-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Genome-Wide Association Study Using Extreme Truncate Selection
Identifies Novel Genes Affecting Bone Mineral Density and Fracture
Risk
Osteoporotic fracture is a major cause of morbidity and mortality worldwide. Low
bone mineral density (BMD) is a major predisposing factor to fracture and is
known to be highly heritable. Site-, gender-, and age-specific genetic effects
on BMD are thought to be significant, but have largely not been considered in
the design of genome-wide association studies (GWAS) of BMD to date. We report
here a GWAS using a novel study design focusing on women of a specific age
(postmenopausal women, age 55–85 years), with either extreme high or low
hip BMD (age - and gender-adjusted BMD z-scores of +1.5 to +4.0,
n = 1055, or −4.0 to −1.5,
n = 900), with replication in cohorts of women drawn from
the general population (n = 20,898). The study replicates
21 of 26 known BMD–associated genes. Additionally, we report suggestive
association of a further six new genetic associations in or around the genes
CLCN7, GALNT3, IBSP, LTBP3, RSPO3, and
SOX4, with replication in two independent datasets. A novel
mouse model with a loss-of-function mutation in GALNT3 is also
reported, which has high bone mass, supporting the involvement of this gene in
BMD determination. In addition to identifying further genes associated with BMD,
this study confirms the efficiency of extreme-truncate selection designs for
quantitative trait association studies.
Published in the journal: . PLoS Genet 7(4): e32767. doi:10.1371/journal.pgen.1001372
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1001372Summary
Osteoporotic fracture is a major cause of morbidity and mortality worldwide. Low
bone mineral density (BMD) is a major predisposing factor to fracture and is
known to be highly heritable. Site-, gender-, and age-specific genetic effects
on BMD are thought to be significant, but have largely not been considered in
the design of genome-wide association studies (GWAS) of BMD to date. We report
here a GWAS using a novel study design focusing on women of a specific age
(postmenopausal women, age 55–85 years), with either extreme high or low
hip BMD (age - and gender-adjusted BMD z-scores of +1.5 to +4.0,
n = 1055, or −4.0 to −1.5,
n = 900), with replication in cohorts of women drawn from
the general population (n = 20,898). The study replicates
21 of 26 known BMD–associated genes. Additionally, we report suggestive
association of a further six new genetic associations in or around the genes
CLCN7, GALNT3, IBSP, LTBP3, RSPO3, and
SOX4, with replication in two independent datasets. A novel
mouse model with a loss-of-function mutation in GALNT3 is also
reported, which has high bone mass, supporting the involvement of this gene in
BMD determination. In addition to identifying further genes associated with BMD,
this study confirms the efficiency of extreme-truncate selection designs for
quantitative trait association studies.Introduction
Osteoporotic fracture is a leading cause of morbidity and mortality in the community, particularly amongst the elderly. In 2004 ten million Americans were estimated to have osteoporosis, resulting in 1.5 million fractures per annum [1]. Hip fracture is associated with a one year mortality rate of 36% in men and 21% in women [2]; and the burden of disease of osteoporotic fractures overall is similar to that of colorectal cancer and greater than that of hypertension and breast cancer [3]. Bone mineral density (BMD) is strongly correlated with bone strength and fracture risk, and its measurement is widely used as a diagnostic tool in the assessment of fracture risk [4]–[6]. BMD is known to be highly heritable, with heritability assessed in both young and elderly twins, and in families, to be 60–90% [7]–[14]. Although the extent of covariance between BMD and fracture risk is uncertain, of the 26 genes associated with BMD at genome-wide significant levels to date, nine have been associated with fracture risk (reviewed in [15]), supporting the use of BMD as an intermediate phenotype in the search for genes associated with fracture risk.
There is considerable evidence from genetic studies in humans [12], [16], [17], and in mice [18], indicating that the genes that influence BMD at different sites, and in the different genders, overlap but are not identical. Thus far all genome-wide association studies (GWAS) of BMD have studied cohorts of a wide age range, and with one exception have included both men and women; when only women have been studied, both pre - and postmenopausal women have been included. Therefore, to identify genes involved in osteoporosis in the demographic at highest risk of osteoporotic fracture we have performed a GWAS in postmenopausal women selected on the basis of their hip BMD, and replicated the GWAS findings in a large cohort of adult women drawn from the general population.
Results
Considering markers previously reported as associated with BMD, our discovery dataset replicates previously associated SNPs in 21 of the 26 genes reported to date to have genome-wide significant associations (Table S6) (P<0.05, association in the same direction as initially reported, or, in the case of LRP5 and GPR177, with the next flanking SNP genotyped) [17], [21], [22], [23], [28], [32], [33], [34]. Replicated genes include ARHGAP1, CTNNB1, ESR1, FAM3C, FLJ42280, FOXL1, GPR177, HDAC5, JAG1, LRP5, MARK3, MEF2C, MEPE, OPG, RANK, RANKL, SOST, SOX6, SP7 (Osterix), STARD3NL and ZBTB40. Considering the combined Anglo-Australasian Osteoporosis Genetics Consortium (AOGC) and deCODE/TwinsUK/Rotterdam cohorts, 97 SNPs from six loci achieved P<5×10−8 at the femoral neck (FN), of which four had previously been reported (FLJ42280, MEF2C, SOX6, ZBTB40). At the lumbar spine (LS), six SNPs from two known loci (RANKL, OPG) achieved P<5×10−8. No support was seen for previously reported associations involving SNPs in ADAMTS18, CRHR1, DCDC5, MHC, or SBTBN1 (P>0.05).
This study also identifies and replicates two novel loci with confirmed association with BMD in GALNT3 (MIM: 601756) and at chromosome 6q22 near RSPO3 (MIM: 610574), and provides strong evidence of a further four BMD-associated loci (CLCN7 (MIM: 602727), IBSP (MIM: 147563), LTBP3 (MIM: 602090), SOX4 (MIM: 184430)) (Table 1). Although these did not achieve ‘genome-wide significance’ in the discovery set alone, they achieved P-values in the AOGC-discovery cohort of P<10−4, and support in the AOGC-replication cohort, TwinsUK, Rotterdam and deCODE cohorts; and all have additional evidence supporting their role in bone. Support was also seen for TGFBR3 (MIM: 600742), a gene previously reported to have suggestive association with BMD [33].
Tab. 1. Findings for novel replicated associations. 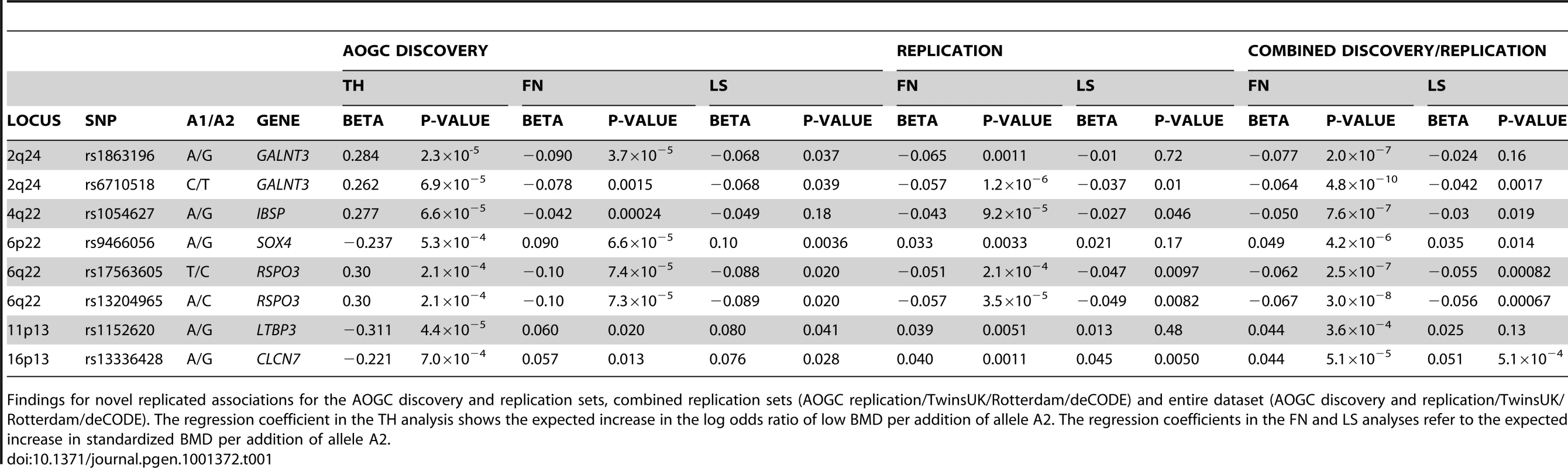
GALNT3
SNPs at chromosome 2q24, in and around GALNT3, achieved near genome-wide significance in our discovery cohort (peak P-value rs1863196, total hip (TH) P = 2.3×10−5; LS P = 0.037) (Figure 1A). This SNP was not typed or imputed by either the Rotterdam or the TwinsUK cohorts, but a nearby SNP showed strong association in both AOGC and the combined replication cohorts (rs6710518; AOGC discovery, TH P = 6.9×10−5; combined replication sets, FN P = 2.7×10−7). In the combined datasets the finding achieved genome-wide significance at the FN (P = 1.7×10−10). Strong association was also seen with this SNP at LS (P = 7.5×10−5). Another marker within GALNT3, rs4667492, was also associated with fracture risk, including vertebral fractures (OR = 0.89; 95%CI = 0.80–0.99; P = 0.032) and overall low trauma fractures (OR = 0.92; 95%CI = 0.85–0.99; P = 0.024).
Fig. 1. SNP association plots for BMD-associated regions. 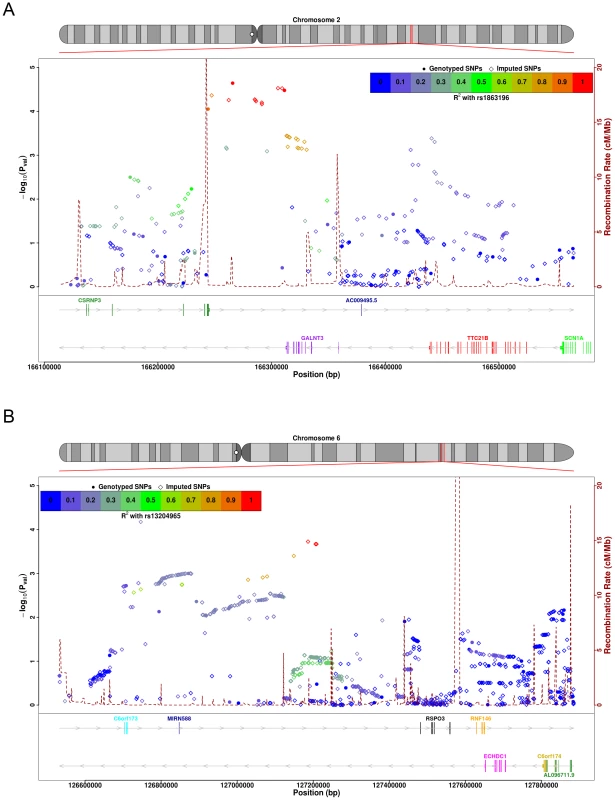
We have recently identified a mouse with an N-ethyl-N-nitrosourea induced loss-of-function GALNT3 mutation (Trp589Arg), that develops hyperphosphataemia with extraskeletal calcium deposition, and hence represents a model for FTC [35]. To establish further the association of GALNT3 and BMD, we determined BMD in these GALNT3 mutant mice. This revealed that homozygous (−/−) GALNT3 mutant male and female adult mice had a higher areal BMD than their wild-type (+/+) litter mates, with heterozygous (+/−) mice having intermediate BMD (Figure 2). This loss-of-function GALNT3 mutation is predicted to lead to a reduced glycosylation of FGF23, which increases its breakdown and leads to reduced serum FGF23 concentrations [35].
RSPO3
A novel genome-wide significant association was also seen at markers on chromosome 6q22-23 (Figure 1B). In the combined dataset, marker rs13204965 achieved genome-wide significance at this locus at the FN (P = 2.2×10−9), with strong support in both the AOGC discovery set, and the combined replication sets (AOGC-discovery, TH P = 2.1×10−4; combined replication P = 3.5×10−5). Strong association was also seen with LS BMD (rs13204965 P = 0.00067). The peak of association at this locus lies within a cDNA fragment, AK127472. The nearest gene, RSPO3 (R-spondin-3), is 275 kb telomeric of the strongest associated SNP, but is within the associated linkage disequilibrium region (Figure 1B).
CLCN7
Association was observed at chromosome 16p13 with SNPs in and around CLCN7, which encodes a Cl−/H+ antiporter expressed primarily in osteoclasts, and critical to lysosomal acidification, an essential process in bone resorption. Peak association at this locus was seen with SNP rs13336428 in the discovery set (TH P = 7.0×10−4; LS P = 0.028) (Figure S3A), which was confirmed in the replication set (FN P = 3.6×10−5; LS P = 0.00012), achieving P = 1.7×10−6 at the FN and 1.2×10−5 at LS in the overall cohort. Association has previously been reported between two SNPs in exon 15 of CLCN7 (rs12926089, rs12926669) and FN BMD (P = 0.001–0.003) [36]; no association was seen with either of those SNPs in the current study (P>0.4 at FN and LS).
IBSP
Association was observed with SNPs in IBSP (integrin-binding bone sialoprotein) (Figure S3B), encoded at chromosome 4q22, a gene which has previously had suggestive association reported with BMD in two studies (rs1054627, Styrkarrsdottir et al P = 4.6×10−5 [22]; Koller et al P = 1.5×10−4 [37]). In the current study, moderate association was observed in the discovery set with the same SNP as previously reported (rs1054627, AOGC discovery TH, P = 6.6×10−5), with support in the replication set and strong association overall (FN combined replication P = 9.2×10−5; FN overall association P = 7.6×10−7). Nominal association was observed at LS (rs1054627, P = 0.019).
LTBP3
Association with BMD was also seen at chromosome 11p13, with SNP rs1152620 achieving P = 4.4×10−5 (TH) in the discovery set, P = 0.0051 (FN) in the replication set, and P = 3.6×10−4 overall (Figure S3C). This SNP was also nominally associated with LS BMD in the discovery set (P = 0.041). The nearest gene to this locus is LTBP3 (latent transforming growth factor beta binding protein 3), which is located 292 kb q-telomeric of rs1152620.
SOX4
At chromosome 6p22, SNPs in and around SOX4 (Sex determining region Y box 4) were moderately associated with BMD in our discovery set (most significant association rs9466056, TH P = 5.3×10−4; LS P = 0.0036) (Figure S3D), with support at the hip and LS in the replication set (FN P = 0.00013, LS P = 0.013), achieving association overall with P = 2.6×10−7 (FN) and P = 0.00081 (LS).
Discussion
This study demonstrates convincing evidence of association with six genes with BMD variation, GALNT3, RSPO3, CLCN7, IBSP, LTBP3 and SOX4. Using a moderate sample size, the use of a novel study design also led to the confirmation of 21 of 26 known BMD-associations. This study thus demonstrates the power of extreme-truncate selection designs for association studies of quantitative traits.
GALNT3 encodes N-acetylgalactosaminyltransferase 3, an enzyme involved in 0-glycosylation of serine and threonine residues. Mutations of GALNT3 are known to cause familial tumoral calcinosis (FTC, OMIM 2111900) [38] and hyperostosis-hyperphosphataemia syndrome (HOHP, OMIM 610233) [39]. FTC is characterised by hyperphosphataemia in association with the deposition of calcium phosphate crystals in extraskeletal tissues; whereas in HOHP, hyperphosphataemia is associated with recurrent painful long bone swelling and radiographic evidence of periosteal reaction and cortical hyperostosis. FGF23 mutations associated with FTC cause hyperphosphataemia through effects on expression of the sodium-phosphate co-transporter in the kidney and small intestine, and through increased activation of vitamin D due to increased renal expression of CYP27B1 (25-hydroxyvitamin-D 1 alpha hydroxylase) [40]. It is unclear whether FGF23 has direct effects on the skeleton or if its effects are mediated through its effects on serum phosphate and vitamin D levels. FGF23 signals via a complex of an FGF receptor (FGFR1(IIIc)) and Klotho [41]; mice with a loss-of-function mutation in Klotho develop osteoporosis amongst other abnormalities, and modest evidence of association of Klotho with BMD has been reported in several studies [42], [43], [44], [45]. We saw no association with polymorphisms in Klotho and BMD in the current study (P>0.05 for all SNPs in and surrounding Klotho). To our knowledge, this finding is the first demonstration in humans that genetic variants in the FGF23 pathway are associated with any common human disease.
RSPO3 is one of four members of the R-spondin family (R-spondin-1 to −4), which are known to activate the Wnt pathway, particularly through effects on LRP6, itself previously reported to be BMD-associated [46], [47]. LRP6 is inhibited by the proteins Kremen and DKK1, which combine to induce endocytosis of LRP6, reducing its cell surface levels. R-spondin family members have been shown to disrupt DKK1-dependent association of LRP6 and Kremen, thereby releasing LRP6 from this inhibitory pathway [48]. R-spondin-4 mutations cause anonychia (absence or severe hypoplasia of all fingernails and toenails, OMIM 206800) [49]. No human disease has been associated with R-spondin-3, and knockout of R-spondin-3 in mice is embryonically lethal due to defective placental development [50].
Mutations of CLCN7 cause a family of osteopetroses of differing age of presentation and severity, including infantile malignant CLCN7-related recessive osteopetrosis (ARO), intermediate autosomal osteopetrosis (IAO), and autosomal dominant osteopetrosis type II (ADOII, Albers-Schoenberg disease). These conditions are characterized by expanded, dense bones, with markedly reduced bone resorption. Our data support associations of polymorphisms at this locus with BMD variation in the population.
IBSP is a major non-collagenous bone matrix protein involved in calcium and hydroxyapatite binding, and is thought to play a role in cell-matrix interactions through RGD motifs in its amino acid sequence. IBSP is expressed in all major bone cells including osteoblasts, osteocytes and osteoclasts; and its expression is upregulated in osteoporotic bone [51]. IBSP knockout mice have low cortical but high trabecular bone volume, with impaired bone formation, resorption, and mineralization [52]. IBSP lies within a cluster of genes including DMP1, MEPE, and SPP1, all of which have known roles in bone and are strong candidate genes for association with BMD. MEPE has previously been associated with BMD at genome-wide significance [17]. In the current study the strongest association was seen with an SNP in IBSP, rs1054627, as was the case with two previous studies [22], [37]. Linkage disequilibrium between this SNP, and the previously reported BMD-associated SNP rs1471403 in MEPE, is modest (r2 = 0.16). Whilst out study supports the association of common variants in IBSP in particular with BMD, further studies will be required to determine if more than one of these genes is BMD-associated.
Recessive mutations of LTBP3 have been identified as the cause of dental agenesis in a consanguineous Pakistani family (OMIM 613097) [53]. Affected family members had base of skull thickening, and elevated axial but not hip BMD. LTBP3−/− mice develop axial osteosclerosis with increased trabecular bone thickness, as well as craniosynostosis [54]. LTBP3 is known to bind TGFβ1, -β2 and -β3, and may influence chondrocyte maturation and enchondral ossification by effects on their bioavailability [54].
Our study also confirms the previously reported association of another TGF pathway gene, TGFBR3, encoded at chromosome 1p22, with BMD [33] (Figure S3E). In that study, association was observed in four independent datasets, but overall the findings did not achieve genome-wide significance at any individual SNP (most significant SNP rs17131547, P = 1.5×10−6). In our discovery set, peak association was seen at this locus with SNP rs7550034 (TH P = 1.5×10−4), which lies 154 kb q-telomeric of rs17131547, but still within TGFBR3 (rs17131547 was not typed or imputed in our dataset) (Figure S3E). This supports TGFBR3 as a true BMD-associated gene.
This study also demonstrated that SOX4 polymorphisms are associated with BMD variation. Both SOX4 and SOX6 are cartilage-expressed transcription factors known to play essential roles in chondrocyte differentiation and cartilage formation, and hence endochondral bone formation. SOX6 has previously been reported to be BMD-associated at genome-wide significant levels [17]. Whilst SOX4−/ − mice develop severe cardiac abnormalities and are non-viable, SOX4+/− mice have osteopaenia with reduced bone formation but normal resorption rates, and diminished cortical and trabecular bone volume [55]. Our data suggest that SOX4 polymorphisms contribute to the variation in BMD in humans.
This study has a unique design amongst GWAS of BMD reported to date, using an extreme-truncate ascertainment scheme, focusing on a specific skeletal site (TH), and with recruitment of a narrow age - and gender-group (post-menopausal women age 55–85 years). Our goal in employing this scheme was to increase the study power by reducing heterogeneity due to age-, gender - and skeletal site-specific effects. Whilst osteoporotic fracture can occur at a wide range of skeletal sites, hip fracture in postmenopausal women is the major cause of morbidity and mortality due to osteoporosis. To date, with only one exception, all GWAS of BMD have studied cohorts unselected for BMD [28], and no study has restricted its participants to postmenopausal women ascertained purely on the basis of hip BMD. Assuming marker-disease-associated allele linkage disequilibrium of r2 = 0.9, for alpha = 5×10−8 our study has 80% power to detect variants contributing 0.3% of the additive genetic variance of BMD. An equivalent-powered cohort study would require ∼16,000 unselected cases.
Considering the 26 known genes (or genomic areas) associated with BMD, P-values less than <0.05 were seen in our discovery for 21 of the BMD-associated SNPs. Of the 26 known BMD genes, 16 would have been included in our replication study on the basis of the strength of their BMD association in our discovery cohort, but were not further genotyped as they were known already to be BMD-associated. Had these 16 genes replicated, 22 genes would have been identified in this single study, demonstrating the power of the design of the current study.
A potential criticism of studies of highly selected cohorts, such as the AOGC-discovery cohort, is that the associations identified may not be relevant in the general population. However, the confirmation of our findings in replication cohorts of women unselected for BMD confirms that our findings are of broad relevance.
In summary, our study design therefore represents a highly efficient model for future studies of quantitative traits and is one of the first reported studies using an extreme truncate design in any disease. We have identified two new BMD loci at genome-wide significance (GALNT3, RSPO3), with GALNT3 SNPs also associated with fracture. Strong evidence was also demonstrated for four novel loci (CLCN7, IBSP, LTBP3, SOX4). Further support was also provided that TGFBR3 is a true BMD-associated locus. Our discovery cohort replicated 21 of 26 previously identified BMD-associated loci. Our novel findings further advance our understanding of the aetiopathogenesis of osteoporosis, and highlight new genes and pathways not previously considered important in BMD variation and fracture risk in the general population. Our study also provides strong support that the use of extreme truncate selection is an efficient and powerful approach for the study of quantitative traits.
Materials and Methods
Ethics statement
All participants gave written, informed consent, and the study was approved by the relevant research ethics authorities at each participating centre.
Subjects and phenotypes
The discovery sample population included 1128 Australian, 74 New Zealand and 753 British women, between 55–85 years of age, five or more years postmenopausal, with either high BMD (age - and gender-adjusted BMD z-scores of +1.5 to +4.0, n = 1055) or low BMD (age - and gender-adjusted BMD z-scores of −4.0 to −1.5, n = 900) (Tables S1 and S2). BMD z-scores were determined according to the Geelong Osteoporosis Study normative range [19]. Low BMD cases were excluded if they had secondary causes of osteoporosis, including corticosteroid usage at doses equivalent to prednisolone ≥7.5 mg/day for ≥6 months, past or current anticonvulsant usage, previous strontium usage, premature menopause (<45 years), alcohol excess (>28 units/week), chronic renal or liver disease, Cushing's syndrome, hyperparathyroidism, thyrotoxicosis, anorexia nervosa, malabsorption, coeliac disease, rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease, osteomalacia, and neoplasia (cancer, other than skin cancer). Screening blood tests (including creatinine (adjusted for weight), alkaline phosphatase, gamma-glutamyl transferase, 25-hydroxyvitamin D and PTH) were checked in 776 cases, and no differences were found between the high and low BMD groups. Therefore no further screening tests were done of the remaining cases.
Fracture data were analysed comparing individuals who had never reported a fracture after the age of 50 years, with individuals who had had a low or non-high trauma (low trauma fracture = fracture from a fall from standing height or less) osteoporotic fracture (excluding skull, nose, digits, hand, foot, ankle, patella) after the age of 50 years. Vertebral, hip and non-vertebral fractures were considered both independently and combined.
All participants were of self-reported white European ancestry.
DNA was obtained from peripheral venous blood from all cases except those recruited from New Zealand, for whom DNA was obtained from salivary samples using Oragene kits (DNA Genotek, Ontario, Canada). We have previously demonstrated that DNA from these two sources have equivalent genotyping characteristics [20].
After quality control checks including assessment of cryptic relatedness, ethnicity and genotyping quality, 900 individuals with low TH BMD and 1055 individuals with high TH BMD were available for analysis.
The replication cohort consisted of 8928 samples drawn from nine cohort studies, outlined in Tables S3 and S4 (‘AOGC replication cohort’) which were directly genotyped, These replication cases were adult women (age 20–95 years), unselected with regard to BMD, and who were not screened for secondary causes of osteoporosis. Replication was also performed in silico in 11,970 adult women from the TwinsUK and Rotterdam, and deCODE Genetics GWASs [21], [22], [23], in which association data were available at LS and FN.
High and low BMD ascertainment was defined according to the TH score, because this has better measurement precision than FN BMD [24]. However, neither TwinsUK nor the Rotterdam Study had TH BMD on the majority of their datasets and therefore were analysed using the FN measurement for which data were available on the whole cohort. All replication findings at the hip are reported therefore for FN BMD. TH and FN BMD are closely correlated (r = 0.882 in the AOGC dataset), with FN BMD one of the components of the TH BMD measurement.
Genotyping
Genotyping of the discovery cohort (n = 2036) was performed using Illumina Infinium II HumHap300 (n = 140), 370CNVDuo (n = 4), 370CNVQuad (n = 1882) and 610Quad (n = 10) chips at the University of Queensland Diamantina Institute, Brisbane, Australia. Genotype clustering was performed using Illumina's BeadStudio software; all SNPs with quality scores <0.15 and all individuals with <98% genotyping success were excluded. 289499 SNPs were shared across all chip types. Cluster plots from the 500 most strongly associated loci, were manually inspected and poorly clustering SNPs excluded from analysis. Following imputation using the HapMap Phase 2 data, 2,543,887 SNPs were tested for association with TH and LS BMD (Manhattan plot of association findings, Figure S1). After data cleaning, minimal evidence of inflation of test statistics was observed, with a genomic inflation factor (λ) of 1.0282 (qq plot, Figure S2).
A total of 124 SNPs were successfully genotyped in the AOGC replication cohort. These replication study SNPs were selected from the findings of the discovery cohort, either based on the strength of association (P-value) or following analysis with GRAIL (n = 45) [25], using as seed data all SNPs previously reported to be associated with BMD at GWAS significant levels (results for all replication SNPs presented in Table S5). GRAIL is a bioinformatic program that assesses the strength of relationships between genes in regions surrounding input SNPs (usually derived from genetic association studies) and other SNPs or genes associated with the trait of interest, by assessing their co-occurrence in PubMed abstracts. Where genes surrounding input SNPs occur more frequently in abstracts with known associated genes, these SNPs are more likely themselves also to be associated, and can thus be prioritized for inclusion in replication studies.
For the replication study, genotyping was performed either by Applied Biosystems OpenArray (n = 113) or Taqman technology (n = 11) (Applied Biosystems, Foster City, CA, USA), according to the manufacturer's protocol.
Statistical methods
Eleven individuals were removed because of abnormal X-chromosome homozygosity (X-chromosome homozygosity either <−0.14, or >+0.14). Outliers with regard to autosomal heterozygosity (either <0.34225 or >0.357, n = 40) and missingness (>3%, n = 4) were removed. Using an IBS/IBD analysis in PLINK to detect cryptic relatedness, one individual from 35 pairs of individuals with pi-hat >0.12 (equivalent to being 3rd degree relatives or closer) were removed. SNPs with minor allele frequency <1% (n = 561), and those not in Hardy-Weinberg equilibrium (P<10−7, n = 170) were then removed, leaving 288,768 SNPs in total. Nine replication SNPs were removed because of excess missingness (>10%) or because they failed tests of Hardy-Weinberg equilibrium (P<0.001).
To detect and correct for population stratification EIGENSTRAT software was used. We first excluded the 24 regions of long range LD including the MHC identified in Price et al. before running the principal components analysis, as suggested by the authors [26]. Sixteen individuals were removed as ethnic outliers, leaving 1955 individuals in the final discovery dataset.
Imputation analyses were carried out using Markov Chain Haplotyping software (MaCH; http://www.sph.umich.edu/csg/abecasis/MACH/) using phased data from CEU individuals from release 22 of the HapMap project as the reference set of haplotypes. We only analyzed SNPs surrounding disease-associated SNPs that were either genotyped or could be imputed with relatively high confidence (R2≥0.3). For TH measurements, a case-control association analysis of imputed SNPs was performed assuming an underlying additive model and including four EIGENSTRAT eigenvectors as covariates, using the software package MACH2DAT [27] which accounts for uncertainty in prediction of the imputed data by weighting genotypes by their posterior probabilities. For FN and LS BMD analyses, Z-transformed residual BMD scores (in g/cm2) were generated for the entire AOGC cohort after adjusting for the covariates age, age2, and weight, and for centre of BMD measurement. Because the regression coefficient for BMD on genotype would be biased by selection for extremes, we adopted the approach detailed in Kung et al (2009) [28]. Specifically, the regression coefficient of genotype on BMD was estimated, and subsequently transformed to the regression coefficient of BMD on genotype through knowledge of the population variance of the phenotype and the allele frequencies. For fracture data, analysis was by logistic regression. Only SNPs achieving GWAS significance were tested for fracture association. The SNPs used for replication from the Rotterdam Study were analyzed using MACH2QTL implemented in GRIMP [29]. Data from the discovery and replication cohorts were combined using the inverse variance approach as implemented in the program METAL [30].
SNPs associated with BMD were also tested for association with fracture in the AOGC discovery and replication cohorts (hip, vertebral, nonvertebral, and all low trauma fractures, age ≥50 years, as defined above), by logistic regression.
Study power was calculated using the ‘Genetic Power Calculator’ [31].
Mouse BMD analysis
All animal studies were approved by the MRC Harwell Unit Ethical Review Committee and are licensed under the Animal (Scientific Procedures) Act 1986, issued by the UK Government Home Office Department. Dual-energy X-ray absorptiometry (DEXA) was performed using a Lunar Piximus densitometer (GE Medical Systems) and analysed using the Piximus software.
Data availability
Data related to this study will be available to research projects approved by a Data Access Committee including representatives of the University of Queensland Research Ethics Committee. For enquiries regarding access please contact the corresponding author, MAB (matt.brown@uq.edu.au).
Supporting Information
Zdroje
1. US Department of Health and Human Services
2004
Bone health and Osteoporosis: a report of the surgeon
general.
Rockville, MD, USA
2. US Department of Commerce
1993
Hip fracture rates in people aged fifty years and over:
mortality, service use, expenditures, and long-term functional
impairment.
Washington, DC
3. Johnell
O
Kanis
JA
2006
An estimate of the worldwide prevalence and disability associated
with osteoporotic fractures.
Osteoporos Int
17
1726
1733
4. Kanis
JA
Johnell
O
Oden
A
Johansson
H
McCloskey
E
2008
FRAX and the assessment of fracture probability in men and women
from the UK.
Osteoporos Int
19
385
397
5. Henry
MJ
Pasco
JA
Seeman
E
Nicholson
GC
Sanders
KM
2001
Assessment of fracture risk: value of random population-based
samples—the Geelong Osteoporosis Study.
J Clin Densitom
4
283
289
6. Nguyen
ND
Pongchaiyakul
C
Center
JR
Eisman
JA
Nguyen
TV
2005
Identification of high-risk individuals for hip fracture: a
14-year prospective study.
J Bone Miner Res
20
1921
1928
7. Arden
NK
Baker
J
Hogg
C
Baan
K
Spector
TD
1996
The heritability of bone mineral density, ultrasound of the
calcaneus and hip axis length: a study of postmenopausal
twins.
J Bone Miner Res
11
530
534
8. Arden
NK
Spector
TD
1997
Genetic influences on muscle strength, lean body mass, and bone
mineral density: a twin study.
J Bone Miner Res
12
2076
2081
9. Dequeker
J
Nijs
J
Verstraeten
A
Geusens
P
Gevers
G
1987
Genetic determinants of bone mineral content at the spine and
radius: a twin study.
Bone
8
207
209
10. Harris
M
Nguyen
TV
Howard
GM
Kelly
PJ
Eisman
JA
1998
Genetic and environmental correlations between bone formation and
bone mineral density: a twin study.
Bone
22
141
145
11. Nguyen
TV
Howard
GM
Kelly
PJ
Eisman
JA
1998
Bone mass, lean mass, and fat mass: same genes or same
environments?
Am J Epidemiol
147
3
16
12. Duncan
E
Cardon
L
Sinsheimer
J
Wass
J
Brown
M
2003
Site and Gender Specificity of Inheritance of Bone Mineral
Density.
J Bone Miner Res
18
1531
1538
13. Sigurdsson
G
Halldorsson
BV
Styrkarsdottir
U
Kristjansson
K
Stefansson
K
2008
Impact of genetics on low bone mass in adults.
J Bone Miner Res
23
1584
1590
14. Flicker
L
Hopper
J
Rodgers
L
Kaymakci
B
Green
R
1995
Bone density determinants in elderly women: a twin
study.
J Bone Miner Res
10
1607
1613
15. Duncan
EL
Brown
MA
2010
Clinical review 2: Genetic determinants of bone density and
fracture risk—state of the art and future directions.
J Clin Endocrinol Metab
95
2576
2587
16. Naganathan
V
Macgregor
A
Snieder
H
Nguyen
T
Spector
T
2002
Gender differences in the genetic factors responsible for
variation in bone density and ultrasound.
J Bone Miner Res
17
725
733
17. Rivadeneira
F
Styrkarsdottir
U
Estrada
K
Halldorsson
BV
Hsu
YH
2009
Twenty bone-mineral-density loci identified by large-scale
meta-analysis of genome-wide association studies.
Nat Genet
41
1199
1206
18. Orwoll
ES
Belknap
JK
Klein
RF
2001
Gender specificity in the genetic determinants of peak bone
mass.
J Bone Miner Res
16
1962
1971
19. Henry
MJ
Pasco
JA
Nicholson
GC
Seeman
E
Kotowicz
MA
2000
Prevalence of osteoporosis in Australian women: Geelong
Osteoporosis Study.
J Clin Densitom
3
261
268
20. Bahlo
M
Stankovich
J
Danoy
P
Hickey
PF
Taylor
BV
2010
Saliva-Derived DNA Performs Well in Large-Scale, High-Density
Single-Nucleotide Polymorphism Microarray Studies.
Cancer Epidemiol Biomarkers Prev
19
794
798
21. Richards
JB
Rivadeneira
F
Inouye
M
Pastinen
TM
Soranzo
N
2008
Bone mineral density, osteoporosis, and osteoporotic fractures: a
genome-wide association study.
Lancet
371
1505
1512
22. Styrkarsdottir
U
Halldorsson
BV
Gretarsdottir
S
Gudbjartsson
DF
Walters
GB
2009
New sequence variants associated with bone mineral
density.
Nat Genet
41
15
17
23. Styrkarsdottir
U
Halldorsson
BV
Gretarsdottir
S
Gudbjartsson
DF
Walters
GB
2008
Multiple genetic loci for bone mineral density and
fractures.
N Engl J Med
358
2355
2365
24. Shepherd
JA
Fan
B
Lu
Y
Lewiecki
EM
Miller
P
2006
Comparison of BMD precision for Prodigy and Delphi spine and
femur scans.
Osteoporos Int
17
1303
1308
25. Raychaudhuri
S
Plenge
RM
Rossin
EJ
Ng
AC
Purcell
SM
2009
Identifying relationships among genomic disease regions:
predicting genes at pathogenic SNP associations and rare
deletions.
PLoS Genet
5
e1000534
doi:10.1371/journal.pgen.1000534
26. Price
AL
Weale
ME
Patterson
N
Myers
SR
Need
AC
2008
Long-range LD can confound genome scans in admixed
populations.
Am J Hum Genet
83
132
135; author reply 135-139
27. Li
Y
Willer
CJ
Sanna
S
Abecasis
GR
2009
Genotype imputation.
Annu Rev Genomics Hum Genet
10
387
406
28. Kung
AWC
Xiao
S-M
Cherny
S
Li
GHY
Gao
Y
2010
Assocation of JAG1 with bone mineral density and osteoporotic
fractures: a genome-wide association study and follow-up replication
studies.
Am J Hum Genet
86
1
11
29. Estrada
K
Abuseiris
A
Grosveld
FG
Uitterlinden
AG
Knoch
TA
2009
GRIMP: a web - and grid-based tool for high-speed analysis of
large-scale genome-wide association using imputed data.
Bioinformatics
25
2750
2752
30. Willer
CJ
Sanna
S
Jackson
AU
Scuteri
A
Bonnycastle
LL
2008
Newly identified loci that influence lipid concentrations and
risk of coronary artery disease.
Nat Genet
40
161
169
31. Purcell
S
Cherny
SS
Sham
PC
2003
Genetic Power Calculator: design of linkage and association
genetic mapping studies of complex traits.
Bioinformatics
19
149
150
32. Cho
YS
Go
MJ
Kim
YJ
Heo
JY
Oh
JH
2009
A large-scale genome-wide association study of Asian populations
uncovers genetic factors influencing eight quantitative
traits.
Nat Genet
41
527
534
33. Xiong
DH
Liu
XG
Guo
YF
Tan
LJ
Wang
L
2009
Genome-wide association and follow-up replication studies
identified ADAMTS18 and TGFBR3 as bone mass candidate genes in different
ethnic groups.
Am J Hum Genet
84
388
398
34. Timpson
NJ
Tobias
JH
Richards
JB
Soranzo
N
Duncan
EL
2009
Common variants in the region around Osterix are associated with
bone mineral density and growth in childhood.
Hum Mol Genet
18
1510
1517
35. Esapa
C
Head
R
Chan
E
Crane
M
Cheeseman
M
2009
A mouse with a Trp589Arg mutation in
N-acetylgalactosaminyltransferase 3 (Galnt3) provides a model for familial
tumoural calcinosis.
Endocrine Abstracts
19
OC31
36. Pettersson
U
Albagha
OM
Mirolo
M
Taranta
A
Frattini
A
2005
Polymorphisms of the CLCN7 gene are associated with BMD in
women.
J Bone Miner Res
20
1960
1967
37. Koller
DL
Ichikawa
S
Lai
D
Padgett
LR
Doheny
KF
2010
Genome-wide association study of bone mineral density in
premenopausal European-American women and replication in African-American
women.
J Clin Endocrinol Metab
95
1802
1809
38. Topaz
O
Shurman
DL
Bergman
R
Indelman
M
Ratajczak
P
2004
Mutations in GALNT3, encoding a protein involved in O-linked
glycosylation, cause familial tumoral calcinosis.
Nat Genet
36
579
581
39. Frishberg
Y
Topaz
O
Bergman
R
Behar
D
Fisher
D
2005
Identification of a recurrent mutation in GALNT3 demonstrates
that hyperostosis-hyperphosphatemia syndrome and familial tumoral calcinosis
are allelic disorders.
J Mol Med
83
33
38
40. Larsson
T
Davis
SI
Garringer
HJ
Mooney
SD
Draman
MS
2005
Fibroblast growth factor-23 mutants causing familial tumoral
calcinosis are differentially processed.
Endocrinology
146
3883
3891
41. Urakawa
I
Yamazaki
Y
Shimada
T
Iijima
K
Hasegawa
H
2006
Klotho converts canonical FGF receptor into a specific receptor
for FGF23.
Nature
444
770
774
42. Kawano
K
Ogata
N
Chiano
M
Molloy
H
Kleyn
P
2002
Klotho gene polymorphisms associated with bone density of aged
postmenopausal women.
J Bone Miner Res
17
1744
1751
43. Ogata
N
Matsumura
Y
Shiraki
M
Kawano
K
Koshizuka
Y
2002
Association of klotho gene polymorphism with bone density and
spondylosis of the lumbar spine in postmenopausal women.
Bone
31
37
42
44. Riancho
JA
Valero
C
Hernandez
JL
Ortiz
F
Zarrabeitia
A
2007
Association of the F352V variant of the Klotho gene with bone
mineral density.
Biogerontology
8
121
127
45. Yamada
Y
Ando
F
Niino
N
Shimokata
H
2005
Association of polymorphisms of the androgen receptor and klotho
genes with bone mineral density in Japanese women.
J Mol Med
83
50
57
46. Sims
AM
Shephard
N
Carter
K
Doan
T
Dowling
A
2007
Genetic Analyses in a Sample of Individuals With High or Low Bone
Density Demonstrates Association With Multiple Wnt Pathway
Genes.
J Bone Miner Res
23
499
506
47. van Meurs
JB
Rivadeneira
F
Jhamai
M
Hugens
W
Hofman
A
2006
Common genetic variation of the low-density lipoprotein
receptor-related protein 5 and 6 genes determines fracture risk in elderly
white men.
J Bone Miner Res
21
141
150
48. Kim
KA
Wagle
M
Tran
K
Zhan
X
Dixon
MA
2008
R-Spondin family members regulate the Wnt pathway by a common
mechanism.
Mol Biol Cell
19
2588
2596
49. Blaydon
DC
Ishii
Y
O'Toole
EA
Unsworth
HC
Teh
MT
2006
The gene encoding R-spondin 4 (RSPO4), a secreted protein
implicated in Wnt signaling, is mutated in inherited
anonychia.
Nat Genet
38
1245
1247
50. Aoki
M
Mieda
M
Ikeda
T
Hamada
Y
Nakamura
H
2007
R-spondin3 is required for mouse placental
development.
Dev Biol
301
218
226
51. Trost
Z
Trebse
R
Prezelj
J
Komadina
R
Logar
DB
2010
A microarray based identification of osteoporosis-related genes
in primary culture of human osteoblasts.
Bone
46
72
80
52. Malaval
L
Wade-Gueye
NM
Boudiffa
M
Fei
J
Zirngibl
R
2008
Bone sialoprotein plays a functional role in bone formation and
osteoclastogenesis.
J Exp Med
205
1145
1153
53. Noor
A
Windpassinger
C
Vitcu
I
Orlic
M
Rafiq
MA
2009
Oligodontia is caused by mutation in LTBP3, the gene encoding
latent TGF-beta binding protein 3.
Am J Hum Genet
84
519
523
54. Dabovic
B
Chen
Y
Colarossi
C
Obata
H
Zambuto
L
2002
Bone abnormalities in latent TGF-[beta] binding protein
(Ltbp)-3-null mice indicate a role for Ltbp-3 in modulating
TGF-[beta] bioavailability.
J Cell Biol
156
227
232
55. Nissen-Meyer
LS
Jemtland
R
Gautvik
VT
Pedersen
ME
Paro
R
2007
Osteopenia, decreased bone formation and impaired osteoblast
development in Sox4 heterozygous mice.
J Cell Sci
120
2785
2795
Štítky
Genetika Reprodukční medicína
Článek Incorporating Biological Pathways via a Markov Random Field Model in Genome-Wide Association StudiesČlánek Survival Motor Neuron Protein Regulates Stem Cell Division, Proliferation, and Differentiation inČlánek Epigenetic Regulation of Cell Type–Specific Expression Patterns in the Human Mammary Epithelium
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 4- Akutní intermitentní porfyrie
- Růst a vývoj dětí narozených pomocí IVF
- Farmakogenetické testování pomáhá předcházet nežádoucím efektům léčiv
- Pilotní studie: stres a úzkost v průběhu IVF cyklu
- Vliv melatoninu a cirkadiálního rytmu na ženskou reprodukci
-
Všechny články tohoto čísla
- Is Co-Expressed with Closely Adjacent Uncharacterised Genes Spanning a Breast Cancer Susceptibility Locus at 6q25.1
- The Liberation of Embryonic Stem Cells
- Genome-Wide Meta-Analysis Identifies Regions on 7p21 () and 15q24 () As Determinants of Habitual Caffeine Consumption
- A Sustained Dietary Change Increases Epigenetic Variation in Isogenic Mice
- The Exocyst Protein Sec10 Interacts with Polycystin-2 and Knockdown Causes PKD-Phenotypes
- Incorporating Biological Pathways via a Markov Random Field Model in Genome-Wide Association Studies
- Survival Motor Neuron Protein Regulates Stem Cell Division, Proliferation, and Differentiation in
- Identification and Functional Validation of the Novel Antimalarial Resistance Locus in
- Does Positive Selection Drive Transcription Factor Binding Site Turnover? A Test with Drosophila Cis-Regulatory Modules
- Protein Phosphatase 2A Controls Ethylene Biosynthesis by Differentially Regulating the Turnover of ACC Synthase Isoforms
- Ribosomal DNA Deletions Modulate Genome-Wide Gene Expression: “–Sensitive” Genes and Natural Variation
- Reciprocal Sign Epistasis between Frequently Experimentally Evolved Adaptive Mutations Causes a Rugged Fitness Landscape
- Variable Pathogenicity Determines Individual Lifespan in
- Evolution of Vertebrate Transient Receptor Potential Vanilloid 3 Channels: Opposite Temperature Sensitivity between Mammals and Western Clawed Frogs
- Towards Establishment of a Rice Stress Response Interactome
- Mouse Genome-Wide Association and Systems Genetics Identify As a Regulator of Bone Mineral Density and Osteoclastogenesis
- Quantitative Fitness Analysis Shows That NMD Proteins and Many Other Protein Complexes Suppress or Enhance Distinct Telomere Cap Defects
- Highly Precise and Developmentally Programmed Genome Assembly in Requires Ligase IV–Dependent End Joining
- PDP-1 Links the TGF-β and IIS Pathways to Regulate Longevity, Development, and Metabolism
- Genome-Wide Association Study Using Extreme Truncate Selection Identifies Novel Genes Affecting Bone Mineral Density and Fracture Risk
- Eight Common Genetic Variants Associated with Serum DHEAS Levels Suggest a Key Role in Ageing Mechanisms
- 14-3-3 Proteins Regulate Exonuclease 1–Dependent Processing of Stalled Replication Forks
- HDA6 Regulates Locus-Directed Heterochromatin Silencing in Cooperation with MET1
- Epigenetic Regulation of Cell Type–Specific Expression Patterns in the Human Mammary Epithelium
- Enhanced Statistical Tests for GWAS in Admixed Populations: Assessment using African Americans from CARe and a Breast Cancer Consortium
- Beyond Missing Heritability: Prediction of Complex Traits
- An Evolutionary Genomic Approach to Identify Genes Involved in Human Birth Timing
- Long-Lost Relative Claims Orphan Gene: in a Wasp
- PTG Depletion Removes Lafora Bodies and Rescues the Fatal Epilepsy of Lafora Disease
- Chromatin Organization in Sperm May Be the Major Functional Consequence of Base Composition Variation in the Human Genome
- GWAS of Follicular Lymphoma Reveals Allelic Heterogeneity at 6p21.32 and Suggests Shared Genetic Susceptibility with Diffuse Large B-cell Lymphoma
- Loss-of-Function Mutations in Cause Metachondromatosis, but Not Ollier Disease or Maffucci Syndrome
- DNA Damage, Somatic Aneuploidy, and Malignant Sarcoma Susceptibility in Muscular Dystrophies
- The Phylogenetic Origin of Coincided with the Origin of Maternally Provisioned Germ Plasm and Pole Cells at the Base of the Holometabola
- Genome Analysis Reveals Interplay between 5′UTR Introns and Nuclear mRNA Export for Secretory and Mitochondrial Genes
- Genome-Wide Association Analysis of Soluble ICAM-1 Concentration Reveals Novel Associations at the , , , and Loci
- The Complete Spectrum of Yeast Chromosome Instability Genes Identifies Candidate CIN Cancer Genes and Functional Roles for ASTRA Complex Components
- Dynamic Regulation of H3K27 Trimethylation during Differentiation
- Phosphorylation-Dependent Differential Regulation of Plant Growth, Cell Death, and Innate Immunity by the Regulatory Receptor-Like Kinase BAK1
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- PTG Depletion Removes Lafora Bodies and Rescues the Fatal Epilepsy of Lafora Disease
- Evolution of Vertebrate Transient Receptor Potential Vanilloid 3 Channels: Opposite Temperature Sensitivity between Mammals and Western Clawed Frogs
- Survival Motor Neuron Protein Regulates Stem Cell Division, Proliferation, and Differentiation in
- An Evolutionary Genomic Approach to Identify Genes Involved in Human Birth Timing
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání




