-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Microbial Programming of Systemic Innate Immunity and Resistance to Infection
article has not abstract
Published in the journal: . PLoS Pathog 10(12): e32767. doi:10.1371/journal.ppat.1004506
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1004506Summary
article has not abstract
Introduction
A multicellular organism is subject to constant microbial exposure throughout its life. These interactions can be transient or permanent, with microbes ranging from those considered commensal to pathogenic [1]. The initial host response to these myriad microbes relies upon the innate immune system. This highly conserved and ancient arm of host defense has generally been thought of as hardwired and inflexible [2], with only local interactions with microbes at environmental interfaces believed to exert a significant influence on innate immune cell function [3], [4]. It is now becoming clear that this long-held dogma is incorrect, and the ongoing microbial exposure we experience throughout life, whether it is by the microbiota or pathogenic organisms, exerts a systemic influence on the production and function of innate immune cells. In this Pearl, I will describe the mechanistic basis for these systemic effects and discuss how they modulate host defenses to infection by other microbes.
Systemic Regulation of Macrophage, Neutrophil, and Dendritic Cell Function by the Microbiota Enhances Innate Defenses against Infection
Tissue resident macrophages and dendritic cells (DCs), combined with recruited neutrophils, are major innate effector cells that form the first line of host defense to protect against infection and help maintain tissue homeostasis. The impact of microbes on the production and functional programming of macrophages and dendritic cells has generally been assumed limited to the mucosa, with colonizing microbes known to fine-tune the function of these cells at this site. Microbial influences on neutrophils have been thought restricted to severe infections, where there can be a short-term increase in neutrophil production, but neutrophil function has been believed to be subject to minimal microbial influence because of their terminal differentiation and short half-life [5]. Recent work has brought about a reevaluation of these views and has shown that neutrophils are actually subject to microbial regulation throughout their life even in the absence of infection (Fig. 1). This starts with their production, as mice devoid of any live microbial communities (germ-free) produce fewer neutrophils, compared to conventional mice colonized by the microbiota [6]. Functionally, circulating neutrophils in germ-free mice have defects in extravasation from the bloodstream into target tissues in response to microbial signals [7] and also in killing of bacterial pathogens [8]. Thus, signals from the microbiota have a systemic effect on neutrophils promoting their production and antimicrobial capacity. The role of neutrophils has now been shown to extend beyond this acute innate response into the regulation of adaptive immunity. Somatic hypermutation and antibody production by B-cells in the spleen is, in part, controlled by splenic neutrophils and, analogously to the innate function of neutrophils, this novel aspect of neutrophil biology is also thought to be promoted by the microbiota [9]. Like neutrophils, macrophage populations in systemic, nonmucosal tissues are also subject to microbial regulation. In the absence of the microbiota, splenic macrophage numbers are reduced, as are the expression of host defense genes, including those encoding proteins involved in antiviral immunity, such as type I interferon [10], [11]. In another example of the long-range influence the microbiota can have on macrophage function, it has been shown that signals from intestinal bacteria promote reactive oxygen species (ROS) production by alveolar macrophages in the lung in response to bacterial pathogens [12]. In the absence of this stimulation, ROS production during infection is reduced, resulting in attenuated early clearance of bacteria from the lung [12]. Collectively, this suggests that with reduced microbial burden, the host is economical with its resources, diverting fewer to innate cell production and minimizing the production of costly molecules that could lead to tissue damage, such as inflammatory cytokines and ROS. In contrast to neutrophils and macrophages, the number of dendritic cells in nonmucosal tissues is thought to be equivalent between germ-free and conventional mice, suggesting that the microbiota does not regulate dendritic cell production systemically [13]. The microbiota does, however, play a significant role in shaping their function, as splenic dendritic cells isolated from germ-free mice and then stimulated with PRR ligands, express significantly less il6, tnfa, il12, il18, and type I interferon, in comparison to the same cells from conventionalized animals [13]. The proposed mechanistic basis for this is via epigenetic modification of the promoters of the genes encoding these proinflammatory cytokines. Splenic DC isolated from conventional mice have greater trimethylation of histone 3 lysine 4 (H3K4), compared to the same cells from germ-free animals [13], which is characteristic of genes undergoing active transcription, and therefore microbiota-dependent chromatin modifications could be the basis for the increased sensitivity of dendritic cells from conventional mice to microbial stimulation. Further functional defects in dendritic cells have been identified in other work, showing that the migration of dendritic cells from the lung to mediastinal lymph nodes during influenza infection is severely impaired in the absence of the microbiota [14]. Emerging from these data is a clear picture showing that the microbiota not only has a proximal influence on innate cells at the mucosa but also has a distal influence, regulating the function of systemic innate cells during homeostasis. This regulation of innate cell function by bacteria within the microbiota leads to enhanced innate defense to infection by viruses and bacteria. Microbiota-dependent stimulation of neutrophils enhances host defenses against systemic bacterial infection while it has been shown that systemic programming of macrophage and dendritic cells function provides more robust antibacterial and antiviral immunity in the lung [8], [10]–[14].
Fig. 1. Systemic regulation of neutrophil production and function by the microbiota. 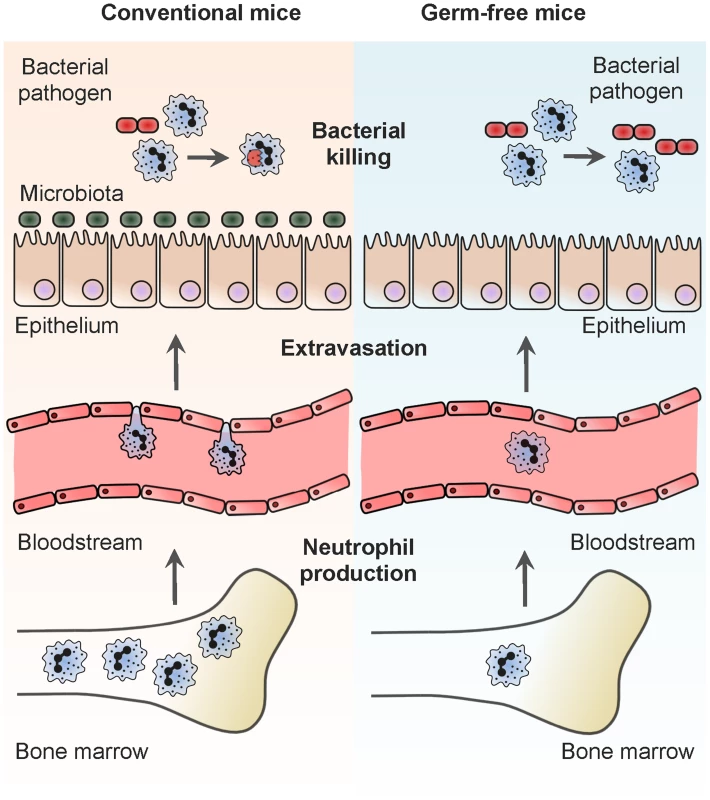
This summarizes the stages in neutrophil production and function that are known to be regulated by the microbiota. Conventional mice are those colonized by the microbiota and germ-free mice are those that have been born and raised in a sterile environment and thus not colonized by any live microbial communities. Neutrophil production: the site of neutrophil production is the bone marrow, and signals from the microbiota, which can be mediated via TLR4, stimulate neutrophil production continually under steady-state conditions. Neutrophil extravasation: to protect against infection, neutrophils are recruited from the bloodstream to inflamed tissues. In the absence of TLR stimulation from the microbiota, the ability of neutrophils to navigate this migratory step, in response to microbial signals, is diminished. Neutrophil killing of bacteria: neutrophils leave the bone marrow fully differentiated and mature. While in the bone marrow, bacterial cell wall peptidoglycan is recognized by Nod1, primes the antibacterial capacity of neutrophils, and facilitates more effective killing of bacterial pathogens at sites of recruitment. Pattern Recognition Establishes the Level of Innate Immune Activation
A common theme apparent from this work is that the mechanistic basis for many of these systemic phenomena involves the activation of pattern recognition receptors (PRRs), host receptors that recognize conserved microbial structures found in all microbes, whether they are considered pathogens or nonpathogens. Steady-state neutrophil production is regulated by Toll-like receptor 4 (Tlr4) activation [6], extravasation is enhanced in response to redundant Toll-like receptor (TLR) activation via MyD88 [7], and bacterial killing is promoted by recognition of bacterial cell wall peptidoglycan by the Nod-like receptor (NLR) 1, Nod1 (Fig. 1) [8]. Similarly, PRR activation can drive the systemic activation of macrophages and dendritic cells, for example, enhanced ROS production by alveolar macrophages is promoted by NLR ligands originating from the intestine [12] and impaired dendritic cell migration during influenza infection in the absence of the microbiota can be rescued by intrarectal administration of lipopolysaccharide (LPS) [14]. But how do microbes within the microbiota and their products at the mucosa exert a systemic effect on innate cells in distal tissues via PRRs? Recent work shows that the boundary between the external microbial environment delimited by the mucosa, and sterile, internal, nonmucosal tissues is more blurred than previously recognized. Multiple studies [8], [9], [15], [16] and our unpublished data (Clarke, T.B. unpublished) have found microbial products throughout the body, and these disseminated products are known to exert some of the systemic effects of the microbiota. For example, peptidoglycan recognized by Nod1 and required for systemic priming of neutrophil function is found in multiple host tissues and accumulates in the bone marrow, where priming takes place [8]. Furthermore, neutrophils in the spleen have been shown to contain bacterial DNA and LPS [9], providing further evidence for the continual dissemination of bacterial products systemically. Another, nonmutually exclusive mechanism can be through local PRR activation by the microbiota at environmental interfaces leading to the production of host signaling molecules that can exert systemic effects on systemic innate cells. TLR signaling resulting in mucosal IL-17A production has been shown to be important for promoting neutrophil granulocytosis in neonatal mice [17] and provides an example of this second mechanism. Taken together, these studies are beginning to show that PRRs act as homeostatic regulators of the systemic immune system and gauge the required level of innate cell activity through continual tonic engagement by microbial products.
Programming of Innate Immunity Is Not Limited to the Microbiota
In the examples outlined above, programming of innate cell function was achieved by microbes and microbial products from the microbiota. However, these are not the only microbes we interact with, and this, therefore, raises the question of whether microbial modulation of innate immunity is somehow unique to the microbiota or can also be mediated by other microbial encounters. For humans, these interactions can be infection with pathogenic microbes or vaccination. Vaccination with Bacillus Calmette-Guérin (BCG), an attenuated strain of Mycobacterium bovis, protects against tuberculosis (TB), but its effects on the immune system extend far beyond specific protection against TB [18]. BCG vaccination has been shown to afford nonspecific protection against infection by a number of pathogens, including Schistosoma mansoni and Listeria monocytogenes [18]. This is, in part, mediated by programming of circulating monocytes, macrophage, and dendritic cell precursors that are also potent innate cells prior to their differentiation [19]. BCG vaccination increases the expression of activation markers CD14 and CD11b on circulating monocytes, and upon stimulation of these cells by PRR ligands or bacteria, they produce significantly more IL-1β and TNFα than the same cells from unvaccinated individuals [19]. Using mouse models, these effects were shown to be independent of the adaptive immune system and required PRR activation, specifically, the NLR Nod2 [19]. Again, in line with the effects of the microbiota on innate cells, the promoters of genes showing increased production upon stimulation had greater levels of trimethylation of H3K4 [19].
The ability of one infection to influence early host responses to infection by another, unrelated pathogen has also been known for a long time. The mechanistic basis for this has been previously ascribed to heterologous immunity [20], a phenomena whereby the memory lymphocytes that developed in response to one pathogen recognize, and are activated by, a crossreactive antigenic epitope present on another unrelated pathogen [21]. In addition to these effects on adaptive immunity, infection-induced changes in innate cell function have now been shown to shape host defense systemically. Intranasal infection with herpesvirus promotes macrophage production of type II interferon, and this helps protect against systemic bacterial infection, independently of any component of adaptive immunity [22]. However, chronic immune activation because of infection or excessive microbial stimulation can also result in defects in immune function [23]. These detrimental effects have been most comprehensively studied from the perspective of the adaptive immune system, with comparatively less known of the systemic effects of chronic infection on innate cells and how this influences subsequent infection [23]. However, it is well established that the extreme microbial stimulation that occurs during sepsis causes lasting alterations in innate cell function, including reduced ROS production by neutrophils in the bone marrow that can lead to poorer clearance of bacterial lung infection [24]. Additionally, it is known that patients with concurrent intestinal helminth and TB infection have reduced natural killer cell numbers in comparison to those with TB alone [25]. Thus, more information is required to understand the beneficial and detrimental effects different microbial groups can have on innate immunity.
Conclusion
It is clear from this work that innate immunity is a far more flexible and responsive system than previously appreciated. The innate immune system is constantly responding to its surroundings and adjusting the production and function of innate effector cells to match the microbial challenge of its current environment. This is not limited to the mucosa but is also true of the systemic immune compartment, showing the profound influence microbes have on shaping the entire immune system. This fundamental microbial programming of innate immunity, often through PRR signaling, allows economical use of resources, avoiding unnecessary production of potentially tissue damaging molecules like ROS, while gauging the required level of immune activation to effectively deal with infection. Microbial programming of innate immunity resulting in enhanced protection against infection can be mediated by myriad microbes, ranging from members of the microbiota to pathogens, suggesting that we need to move beyond this simplistic dichotomy to fully comprehend the microbial groups that regulate different aspects of immune function. It might then be possible to harness the power of specific microbial groups to exploit the flexibility to the innate immune system for therapeutic benefit to improve responses to vaccination and help protect against infections.
Zdroje
1. HondaK, LittmanDR (2012) The microbiome in infectious disease and inflammation. Annu Rev Immunol 30 : 759–795.
2. JanewayCAJr, MedzhitovR (2002) Innate immune recognition. Annu Rev Immunol 20 : 197–216.
3. ArtisD (2008) Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat Rev Immunol 8 : 411–420.
4. JarchumI, PamerEG (2011) Regulation of innate and adaptive immunity by the commensal microbiota. Curr Opin Immunol 23 : 353–360.
5. ManzMG, BoettcherS (2014) Emergency granulopoiesis. Nat Rev Immunol 14 : 302–314.
6. BuglS, WirthsS, RadsakMP, SchildH, SteinP, et al. (2013) Steady-state neutrophil homeostasis is dependent on TLR4/TRIF signaling. Blood 121 : 723–733.
7. KarmarkarD, RockKL (2013) Microbiota signalling through MyD88 is necessary for a systemic neutrophilic inflammatory response. Immunology 140 : 483–492.
8. ClarkeTB, DavisKM, LysenkoES, ZhouAY, YuY, et al. (2010) Recognition of peptidoglycan from the microbiota by Nod1 enhances systemic innate immunity. Nat Med 16 : 228–231.
9. PugaI, ColsM, BarraCM, HeB, CassisL, et al. (2012) B cell-helper neutrophils stimulate the diversification and production of immunoglobulin in the marginal zone of the spleen. Nat Immunol 13 : 170–180.
10. AbtMC, OsborneLC, MonticelliLA, DoeringTA, AlenghatT, et al. (2012) Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity 37 : 158–170.
11. KhosraviA, YanezA, PriceJG, ChowA, MeradM, et al. (2014) Gut microbiota promote hematopoiesis to control bacterial infection. Cell Host Microbe 15 : 374–381.
12. ClarkeTB (2014) Early innate immunity to bacterial infection in the lung is regulated systemically by the commensal microbiota via NLR ligands. Infect Immun 82 : 4596–4606.
13. GanalSC, SanosSL, KallfassC, OberleK, JohnerC, et al. (2012) Priming of natural killer cells by nonmucosal mononuclear phagocytes requires instructive signals from commensal microbiota. Immunity 37 : 171–186.
14. IchinoheT, PangIK, KumamotoY, PeaperDR, HoJH, et al. (2011) Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc Natl Acad Sci U S A 108 : 5354–5359.
15. NakatsujiT, ChiangHI, JiangSB, NagarajanH, ZenglerK, et al. (2013) The microbiome extends to subepidermal compartments of normal skin. Nat Commun 4 : 1431.
16. AagaardK, MaJ, AntonyKM, GanuR, PetrosinoJ, et al. (2014) The placenta harbors a unique microbiome. Sci Transl Med 6 : 237ra265.
17. DeshmukhHS, LiuY, MenkitiOR, MeiJ, DaiN, et al. (2014) The microbiota regulates neutrophil homeostasis and host resistance to Escherichia coli K1 sepsis in neonatal mice. Nat Med 20 : 524–530.
18. BennCS, NeteaMG, SelinLK, AabyP (2013) A small jab - a big effect: nonspecific immunomodulation by vaccines. Trends Immunol 34 : 431–439.
19. KleinnijenhuisJ, QuintinJ, PreijersF, JoostenLA, IfrimDC, et al. (2012) Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci U S A 109 : 17537–17542.
20. ClarkIA (2001) Heterologous immunity revisited. Parasitology 122 Suppl: S51–59
21. WelshRM, SelinLK (2002) No one is naive: the significance of heterologous T-cell immunity. Nat Rev Immunol 2 : 417–426.
22. BartonES, WhiteDW, CathelynJS, Brett-McClellanKA, EngleM, et al. (2007) Herpesvirus latency confers symbiotic protection from bacterial infection. Nature 447 : 326–329.
23. StelekatiE, WherryEJ (2012) Chronic bystander infections and immunity to unrelated antigens. Cell Host Microbe 12 : 458–469.
24. DelanoMJ, ThayerT, GabrilovichS, Kelly-ScumpiaKM, WinfieldRD, et al. (2011) Sepsis induces early alterations in innate immunity that impact mortality to secondary infection. J Immunol 186 : 195–202.
25. Resende CoT, HirschCS, ToossiZ, DietzeR, Ribeiro-RodriguesR (2007) Intestinal helminth co-infection has a negative impact on both anti-Mycobacterium tuberculosis immunity and clinical response to tuberculosis therapy. Clin Exp Immunol 147 : 45–52.
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek Selective Susceptibility of Human Skin Antigen Presenting Cells to Productive Dengue Virus InfectionČlánek P47 Mice Are Compromised in Expansion and Activation of CD8 T Cells and Susceptible to InfectionČlánek Molecular Evolution of Broadly Neutralizing Llama Antibodies to the CD4-Binding Site of HIV-1
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2014 Číslo 12- Stillova choroba: vzácné a závažné systémové onemocnění
- Diagnostika virových hepatitid v kostce – zorientujte se (nejen) v sérologii
- Perorální antivirotika jako vysoce efektivní nástroj prevence hospitalizací kvůli COVID-19 − otázky a odpovědi pro praxi
- Choroby jater v ordinaci praktického lékaře – význam jaterních testů
- Diagnostický algoritmus při podezření na syndrom periodické horečky
-
Všechny články tohoto čísla
- Microbial Programming of Systemic Innate Immunity and Resistance to Infection
- Unique Features of HIV-1 Spread through T Cell Virological Synapses
- Measles Immune Suppression: Functional Impairment or Numbers Game?
- Cellular Mechanisms of Alpha Herpesvirus Egress: Live Cell Fluorescence Microscopy of Pseudorabies Virus Exocytosis
- Rubella Virus: First Calcium-Requiring Viral Fusion Protein
- Plasma Membrane-Located Purine Nucleotide Transport Proteins Are Key Components for Host Exploitation by Microsporidian Intracellular Parasites
- Selective Susceptibility of Human Skin Antigen Presenting Cells to Productive Dengue Virus Infection
- Loss of Dynamin-Related Protein 2B Reveals Separation of Innate Immune Signaling Pathways
- Intraspecies Competition for Niches in the Distal Gut Dictate Transmission during Persistent Infection
- Unveiling the Intracellular Survival Gene Kit of Trypanosomatid Parasites
- Extreme Divergence of Tropism for the Stem-Cell-Niche in the Testis
- HTLV-1 Tax-Mediated Inhibition of FOXO3a Activity Is Critical for the Persistence of Terminally Differentiated CD4 T Cells
- P47 Mice Are Compromised in Expansion and Activation of CD8 T Cells and Susceptible to Infection
- Hypercytotoxicity and Rapid Loss of NKp44 Innate Lymphoid Cells during Acute SIV Infection
- Molecular Evolution of Broadly Neutralizing Llama Antibodies to the CD4-Binding Site of HIV-1
- Crystal Structure of Calcium Binding Protein-5 from and Its Involvement in Initiation of Phagocytosis of Human Erythrocytes
- Chronic Parasitic Infection Maintains High Frequencies of Short-Lived Ly6CCD4 Effector T Cells That Are Required for Protection against Re-infection
- Specific Dysregulation of IFNγ Production by Natural Killer Cells Confers Susceptibility to Viral Infection
- HSV-2-Driven Increase in the Expression of αβ Correlates with Increased Susceptibility to Vaginal SHIV Infection
- Murine Anti-vaccinia Virus D8 Antibodies Target Different Epitopes and Differ in Their Ability to Block D8 Binding to CS-E
- Brothers in Arms: Th17 and Treg Responses in Immunity
- Granulocytes Impose a Tight Bottleneck upon the Gut Luminal Pathogen Population during Typhimurium Colitis
- A Negative Feedback Modulator of Antigen Processing Evolved from a Frameshift in the Cowpox Virus Genome
- Discovery of Replicating Circular RNAs by RNA-Seq and Computational Algorithms
- The Non-receptor Tyrosine Kinase Tec Controls Assembly and Activity of the Noncanonical Caspase-8 Inflammasome
- Targeted Changes of the Cell Wall Proteome Influence Ability to Form Single- and Multi-strain Biofilms
- Apoplastic Venom Allergen-like Proteins of Cyst Nematodes Modulate the Activation of Basal Plant Innate Immunity by Cell Surface Receptors
- The Toll-Dorsal Pathway Is Required for Resistance to Viral Oral Infection in
- Anti-α4 Antibody Treatment Blocks Virus Traffic to the Brain and Gut Early, and Stabilizes CNS Injury Late in Infection
- Initiation of ART during Early Acute HIV Infection Preserves Mucosal Th17 Function and Reverses HIV-Related Immune Activation
- Microbial Urease in Health and Disease
- Emergence of MERS-CoV in the Middle East: Origins, Transmission, Treatment, and Perspectives
- Blocking Junctional Adhesion Molecule C Enhances Dendritic Cell Migration and Boosts the Immune Responses against
- IL-28B is a Key Regulator of B- and T-Cell Vaccine Responses against Influenza
- A Natural Genetic Variant of Granzyme B Confers Lethality to a Common Viral Infection
- Neutral Sphingomyelinase in Physiological and Measles Virus Induced T Cell Suppression
- Differential PfEMP1 Expression Is Associated with Cerebral Malaria Pathology
- The Role of the NADPH Oxidase NOX2 in Prion Pathogenesis
- Rapid Evolution of Virus Sequences in Intrinsically Disordered Protein Regions
- The Central Role of cAMP in Regulating Merozoite Invasion of Human Erythrocytes
- Expression of Suppressor of Cytokine Signaling 1 (SOCS1) Impairs Viral Clearance and Exacerbates Lung Injury during Influenza Infection
- Cellular Oxidative Stress Response Controls the Antiviral and Apoptotic Programs in Dengue Virus-Infected Dendritic Cells
- SUMOylation by the E3 Ligase TbSIZ1/PIAS1 Positively Regulates VSG Expression in
- Monocyte Recruitment to the Dermis and Differentiation to Dendritic Cells Increases the Targets for Dengue Virus Replication
- Oral Streptococci Utilize a Siglec-Like Domain of Serine-Rich Repeat Adhesins to Preferentially Target Platelet Sialoglycans in Human Blood
- SV40 Utilizes ATM Kinase Activity to Prevent Non-homologous End Joining of Broken Viral DNA Replication Products
- Amphipathic α-Helices in Apolipoproteins Are Crucial to the Formation of Infectious Hepatitis C Virus Particles
- Proteomic Analysis of the Acidocalcisome, an Organelle Conserved from Bacteria to Human Cells
- Experimental Cerebral Malaria Pathogenesis—Hemodynamics at the Blood Brain Barrier
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Plasma Membrane-Located Purine Nucleotide Transport Proteins Are Key Components for Host Exploitation by Microsporidian Intracellular Parasites
- Rubella Virus: First Calcium-Requiring Viral Fusion Protein
- Emergence of MERS-CoV in the Middle East: Origins, Transmission, Treatment, and Perspectives
- Unique Features of HIV-1 Spread through T Cell Virological Synapses
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



