genes were found in the D. magna EST database [27] and we cloned and sequenced cDNAs encoding each.
These newly identified DM-domain genes showed greater sequence similarity at the amino acid sequence level to known insect Dsx genes than to the previously identified Dsx-related genes. Therefore, these were designated Daphnia magna Dsx (DapmaDsx). One of the DapmaDsx genes (DapmaDsx1) encodes a protein of 330 amino acids from two mRNAs (DapmaDsx1-α and DapmaDsx1-β) that differ only in their 5'UTR (Figure 1A and Figure S2). The other (DapmaDsx2) encodes a protein of 314 amino acids (Figure S3). The predicted protein products of these two genes share 38% overall amino acid identity and both contain a DM-domain (Figure 1B). In addition, both genes also have an oligomerization domain that is characteristic for insect Dsx homologs (Figure 1C). Dimerization, which enhances specific DNA binding, is mediated by an extensive non-polar interface conserved within oligomerization domain. Importantly, in DapmaDsx2, two of three non-polar amino acids important in formation of the interface are substituted with the acidic amino acid, aspartic acid (Figure 1C). Phylogenetic analysis of these DM-domain genes confirmed that Daphnia Dsx genes are most closely related to the insect Dsx genes, but that the two Daphnia genes are paralogs that duplicated, forming a tandem gene cluster after the divergence of insects and crustaceans (Figure 1A and Figure S4).
Fig. 1. Daphnia Dsx genes. 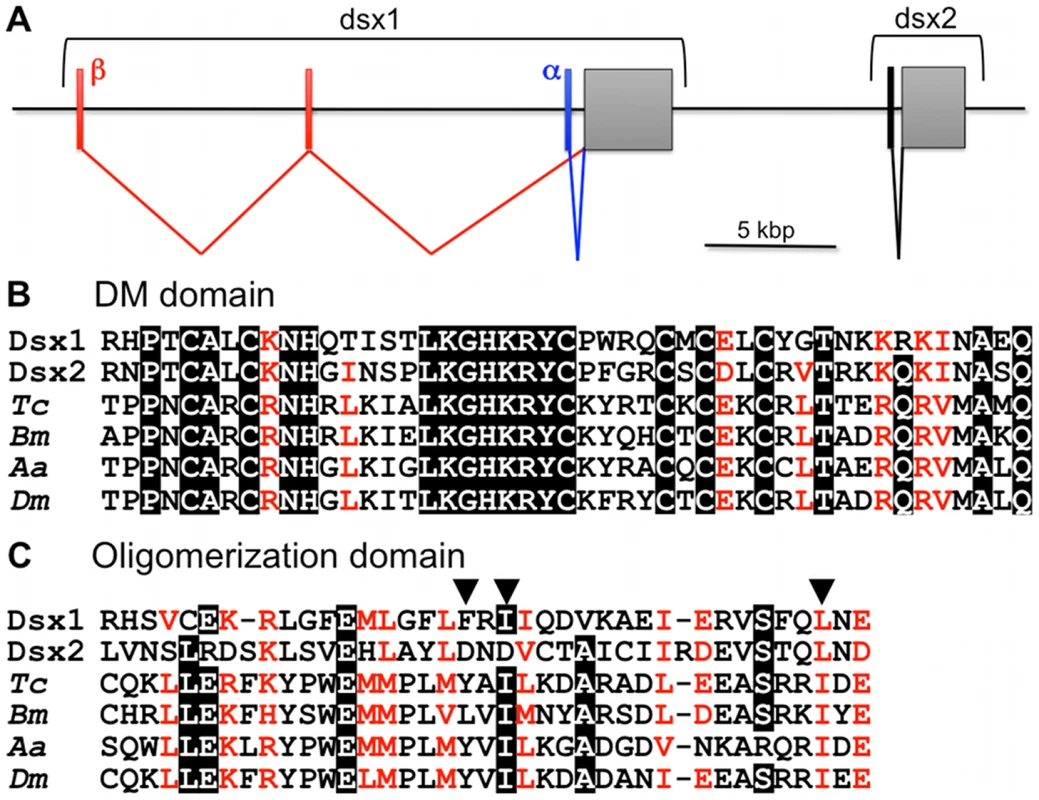
(A) Genomic organization of two dsx genes in Daphnia magna. Coding exons encoding Dsx1 and Dsx2 are indicated as grey boxes, respectively. Exons comprising DSX1-α, DSX1-β and DSX2 5'UTR are indicated as blue, red and black bars. (B, C) Alignment of deduced amino acid sequences of DM domains and oligomerization domains of dsx genes. Amino acid sequences were aligned using CLUSTAL-W. Identical amino acids are highlighted in black. Similar amino acids are shown in red. Positions of non-polar amino acids important in formation of the hydrophobic interface between oligomerization domains in Drosophila Dsx protein were indicated with solid triangles [36]. Tc, Tribolium castaneum: Bm, Bombyx mori; Aa, Aedes aegypti; Dm, Drosophila melanogaster. Sexually Dimorphic Expression of Daphnia Dsx
We next examined expression levels of DapmaDsx genes during development by quantitative real time PCR. Expression of both DapmaDsx1 and DapmaDsx2 genes increased over 72 h exclusively in male embryos (Figure 2A, dsx1, dsx2). This temporal expression correlates well with the development of sexually dimorphic organs (e.g., gonads and first antennae), in which morphological sex differences are observed at 72 h after ovulation when hatchlings begin to swim out from the brood chamber. This temporal expression pattern suggests that one or both of the Daphnia Dsx genes might play a critical role in sexual differentiation. To examine whether the two different DapmaDsx1 promoters are used in a temporally independent manner, the expression levels of DapmaDsx1-α and DapmaDsx1-β mRNAs were evaluated. Both mRNAs increased only in males during early development, suggesting that both promoters function primarily during male development (Figure 2A, dsx1-α). DapmaDsx1-β mRNA could be detected immediately post-ovulation in males and females (Figure 2A, dsx1-β), suggesting that maternal DapmaDsx1-β mRNA is transferred to ovulated eggs.
Fig. 2. Temporal and spatial dimorphic gene expression of Dsx genes during development. 
(A) Male and female embryos were obtained and gene expression levels of Dsx1 and Dsx2 were determined at 18 h, 42 h and 72 h after ovulation by quantitative RT-PCR using primers corresponding to Dsx1 and Dsx2 coding sequence (CDS). Dsx1-α and DSX1-β transcripts were also quantified using primers specific to each 5'UTR. Copy numbers were estimated by quantification compared with an external standard and dividing by the number of embryos used. Bars indicate S.E.M. (B) Expression of dsx genes in adult gonads was quantified. PCR primers corresponding to Dsx1 CDS, two types of 5'UTRs of Dsx1 gene and Dsx2 CDS were used for quantitative PCR. Bars indicate S.E.M. (C) Schematic illustration of D. magna late embryo. The red boxes indicate the areas shown in panel D. (D) Whole mount in situ hybridization in late embryos using DIG-labeled probes corresponding to Dsx1 and Dsx2. The heads and the thoracic segments are magnified. Bars indicate 50 µm (heads) and 100 µm (thoracic segments). CE: Compound eye, Oc: Ocellus, An1: First antennae, An2: Second antennae, e: Epipod, T1-5: First to fifth thoracic segments. We next examined whether DapmaDsx genes are expressed in male specific structures. Both DapmaDsx1 and DapmaDsx2 genes were highly expressed in the testis (Figure 2B). Whereas DapmaDsx1-α mRNA was expressed exclusively in the testis, DapmaDsx1-β mRNA was expressed in both testis and ovary. Whole mount in situ hybridization showed that DapmaDsx1 and DapmaDsx2 could both be detected in the first antennae and the first thoracic segments (Figure 2C and 2D), both of which are known to show sexually dimorphic characteristics. DapmaDsx2 is also expressed in female first thoracic segments though apparently more weakly than in males. Male-specific expression was also observed in the compound eye, whose sex difference has not been reported in this species to date. Taken together, these male specific expression patterns are regulated temporally and spatially, supporting strongly the involvement of DapmaDsx genes in male differentiation in Daphnia.
Daphnia Dsx1 Is Necessary for Male Trait Development
The sexually dimorphic expression of DapmaDsx mRNAs led us to hypothesize that the expression level of the DapmaDsx transcripts could mediate sex determination. To test this hypothesis, we established a technique to introduce exogenous genes into ovulated eggs and developed a dsRNA-based gene knockdown technique for D. magna [41]. Eggs induced to become males by fenoxycarb exposure to the mother during a critical stage of oocyte development [22], [26] were injected with Dsx-specific, or control dsRNAs, grown to the swimming juveniles and evaluated the phenotypes.
At the third instar stage, microinjection of the DapmaDsx1-specific dsRNAs resulted in development of the shortened first antenna whose length was the same as that of females in all of the DapmaDsx1-dsRNA-injected juveniles (Figure 3A). At the fifth instar stage, we dissected the feminized daphnids and found that the first thoracic appendage lacks a hook used in clasping the females and has a female-like long filament instead (Figure 3B). Correspondingly, repression of DapmaDsx1 resulted in the development of ovaries during which oocytes accumulate yolk granules and lipid droplets as well as those of wild-type females (Figure 3C). In contrast, microinjection of DapmaDsx2-dsRNAs did not induce the formation of either female-like somatic or gonadal tissues (Figure 3A–3C, Table 1).
Fig. 3. Dimorphic development of Daphnia magna. 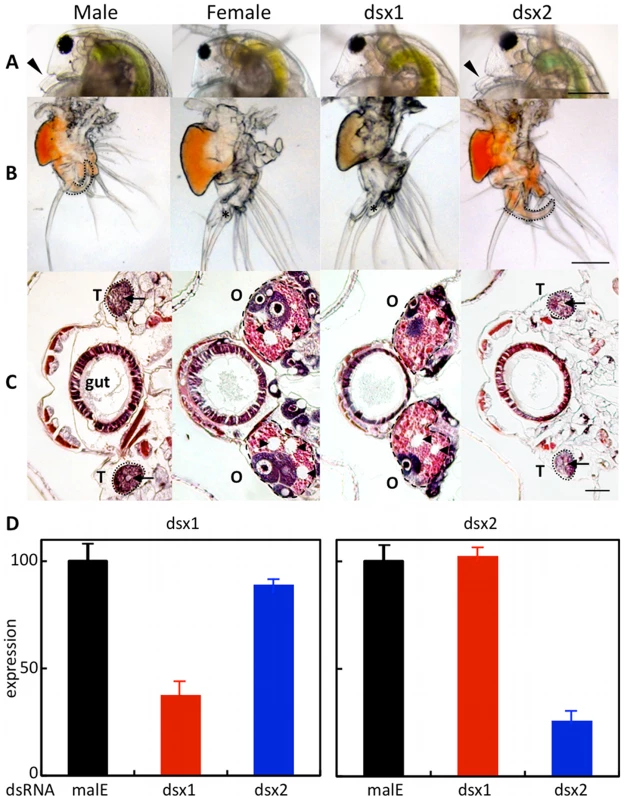
Eggs induced to become males were obtained from D. magna. After injection of the synthesized dsRNA, sexually dimorphic phenotypes were examined at the fifth or sixth instar except first antennae (third instar). The first two columns represent normal male and female phenotypes, respectively. The third and fourth columns represent phenotypes of individuals injected with #1-dsRNA of dsx1 and dsx2, respectively. (A): Lateral view of the head. Arrowheads indicate the first antennae. (B): First thoracic limb. Dotted line shows the outline of the stout chitinized hook. A female-type long filament corresponding to the hook is labeled with an asterisk. (C): Gonad. Daphnids were embedded in paraffin and sectioned, following by standard hematoxylin and eosin staining. Dorsal is left, ventral is right. Dotted circled lines show gonads at both sides of a gut. T and O indicate testis and ovary, respectively. Arrowheads indicate large lipid droplets lying among the eosinophilic yolk granules. Arrows indicate lumens into which the mature spermatozoa are released. (D): Gene expression profile of Dsx1 and Dsx2 in embryos injected with dsRNA of Dsx1 (left panel) and in dsRNA of Dsx2 (right panel). The MalE gene from E. coli was used as a control gene. Bars in (A), (B) and (C) indicate 200, 100, 50 µm, respectively. Tab. 1. Summary of RNA interference using dsRNA. 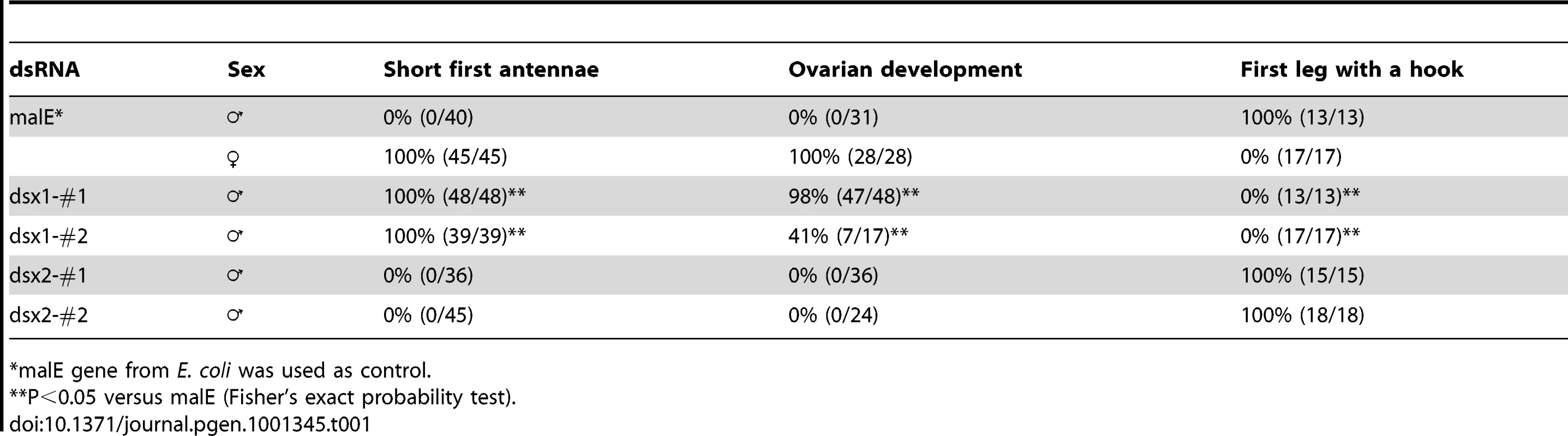
*malE gene from E. coli was used as control. The quantity of DapmaDsx1 and DapmaDsx2 mRNA was decreased to 40% and 20%, respectively, relative to control-dsRNA-injected embryos. Reduction of transcripts from either of the two DapmaDsx genes did not change the mRNA level of the other (Figure 3D), suggesting that the DapmaDsx genes do not regulate each other's expression or stability. We confirmed the specificity of the knockdown by using non-overlapping dsRNAs for each DapmaDsx gene (Figure S5). As expected, only DapmaDsx1 RNAi induced the same phenotypic changes (Table 1). Since the size of testes appears to be somewhat reduced following the DapmaDsx2-dsRNA injection (Figure 3C), we cannot rule out some low-level requirement for DapmaDsx2 function. However, these results suggest that DapmaDsx1 is necessary for sex determination.
To ask whether expression of DapmaDsx1 or Dsx2 might be sufficient to trigger male development in females, we developed a technique for transient transgenesis in Daphnia embryos by microinjection of capped, polyadenylated mRNAs into ovulated eggs. When the DapmaDsx1 or DapmaDsx2 mRNA was introduced into female eggs, only DapmaDsx1 mRNA partially induced elongation of the first antenna in females at 72 h after microinjection (Figure 4 and Table 2). Unfortunately, due to the transient nature of this method, the masculinization of the first antennae was observed only in the first instar juveniles, whose gonads were too small to evaluate the sex-reversal. Taken together, these data show that although they are similar in amino acid sequence and expression pattern, DapmaDsx1 and not DapmaDsx2 plays a primary role for male trait development.
Fig. 4. Elongation of 1st antenna by the expression of Dsx1 gene. 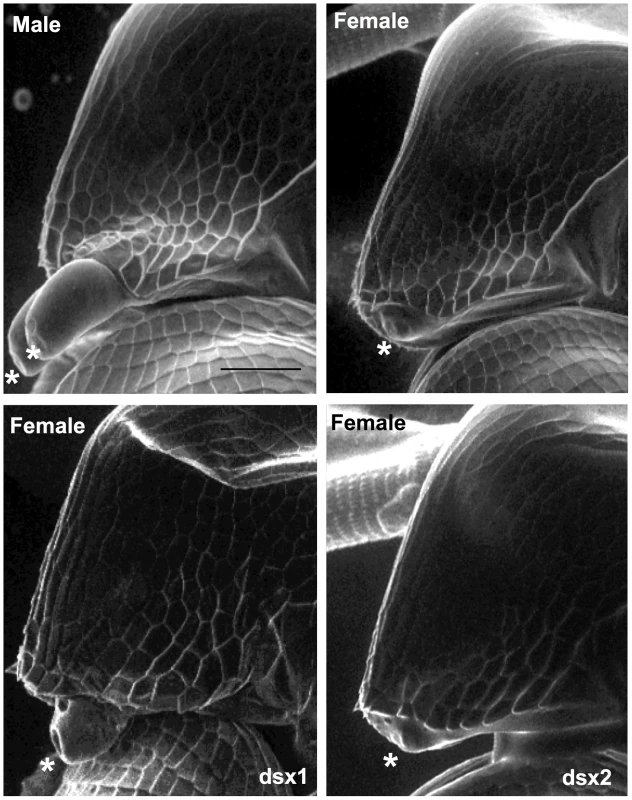
The mRNA was injected to embryos within one hour after ovulation and observed using electron microscope after 72 h. Male and female indicate normal phenotype of each sex. Dsx1 and Dsx2 indicate mRNA of Dsx1 and Dsx2, which were injected to female eggs, respectively. Asterisk indicates first antennae. Bar indicates 100 µm. Tab. 2. Summary of effects caused by ectopic expression of Dsx genes in female embryos. 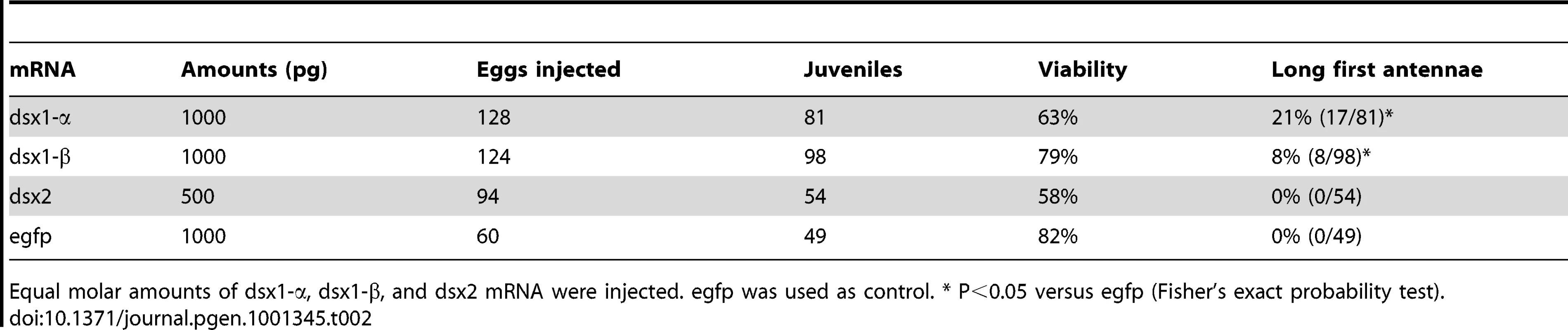
Equal molar amounts of dsx1-α, dsx1-β, and dsx2 mRNA were injected. egfp was used as control. * P<0.05 versus egfp (Fisher's exact probability test). Daphnia Dsx1 Gene Lacks the Phylogenetically Conserved Splicing Variants
Recent phylogenetic analyses [44]-[46] and developmental genetics [47], [48] suggest that insects may be a sister group to branchiopod crustaceans, a group that includes daphnids (water fleas) and brine shrimp Artemia [42]. As it is known that insect Dsx genes express sex-specific variants in their coding sequences, we next examined whether the sex-specific splicing of the Daphnia Dsx1 gene also occurs. Only a single amplified cDNA could be detected from either male or female by RT-PCR with primers to amplify a coding sequence, although PCR products obtained from females were very faint (Figure 5A). We sequenced the cDNA fragments and confirmed no sex differences of the fragments.
Fig. 5. Structure of Dsx1 mRNAs expressed in males and females. 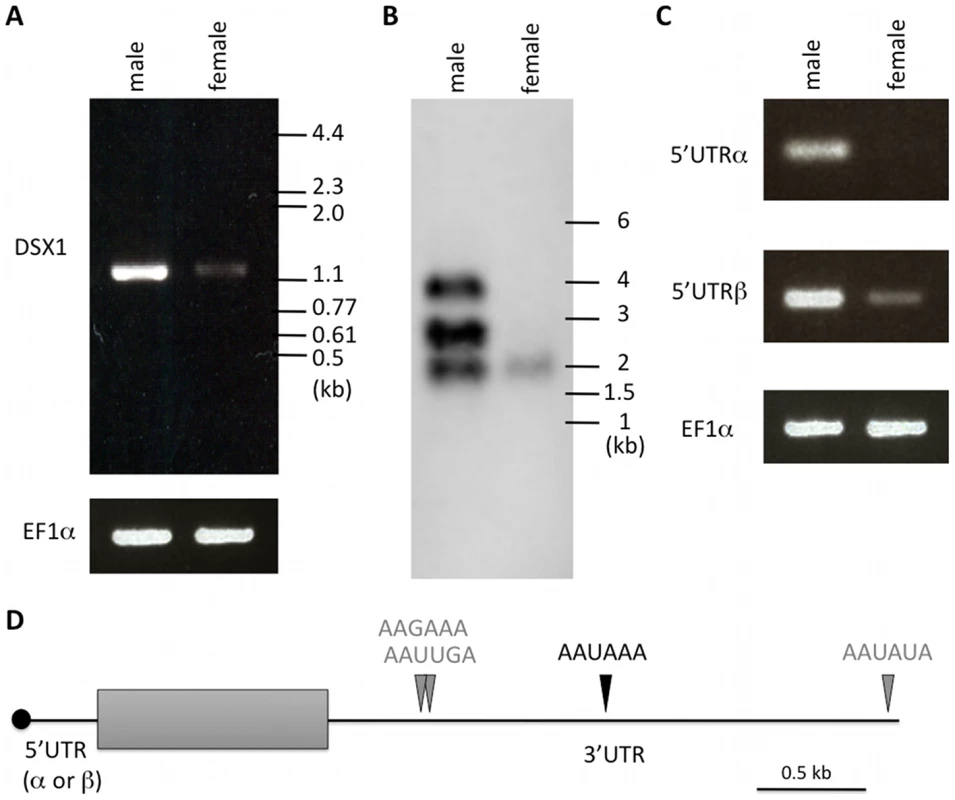
(A) RT-PCR using oligonucleotides corresponding to 5'- and 3'-ends of Dsx1 CDS. The amplified cDNAs were resolved by agarose gel electrophoresis. (B) A northern blot probed for dsx1 mRNAs. Migration of markers with lengths indicated (kb) is shown at the right. (C) RT-PCR using oligonucleotides to amplify 5'UTR-α and -β of Dsx1 gene. The amplified cDNAs were resolved by agarose gel electrophoresis. (D) Schematic illustration of Dsx1 mRNAs with alternative isoforms due to usage of alternative promoters and polyadenylation signals. A grey box shows protein coding region; black line represents untranslated regions. Canonical and non-canonical functional polyadenylation signals identified by 3'RACE are indicated with black and grey arrowheads. Further, to find altered DapmaDsx1 mRNA length and abundance, we used a northern blot with an antisense probe hybridized to the coding region. In male adults, three transcripts of approximately 4 kb, 2.8 kb, and 2 kb were detected. Of the three, 2.8 kb transcripts were the most abundant. In contrast, only faint 2 kb transcripts were detected in female adults (Figure 5B). As length differences between 5'UTR-α and -β were only 112 bases, one explanation for large differences of the transcript lengths might be the 3'UTR lengths. To determine the 3'end of DapmaDsx1 mRNAs, 3'RACE was performed. We identified four tandem polyadenylation sites located downstream of the stop codon and found that alternative usage of those sites was correlated well with production of the three DapmaDsx1 transcripts. The canonical AAUAAA signal was used for 2.8 kb transcripts, the variant AAUAUA for the 4-kb, and for the shortest 2 kb transcripts the two variant signals were used (AAGAAA or AAUUGA, Figure 5D, Figure S2). Interestingly, RT-PCR analysis with primers to discriminate between 5'UTR-α and -β showed that female 2 kb transcripts were only DapmaDsx1-β mRNAs with the shortest 3'UTR that presumably were supplied to eggs as maternal mRNAs (Figure 5C).
Discussion
We report here that gain - and loss-of-function analyses in environmental sex-determining Daphnia have allowed us to characterize the function of DapmaDsx1 in sexual dimorphism and provide insight into the molecular relationship between GSD and ESD. We and others previously showed that exposure to juvenile hormone analogs reliably produces male Daphnia [25], [49]. This finding enabled us to examine genes in embryos directed to either male or female. Together with the growing genome and transcriptome resources and gene manipulation techniques for Daphnia [27]-[30], , this species is a first crustacean model that provides novel insights into understanding evolution of the sex-determining pathway.
The molecular mechanisms of genetic sex determination have been well studied in a few model organisms, such as the mouse, fruit fly and nematode. DM-domain genes are highly conserved and involved in sexual differentiation of these species [38]. The DM-domain gene was also identified as a sex-determining gene in some populations of fish of the genus Medaka [51]. Moreover, recently, molecular analyses of GSD in the frog Xenopus laevis [52] and chicken Gallus gallus [53] demonstrate deep conservation of DM-domain genes in GSD. To our knowledge, this study is the first in vivo demonstration that the Dsx gene, a fundamental genetic component that is functionally conserved in animals using GSD, can also implement ESD. Interestingly, in reptiles with temperature-dependent sex determination, the Dsx ortholog, Dmrt1 is regulated by temperature [54]-[56]. These indicate that DM-domain genes also play an important role in environmental sex determining organisms, supporting the hypothesis that both ESD and GSD have the same origin and share similar genetic components in their sex determining pathways.
Crustacean and Insect Doublesex Share Molecular Characteristics to Function as a Major Effector of Sexual Dimorphism
DapmaDsx1, one of two Dsx homologs from Daphnia, shares several important characteristic features of Drosophila Dsx protein to function as a major effector of sexual differentiation [40], [57], [58]. First, DapmaDsx1 protein is composed of two domains, the phylogenetically conserved DM-domain [35] and the insect-specific oligomerization domain. In contrast, the DapmaDsx2 protein appears to be unable to regulate sexual differentiation and contains mutations at important amino acids of oligomerization domain [36], suggesting that this domain might be necessary for establishing sexual dimorphic traits. Second, male-specific expression of DapmaDsx1 is regulated temporally and spatially during development. Although it has long been believed that Drosophila Dsx gene was cell-autonomously expressed in all cells [59], Robinette et al. [60] recently reported that Drosophila also exclusively expressed Dsx in sexually dimorphic tissues and cells. Third, and perhaps most importantly, knock-down of DapmaDsx1 in male embryos resulted in the production of female traits including ovarian maturation whereas ectopic expression of DapmaDsx1 in female embryos resulted in partial masculinization of the first antennae. These results suggest that Dsx gene expression in sexually dimorphic tissues is a key process to induce sexual differentiation in crustacean Daphnia and insects.
Factors Regulating Expression of Doublesex Are Independently Co-Opted during Evolution in Each Lineage
In the fruit fly D. melanogaster, Dsx is spliced in the coding region by the Tra protein in a sex-dependent manner [34]. The female-specific RNA produced by alternative splicing is a functional mediator of Tra activity [61]. The female-specific splice variant of the Tra homologs encodes a functional protein not only in the Mediterranean fruit fly [62] and the house fly [63], but also in the honeybee Apis mellifera [64]. In all insect species studied to date (except the silkworm Bombyx mori) [65], Tra regulates sex-specific splicing of Dsx, which produces different mRNAs and proteins, resulting in sex-specific transcriptional activation and repression [66] (Figure 6). Sex-specific splicing of the Dsx gene by the Tra protein might be ancestral in insects. In contrast, DapmaDsx genes do not encode sex-specific Dsx proteins, but instead exhibit sexually dimorphic differences in the abundance of its transcripts. Interestingly, Daphnia has a homolog of the Tra protein but the D. magna Tra gene does not display any detectable sexually dimorphic differences in expression or splicing patterns [26]. We also performed knock-down of the D. magna Tra gene, but could not find any effect for development of sexually dimorphic traits (data not shown). This is consistent with the apparent lack of a sex-specific splicing in the Dsx1 gene. Although it is not yet clear if juvenile hormone directly activates the transcription of DapmaDsx genes, this remains an interesting possibility for future study. We found several motifs in promoter regions of the Dsx1 and Dsx2 genes, which resemble JH-responsive elements previously reported in D. melanogaster [67] and D. magna [68] (data not shown), suggesting that these motifs possibly function as elements to regulate the JH-dependent gene expression. The detection of unfavorable environmental conditions by Daphnia could be transmitted to the endocrine system, leading to the release of juvenile hormone to convey the environmental signals to sexually dimorphic cells. This would be a simple and elegant type of sex determination cascade. Understanding the molecular nature by which the transcription of the DapmaDsx genes is regulated remains an important future goal that will greatly enhance our understanding of not only sex determination, but also invertebrate hormonal systems.
Fig. 6. Simplified view of sex-determining pathways in the branchiopod crustacean Daphnia and insects. 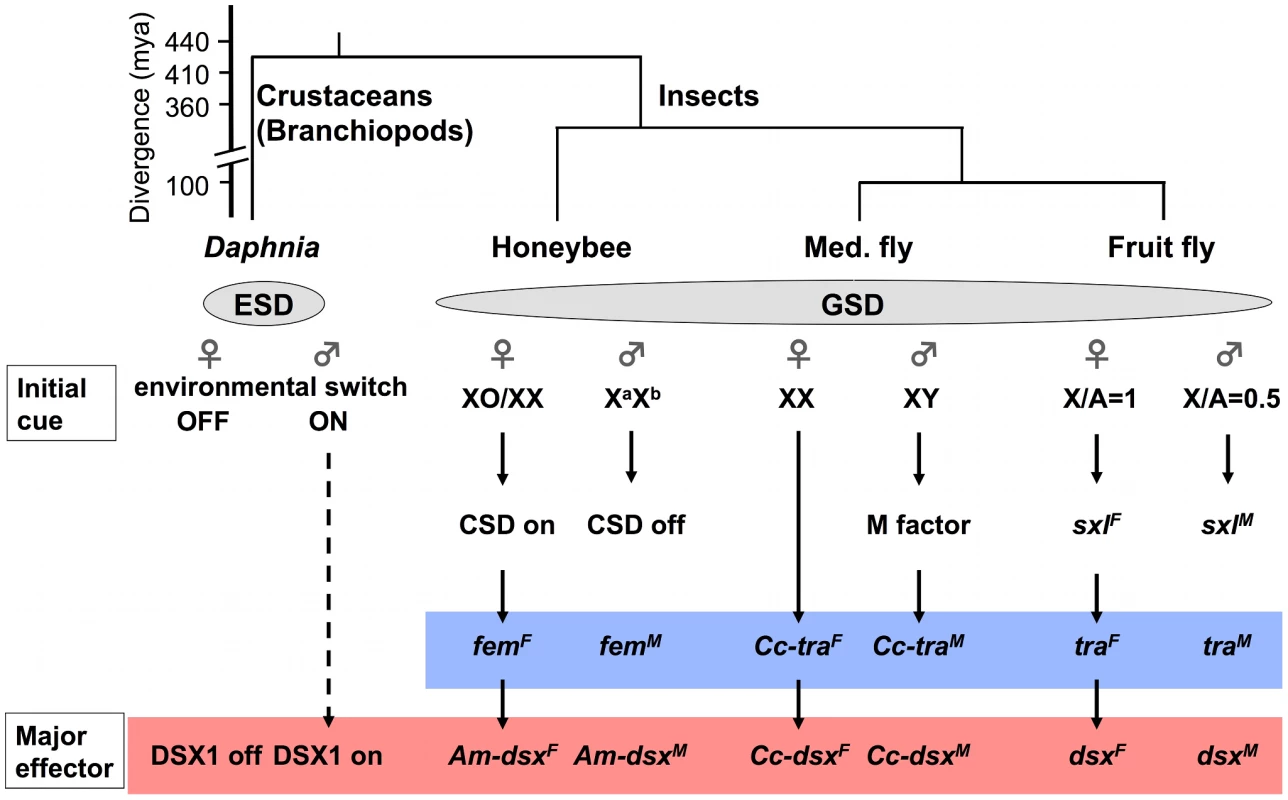
An ESD pathway in Daphnia is compared with GSD pathways in insect model species, honeybee (Apis mellifera [64]), Mediterranean fruit fly (Med. fly, Ceratitis capitata, [62]) and fruit fly (Drosophila melanogaster [78]). Conserved Doublesex and Transformer homologs are indicated with red and blue boxes, respectively. Phylogenetic relationships among the four species are shown above the pathways [42], [64], [71]. CSD, complementary sex determiner; fem, feminizer; Am, Apis mellifera: Cc, Ceratits capitata; sxl, sex lethal; mya, million years ago. Interestingly, expression of the DapmaDsx1 gene utilizes alternative polyadenylation at tandem poly(A) sites, which can yield transcripts that have identical protein-coding sequences but different 3'UTR sequences. Alternative polyadenylation is often associated with tissue - or developmental stage-dependent gene expression [69], [70]. We found that female DapmaDsx1-β mRNAs exclusively use the most promoter-proximal polyadenylation signals. The presence of alternative polyadenylation sites in Dsx genes has been reported in three insects, D. melanogaster, the phorid fly Megaselia scalaris and the mosquito Anopheles gambiae, indicating that regulation of the 3'UTR length might be a common mode to regulate expression of Dsx genes.
Despite having last shared a common ancestor with insects about 400 million years ago [42], [71] and differences of the initial cue to determine sex, the DapmaDsx1 maintained the domain structure essential for establishing sexual dimorphism, while regulation of its expression by other factors became complex and diverse. This is consistent with the prediction that new signals are co-opted upstream of a cascade during the course of evolution [72], [73]. Thus, we have established that there were no boundaries between GSD and ESD in evolution of sex-determining genes at their most fundamental level.
Materials and Methods
Daphnia Strain and Culture Conditions
The Daphnia magna strain (NIES clone) was obtained from the National Institute for Environmental Studies (NIES; Tsukuba, Japan) and maintained as described previously [25]. In order to obtain male embryos, adult D. magna (about 2 weeks of age) were treated with a synthetic juvenile hormone mimic, fenoxycarb (1 µg/L), and eggs ovulated into the brood chamber were collected.
Cloning of DSX-Like Genes
The amino acid sequence of the Drosophila melanogaster Dsx gene was retrieved from NCBI database (http://www.ncbi.nlm.nih.gov/) and used to search the D. magna EST database for related sequences. Two EST sequences were identified to have similarities with the Drosophila Dsx gene. The harvested daphnids were briefly washed and homogenized using the Physcotron NS-310E (Nichion, Tokyo, Japan). Total RNA was extracted with TRIzol reagent according to the manufacturer's protocol (Invitrogen, Carlsbad, CA, USA). Poly (A)+ RNA was isolated from purified total RNA using Fast Track (Invitrogen) and converted to cDNA using Superscript III and random primers (both Invitrogen) according to the manufacturer's protocol. cDNAs corresponding to the EST sequences were obtained by PCR, and full length cDNAs were obtained by RACE (Cap Fishing, SeeGene, Seoul, South Korea) using the oligonucleotide sequences shown in Table S1. Sequence data from this article have been deposited with the DDBJ/EMBL/Genbank Data Libraries under Accession No. AB569296, AB569297, AB569298.
Phylogenetic Analysis of the DM Domain Genes
A phylogenetic tree of DM domain genes including newly cloned D. magna Dsx genes was constructed using amino acid sequences of DM-domain genes used in the previous study [43] and insect Dsx genes listed in Table S2. A multiple alignment was constructed using Clustal W [74] with the following settings (pairwise alignment parameters: gap opening penalty 6.00, gap extension penalty 0.21, identity protein weight matrix; multiple alignment parameters: gap opening penalty 10.00, gap extension penalty 0.24, delay divergent cutoff 30%, gap separation distance 4). Phylogenetic reconstruction was performed using the p-distance algorithm and the neighbor-joining method implemented in MEGA version 4 [75]. The phylogenetic tree was rooted to vertebrate DMRT7 outgroups (mouse; NP_082008, bovine; NP_0010332710, data not shown).
Quantitative PCR
Embryos were obtained from D. magna at two weeks of age. Ovulation occurred just after molting and was assigned to be 0 h. The embryos were collected 18 h, 42 h and 72 h after ovulation. Gonads were isolated and specific mRNAs were quantified as described previously [43]. The oligonucleotide sequences for PCR were indicated in Table S3.
Whole Mount In Situ Hybridization
Templates for the probe preparation were synthesized by PCR using gene-specific primers containing the T7 polymerase promoter sequence at their 5'-ends (Table S4). DIG-labeled probes were prepared as described by Butler et al. [76] and subjected to alkaline hydrolysis. Whole mount in situ hybridization was performed as described by Sagawa et al. [17]. Both antisense and sense probes were used to confirm the specificity of staining.
Double-Strand RNA (dsRNA) Preparation
Double-stranded RNA was synthesized using the MEGAscript high yield transcription kit (Ambion, Austin, TX, USA). Templates were prepared by PCR using gene-specific primers with the T7 polymerase promoter sequence at their 5'-ends (Table S5). The synthesized RNAs were purified using phenol/chloroform. Following ethanol precipitation, the RNA was resuspended in DNase/RNase-free distilled water (Invitrogen, Tokyo, Japan) and annealed [41]. Sequences corresponding to each dsRNAs were shown in Figure S2. dsRNA lengths were: Dsx1-#1, 778 bp: Dsx1-#2, 579 bp: Dsx2-#1, 703 bp: Dsx2-#2 : 448 bp.
Synthesis of Capped and Polyadenylated mRNAs
DSX1-α, DSX1-β and DSX2 cDNAs were subcloned into pCS2 vector and used for RNA synthesis. To synthesize the control RNA, pEGFP-C1 vector was used. In vitro transcription with T7 RNA polymerase and poly-A tail addition were performed according to the manufacturers' protocol using commercial kits [mMESSAGE mMACHINE, and Poly(A) Tailing kit, respectively, Ambion]. Templates were prepared by PCR using primers corresponding to the 5' - and 3'-ends of the mRNA sequences. The T7 polymerase promoter sequence was attached to the 5' end of the forward primer.
Microinjection
Eggs were obtained from D. magna at two weeks of age just after the ovulation and placed in ice-cold M4 media [77] containing 40 mM sucrose (M4-sucrose). The synthesized dsRNA (1 mg/ml) containing 1 mM Chromeo 494 fluorescent dye (Active Motif Chromeon GmbH, Tegernheim, Germany) or mRNA was injected and incubated in a 96-well plate for appropriate times [41]. Equal molar amounts of DSX1-α, DSX1-β and Dsx2 mRNA were injected for the gain-of-function study. Injection volume was approximately 0.3 nL.
Microscopy
Embryos were dissected off the yolk, and photographed with a Zeiss Axioplan 2 Imaging microscope (Zeiss, Oberkochen, Germany). Adults and juveniles were observed and photographed using a Leica MZ APO dissecting microscope (Leica Microsystems Heidelberg GmbH, Mannheim, Germany). mRNA-injected juveniles were directly observed using an environmental scanning electron microscope (XL30 ESEM; Philips, Hillsboro, OR, USA).
Northern Blot Analysis
3 µg and 10 µg of male and female poly (A)+ RNA were used respectively. The RNAs were separated by electrophoresis on a 1.0% formaldehyde–agarose gel and then transferred to positively charged nylon membranes (Hybond-N+; GE Healthcare, Little Chalfont, England). RNA probes were prepared with a DIG RNA labeling kit (Roche Diagnostic GmbH, Manheim, Germany). Primers to amplify templates for the probe preparation were (5'-3'): forward: AAGAATTGTCCGTGGGGGCAC and reverse: TAATACGACTCACTATAGGGAAAGTTTGGTGTAGGGAG. The membranes were hybridized with DIG-labeled RNA probes for 11 hr at 68 °C with DIG easy hyb (Roche Diagnostic). DIG-labeled RNA was detected with an alkaline phosphatase-conjugated anti-DIG antibody using CDP star (Roche Diagnostic) according to the manufacture's protocol.
Supporting Information
Zdroje
1. MarinI
BakerBS
1998
The evolutionary dynamics of sex determination.
Science
281
1990
1994
2. ZarkowerD
2001
Establishing sexual dimorphism: conservation amidst diversity?
Nat Rev Genet
2
175
185
3. BullJJ
1985
Sex determining mechanisms: an evolutionary perspective.
Experientia
41
1285
1296
4. CrewsD
BullJJ
2009
Mode and tempo in environmental sex determination in vertebrates.
Semin Cell Dev Biol
20
251
255
5. KorpelainenH
1990
Sex-Ratios and conditions required for environmental sex determination in animals.
Biol Rev Camb Philos Soc
65
147
184
6. BullJJ
VogtRC
1979
Temperature-dependent sex Determination in turtles.
Science
206
1186
1188
7. OrganCL
JanesDE
2008
Evolution of sex chromosomes in Sauropsida.
Integ Comp Biol
48
512
519
8. WarnerDA
ShineR
2008
The adaptive significance of temperature-dependent sex determination in a reptile.
Nature
451
566-U565
9. BullJJ
1981
Evolution of environmental sex determination from genotypic sex determination.
Heredity
47
173
184
10. BulmerMG
BullJJ
1982
Models of polygenic sex determination and sex-ratio control.
Evolution
36
13
26
11. EpperF
BryantPJ
1983
Sex-specific control of growth and differentiation in the Drosophila genital disk, studied using a temperature-sensitive transformer-2 mutation.
Dev Biol
100
294
307
12. HodgkinJ
2002
Exploring the envelope: Systematic alteration in the sex-determination system of the nematode Caeraorhabditis elegans.
Genetics
162
767
780
13. ShoemakerCM
CrewsD
2009
Analyzing the coordinated gene network underlying temperature-dependent sex determination in reptiles.
Semin Cell Dev Biol
20
293
303
14. HebertPDN
1978
The population biology of Daphnia (Crustacea, Daphnidae).
Biol Rev
53
387
426
15. OlmsteadAW
LeBlancGA
2000
Effects of endocrine-active chemicals on the development of sex characteristics of Daphnia magna.
Environ Toxicol Chem
19
2107
2113
16. MitchellSE
2001
Intersex and male development in Daphnia magna.
Hydrobiologia
442
145
156
17. SagawaK
YamagataH
ShigaY
2005
Exploring embryonic germ line development in the water flea, Daphnia magna, by zinc-finger-containing VASA as a marker.
Gene Expr Patterns
5
669
678
18. ZaffagniniF
1987
Reproduction in Daphnia.
Mem Inst Ital Idrobiol
45
245
284
19. OjimaY
1958
A cytological study on the development and maturation of the parthenogenetic and sexual eggs of Daphnia pulex (Crustacea–Cladocera).
Kwansei Gakuen Univ Ann Stud
6
123
176
20. StrossRG
HillJC
1965
Diapause induction in Daphnia requires two stimuli.
Science
150
1462
1464
21. HebertPD
1987
Genotypic characteristics of cyclic parthenogens and their obligately asexual derivatives.
Experientia Suppl
55
175
195
22. OlmsteadAW
LeblancGA
2002
Juvenoid hormone methyl farnesoate is a sex determinant in the crustacean Daphnia magna.
J Exp Zool
293
736
739
23. OdaS
TatarazakoN
WatanabeH
MoritaM
IguchiT
2005
Production of male neonates in four cladoceran species exposed to a juvenile hormone analog, fenoxycarb.
Chemosphere
60
74
78
24. BantaAM
BrownLA
1929
Control of sex in cladocera. III. Localization of the critical period for control of sex.
Proc Natl Acad Sci U S A
15
71
81
25. TatarazakoN
OdaS
WatanabeH
MoritaM
IguchiT
2003
Juvenile hormone agonists affect the occurrence of male Daphnia.
Chemosphere
53
827
833
26. KatoY
KobayashiK
OdaS
TatarazakoN
WatanabeH
2010
Sequence divergence and expression of a transformer gene in the branchiopod crustacean, Daphnia magna.
Genomics
95
160
165
27. WatanabeH
TatarazakoN
OdaS
NishideH
UchiyamaI
2005
Analysis of expressed sequence tags of the water flea Daphnia magna.
Genome
48
606
609
28. ColbourneJK
SinganVR
GilbertDG
2005
wFleaBase: The Daphnia genome database.
BMC Bioinformatics
6
45
29. ShawJR
PfrenderME
EadsBD
KlaperR
CallaghanA
ColsonI
JansenB
GilbertD
ColbourneJK
2008
Daphnia as an emerging model for toxicological genomics.
Adv Exp Biol
2
165
219
327-328
30. ColbourneJK
PfrenderME
GilbertD
ThomasWK
TuckerA
2011
The ecoresponsive genome of Daphnia pulex.
Science
331
555
561
31. SchuttC
NothigerR
2000
Structure, function and evolution of sex-determining systems in Dipteran insects.
Development
127
667
677
32. ZarkowerD
2006
Somatic sex determination.
WormBook
1
12
33. SekidoR
Lovell-BadgeR
2009
Sex determination and SRY: down to a wink and a nudge?
Trends Genet
25
19
29
34. BurtisKC
BakerBS
1989
Drosophila doublesex gene controls somatic sexual differentiation by producing alternatively spliced mRNAs encoding related sex-specific polypeptides.
Cell
56
997
1010
35. VolffJN
ZarkowerD
BardwellVJ
SchartlM
2003
Evolutionary dynamics of the DM domain gene family in metazoans.
J Mol Evol
57
Suppl 1
S241
249
36. BayrerJR
ZhangW
WeissMA
2005
Dimerization of doublesex is mediated by a cryptic ubiquitin-associated domain fold - Implications for sex-specific gene regulation.
J Biol Chem
280
32989
32996
37. ShenMM
HodgkinJ
1988
mab-3, a gene required for sex-specific yolk protein expression and a male-specific lineage in C. elegans.
Cell
54
1019
1031
38. RaymondCS
ShamuCE
ShenMM
SeifertKJ
HirschB
1998
Evidence for evolutionary conservation of sex-determining genes.
Nature
391
691
695
39. RaymondCS
MurphyMW
O'SullivanMG
BardwellVJ
ZarkowerD
2000
Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation.
Genes Dev
14
2587
2595
40. WilliamsTM
CarrollSB
2009
Genetic and molecular insights into the development and evolution of sexual dimorphism.
Nature Rev Genet
10
883
883
41. KatoY
ShigaY
KobayashiK
TokishitaS
YamagataH
2011
Development of an RNA interference method in the cladoceran crustacean Daphnia magna.
Dev Genes Evol In press
42. GlennerH
ThomsenPF
HebsgaardMB
SorensenMV
WillerslevE
2006
The origin of insects.
Science
314
1883
1884
43. KatoY
KobayashiK
OdaS
ColbourneJK
TatarazakoN
2008
Molecular cloning and sexually dimorphic expression of DM-domain genes in Daphnia magna.
Genomics
91
94
101
44. RegierJC
ShultzJW
1997
Molecular phylogeny of the major arthropod groups indicates polyphyly of crustaceans and a new hypothesis for the origin of hexapods.
Mol Biol Evol
14
902
913
45. RegierJC
ShultzJW
KambicRE
2005
Pancrustacean phylogeny: hexapods are terrestrial crustaceans and maxillopods are not monophyletic.
Proc Biol Sci
272
395
401
46. MallattJ
WinchellCJ
2007
Ribosomal RNA genes and deuterostome phylogeny revisited: more cyclostomes, elasmobranchs, reptiles, and a brittle star.
Mol Phylogenet Evol
43
1005
1022
47. AverofM
AkamM
1995
Hox genes and the diversification of insect and crustacean body plans.
Nature
376
420
423
48. AverofM
CohenSM
1997
Evolutionary origin of insect wings from ancestral gills.
Nature
385
627
630
49. OdaS
TatarazakoN
WatanabeH
MoritaM
IguchiT
2005
Production of male neonates in Daphnia magna (Cladocera, Crustacea) exposed to juvenile hormones and their analogs.
Chemosphere
61
1168
1174
50. KatoY
KobayashiK
WatanabeH
IguchiT
2010
Introduction of foreign DNA into the water flea, Daphnia magna, by electroporation.
Ecotoxicology
19
589
592
51. MatsudaM
NagahamaY
ShinomiyaA
SatoT
MatsudaC
2002
DMY is a Y-specific DM-domain gene required for male development in the medaka fish.
Nature
417
559
563
52. YoshimotoS
OkadaE
UmemotoH
TamuraK
UnoY
2008
A W-linked DM-domain gene, DM-W, participates in primary ovary development in Xenopus laevis.
Proc Natl Acad Sci U S A
105
2469
2474
53. SmithCA
RoeszlerKN
OhnesorgT
CumminsDM
FarliePG
2009
The avian Z-linked gene DMRT1 is required for male sex determination in the chicken.
Nature
461
267
271
54. KettlewellJR
RaymondCS
ZarkowerD
2000
Temperature-dependent expression of turtle Dmrt1 prior to sexual differentiation.
Genesis
26
174
178
55. MurdockC
WibbelsT
2006
Dmrt1 expression in response to estrogen treatment in a reptile with temperature-dependent sex determination.
J Exp Zool B Mol Dev Evol
306
134
139
56. ShoemakerC
RamseyM
QueenJ
CrewsD
2007
Expression of Sox9, Mis, and Dmrt1 in the gonad of a species with temperature-dependent sex determination.
Dev Dyn
236
1055
1063
57. HildrethPE
1965
Doublesex, recessive gene that transforms both males and females of Drosophila into intersexes.
Genetics
51
659
678
58. OliverB
KimYJ
BakerBS
1993
Sex-lethal, master and slave: a hierarchy of germ-line sex determination in Drosophila.
Development
119
897
908
59. BakerBS
RidgeKA
1980
Sex and the single cell. I. On the action of major loci affecting sex determination in Drosophila melanogaster.
Genetics
94
383
423
60. RobinettCC
VaughanAG
KnappJM
BakerBS
2010
Sex and the single cell. II. There is a time and place for sex.
PLoS Biol
8
e1000365
doi:10.1371/journal.pbio.1000365
61. McKeownM
BeloteJM
BakerBS
1987
A molecular analysis of transformer, a gene in Drosophila melanogaster that controls female sexual differentiation.
Cell
48
489
499
62. PaneA
SalveminiM
Delli BoviP
PolitoC
SacconeG
2002
The transformer gene in Ceratitis capitata provides a genetic basis for selecting and remembering the sexual fate.
Development
129
3715
3725
63. HedigerM
HenggelerC
MeierN
PerezR
SacconeG
2010
Molecular characterization of the key switch F provides a basis for understanding the rapid divergence of the sex-determining pathway in the housefly.
Genetics
184
155
170
64. GempeT
HasselmannM
SchiottM
HauseG
OtteM
2009
Sex determination in honeybees: Two separate mechanisms induce and maintain the female pathway.
Plos Biol
7
e1000222
doi:10.1371/journal.pbio.1000222
65. FujiiT
ShimadaT
2007
Sex determination in the silkworm, Bombyx mori: A female determinant on the W chromosome and the sex-determining gene cascade.
Semin Cell Dev Biol
18
379
388
66. VerhulstEC
van de ZandeL
BeukeboomLW
2010
Insect sex determination: it all evolves around transformer.
Curr Opin Genet Dev
20
376
383
67. LiY
ZhangZ
RobinsonGE
PalliSR
2007
Identification and characterization of a juvenile hormone response element and its binding proteins.
J Biol Chem
282
37605
37617
68. GorrTA
RiderCV
WangHY
OlmsteadAW
LeBlancGA
2006
A candidate juvenoid hormone receptor cis-element in the Daphnia magna hb2 hemoglobin gene promoter.
Mol Cell Endocrinol
247
91
102
69. HughesTA
2006
Regulation of gene expression by alternative untranslated regions.
Trends in Genetics
22
119
122
70. JiZ
LeeJY
PanZH
JiangBJ
TianB
2009
Progressive lengthening of 3' untranslated regions of mRNAs by alternative polyadenylation during mouse embryonic development.
Proc Natl Acad Sci U S A
106
7028
7033
71. RonshaugenM
McGinnisN
McGinnisW
2002
Hox protein mutation and macroevolution of the insect body plan.
Nature
415
914
917
72. WilkinsAS
1995
Moving up the hierarchy - a hypothesis on the evolution of a genetic sex determination pathway.
Bioessays
17
71
77
73. NothigerR
SteinmannzwickyM
1985
A single principle for sex determination in insects.
Cold Spring Harb Symp Quant Biol
50
615
621
74. ThompsonJD
HigginsDG
GibsonTJ
1994
CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice.
Nucleic Acids Res
22
4673
4680
75. TamuraK
DudleyJ
NeiM
KumarS
2007
MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0.
Mol Biol Evol
24
1596
1599
76. ButlerK
ZornAM
GurdonJB
2001
Nonradioactive in situ hybridization to Xenopus tissue sections.
Methods
23
303
312
77. ElendtBP
BiasWR
1990
Trace nutrient deficiency in Daphnia magna cultured in standard medium for toxicity testing. Effects of the optimization of culture conditions on life history parameters of D. magna.
Water Res
24
1157
1167
78. PomiankowskiA
NothigerR
WilkinsA
2004
The evolution of the Drosophila sex-determination pathway.
Genetics
166
1761
1773
Štítky
Genetika Reprodukční medicína
Článek Genetic Regulation by NLA and MicroRNA827 for Maintaining Nitrate-Dependent Phosphate Homeostasis inČlánek c-di-GMP Turn-Over in Is Controlled by a Plethora of Diguanylate Cyclases and PhosphodiesterasesČlánek Viral Genome Segmentation Can Result from a Trade-Off between Genetic Content and Particle Stability
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 3- Růst a vývoj dětí narozených pomocí IVF
- Délka menstruačního cyklu jako marker ženské plodnosti
- Vztah užívání alkoholu a mužské fertility
- Akutní intermitentní porfyrie
- Intrauterinní inseminace a její úspěšnost
-
Všechny články tohoto čísla
- Whole-Exome Re-Sequencing in a Family Quartet Identifies Mutations As the Cause of a Novel Skeletal Dysplasia
- Origin-Dependent Inverted-Repeat Amplification: A Replication-Based Model for Generating Palindromic Amplicons
- Testing for an Unusual Distribution of Rare Variants
- Limited dCTP Availability Accounts for Mitochondrial DNA Depletion in Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE)
- FUS Transgenic Rats Develop the Phenotypes of Amyotrophic Lateral Sclerosis and Frontotemporal Lobar Degeneration
- Repeat Associated Non-ATG Translation Initiation: One DNA, Two Transcripts, Seven Reading Frames, Potentially Nine Toxic Entities!
- Initial Mutations Direct Alternative Pathways of Protein Evolution
- Dopamine Signalling in Mushroom Bodies Regulates Temperature-Preference Behaviour in
- Sensing of Replication Stress and Mec1 Activation Act through Two Independent Pathways Involving the 9-1-1 Complex and DNA Polymerase ε
- Genetic Regulation by NLA and MicroRNA827 for Maintaining Nitrate-Dependent Phosphate Homeostasis in
- Identification of a Novel Type of Spacer Element Required for Imprinting in Fission Yeast
- Chiasmata Promote Monopolar Attachment of Sister Chromatids and Their Co-Segregation toward the Proper Pole during Meiosis I
- Global Analysis of the Relationship between JIL-1 Kinase and Transcription
- H3K9me2/3 Binding of the MBT Domain Protein LIN-61 Is Essential for Vulva Development
- REVEILLE8 and PSEUDO-REPONSE REGULATOR5 Form a Negative Feedback Loop within the Arabidopsis Circadian Clock
- A Novel Unstable Duplication Upstream of Predisposes to a Breed-Defining Skin Phenotype and a Periodic Fever Syndrome in Chinese Shar-Pei Dogs
- Polycomb Repressive Complex 2 Controls the Embryo-to-Seedling Phase Transition
- A Role for Set1/MLL-Related Components in Epigenetic Regulation of the Germ Line
- Genome-Wide Association Analysis Identifies Variants Associated with Nonalcoholic Fatty Liver Disease That Have Distinct Effects on Metabolic Traits
- A Genome-Wide Association Study of Upper Aerodigestive Tract Cancers Conducted within the INHANCE Consortium
- Ancestral Mutation in Telomerase Causes Defects in Repeat Addition Processivity and Manifests As Familial Pulmonary Fibrosis
- Ultra-Deep Sequencing of Mouse Mitochondrial DNA: Mutational Patterns and Their Origins
- Phenotype Restricted Genome-Wide Association Study Using a Gene-Centric Approach Identifies Three Low-Risk Neuroblastoma Susceptibility Loci
- The Toll-Like Receptor Gene Family Is Integrated into Human DNA Damage and p53 Networks
- Polycomb Targets Seek Closest Neighbours
- Widespread Hypomethylation Occurs Early and Synergizes with Gene Amplification during Esophageal Carcinogenesis
- c-di-GMP Turn-Over in Is Controlled by a Plethora of Diguanylate Cyclases and Phosphodiesterases
- Estimating Divergence Time and Ancestral Effective Population Size of Bornean and Sumatran Orangutan Subspecies Using a Coalescent Hidden Markov Model
- Rif1 Supports the Function of the CST Complex in Yeast Telomere Capping
- A Tradeoff Drives the Evolution of Reduced Metal Resistance in Natural Populations of Yeast
- Quantifying the Underestimation of Relative Risks from Genome-Wide Association Studies
- Population-Based Resequencing of Experimentally Evolved Populations Reveals the Genetic Basis of Body Size Variation in
- Triplet Repeat–Derived siRNAs Enhance RNA–Mediated Toxicity in a Drosophila Model for Myotonic Dystrophy
- The FUN30 Chromatin Remodeler, Fft3, Protects Centromeric and Subtelomeric Domains from Euchromatin Formation
- Viral Genome Segmentation Can Result from a Trade-Off between Genetic Content and Particle Stability
- Environmental Sex Determination in the Branchiopod Crustacean : Deep Conservation of a Gene in the Sex-Determining Pathway
- Systematic Detection of Polygenic Regulatory Evolution
- The SUMO Isopeptidase Ulp2p Is Required to Prevent Recombination-Induced Chromosome Segregation Lethality following DNA Replication Stress
- Uncoupling Antisense-Mediated Silencing and DNA Methylation in the Imprinted Cluster
- Role of the Drosophila Non-Visual ß-Arrestin Kurtz in Hedgehog Signalling
- Differential Genetic Associations for Systemic Lupus Erythematosus Based on Anti–dsDNA Autoantibody Production
- COMPASS-Like Complexes Mediate Histone H3 Lysine-4 Trimethylation to Control Floral Transition and Plant Development
- H3 Lysine 4 Is Acetylated at Active Gene Promoters and Is Regulated by H3 Lysine 4 Methylation
- Diverse Roles and Interactions of the SWI/SNF Chromatin Remodeling Complex Revealed Using Global Approaches
- A Bow-Tie Genetic Architecture for Morphogenesis Suggested by a Genome-Wide RNAi Screen in
- Roles of () in Oocyte Nuclear Architecture, Gametogenesis, Gonad Tumors, and Genome Stability in Zebrafish
- A Molecular Phylogeny of Living Primates
- Roles of the Espin Actin-Bundling Proteins in the Morphogenesis and Stabilization of Hair Cell Stereocilia Revealed in CBA/CaJ Congenic Jerker Mice
- A Cholinergic-Regulated Circuit Coordinates the Maintenance and Bi-Stable States of a Sensory-Motor Behavior during Male Copulation
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Whole-Exome Re-Sequencing in a Family Quartet Identifies Mutations As the Cause of a Novel Skeletal Dysplasia
- Origin-Dependent Inverted-Repeat Amplification: A Replication-Based Model for Generating Palindromic Amplicons
- FUS Transgenic Rats Develop the Phenotypes of Amyotrophic Lateral Sclerosis and Frontotemporal Lobar Degeneration
- Limited dCTP Availability Accounts for Mitochondrial DNA Depletion in Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE)
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



