-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Exome Sequencing Identifies Rare Deleterious Mutations in DNA Repair Genes and as Potential Breast Cancer Susceptibility Alleles
Despite intensive efforts using linkage and candidate gene approaches, the genetic etiology for the majority of families with a multi-generational breast cancer predisposition is unknown. In this study, we used whole-exome sequencing of thirty-three individuals from 15 breast cancer families to identify potential predisposing genes. Our analysis identified families with heterozygous, deleterious mutations in the DNA repair genes FANCC and BLM, which are responsible for the autosomal recessive disorders Fanconi Anemia and Bloom syndrome. In total, screening of all exons in these genes in 438 breast cancer families identified three with truncating mutations in FANCC and two with truncating mutations in BLM. Additional screening of FANCC mutation hotspot exons identified one pathogenic mutation among an additional 957 breast cancer families. Importantly, none of the deleterious mutations were identified among 464 healthy controls and are not reported in the 1,000 Genomes data. Given the rarity of Fanconi Anemia and Bloom syndrome disorders among Caucasian populations, the finding of multiple deleterious mutations in these critical DNA repair genes among high-risk breast cancer families is intriguing and suggestive of a predisposing role. Our data demonstrate the utility of intra-family exome-sequencing approaches to uncover cancer predisposition genes, but highlight the major challenge of definitively validating candidates where the incidence of sporadic disease is high, germline mutations are not fully penetrant, and individual predisposition genes may only account for a tiny proportion of breast cancer families.
Published in the journal: . PLoS Genet 8(9): e32767. doi:10.1371/journal.pgen.1002894
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002894Summary
Despite intensive efforts using linkage and candidate gene approaches, the genetic etiology for the majority of families with a multi-generational breast cancer predisposition is unknown. In this study, we used whole-exome sequencing of thirty-three individuals from 15 breast cancer families to identify potential predisposing genes. Our analysis identified families with heterozygous, deleterious mutations in the DNA repair genes FANCC and BLM, which are responsible for the autosomal recessive disorders Fanconi Anemia and Bloom syndrome. In total, screening of all exons in these genes in 438 breast cancer families identified three with truncating mutations in FANCC and two with truncating mutations in BLM. Additional screening of FANCC mutation hotspot exons identified one pathogenic mutation among an additional 957 breast cancer families. Importantly, none of the deleterious mutations were identified among 464 healthy controls and are not reported in the 1,000 Genomes data. Given the rarity of Fanconi Anemia and Bloom syndrome disorders among Caucasian populations, the finding of multiple deleterious mutations in these critical DNA repair genes among high-risk breast cancer families is intriguing and suggestive of a predisposing role. Our data demonstrate the utility of intra-family exome-sequencing approaches to uncover cancer predisposition genes, but highlight the major challenge of definitively validating candidates where the incidence of sporadic disease is high, germline mutations are not fully penetrant, and individual predisposition genes may only account for a tiny proportion of breast cancer families.
Introduction
Around one in six women who develop breast cancer has a first degree relative with the condition [1]. In the mid 1990s, a classical linkage approach identified germline mutations in two genes, BRCA1 and BRCA2, which are associated with a high risk of developing both breast and ovarian cancer [2], [3]. Although BRCA1 and BRCA2-specific genetic testing is rapidly evolving in the clinical setting, mutations in these genes are successful at explaining only around half of the dominant multi-case breast cancer only families [4], and their contribution to the heritable risk of breast cancer has been estimated to be no more than around 20% of the total [5], [6]. Importantly, the identification and management of individuals with high-risk breast cancer predisposition gene mutations is now well accepted in clinical practice. Although evidence-based risk management is only possible in a relatively small group of families, as it is limited by the identification of an underlying genetic mutation, the benefits for those individuals are well established [7].
Through a candidate gene approach, mutations in other high and moderate penetrance cancer-susceptibility genes have been identified in a further small proportion of families but the underlying etiology of the increased susceptibility to breast cancer in the majority of multi-case breast cancer families remains unknown. Recent advances in massively parallel sequencing technology have provided an agnostic means by which to efficiently identify germline mutations in individuals with inherited cancer syndromes at the individual family or cancer-specific level [8], [9]. The aim of this study is to identify through a whole exome sequencing approach, the underlying familial predisposition to breast cancer in multiple multi-generational breast cancer families in whom no BRCA1 or BRCA2 mutation was identified (BRCA1/2 negative families), and to assess the candidate genes identified by this means in a cohort of familial BRCA1/2 negative breast and ovarian cancer patients.
Results/Discussion
We performed intra-family exome sequence analysis of multiple affected relatives from 15 high-risk, trans-generational breast cancer families in whom full BRCA1 and BRCA2 mutation analysis had been performed and was uninformative in at least one breast cancer-affected family member (Table 1). Sequencing was performed on GAIIx or HiSeq instruments (Illumina). The average read depth achieved for target regions was 83.19 and at least 80% (average 89.12%) of the capture target regions were covered by 10 or more sequence reads for all samples (Table S1). Following data filtering, an average of 35 overtly deleterious and 284 non-synonymous mutations were identified per individual (Table S1).
Tab. 1. Characteristics of 15 high-risk breast cancer families selected for exome sequencing. 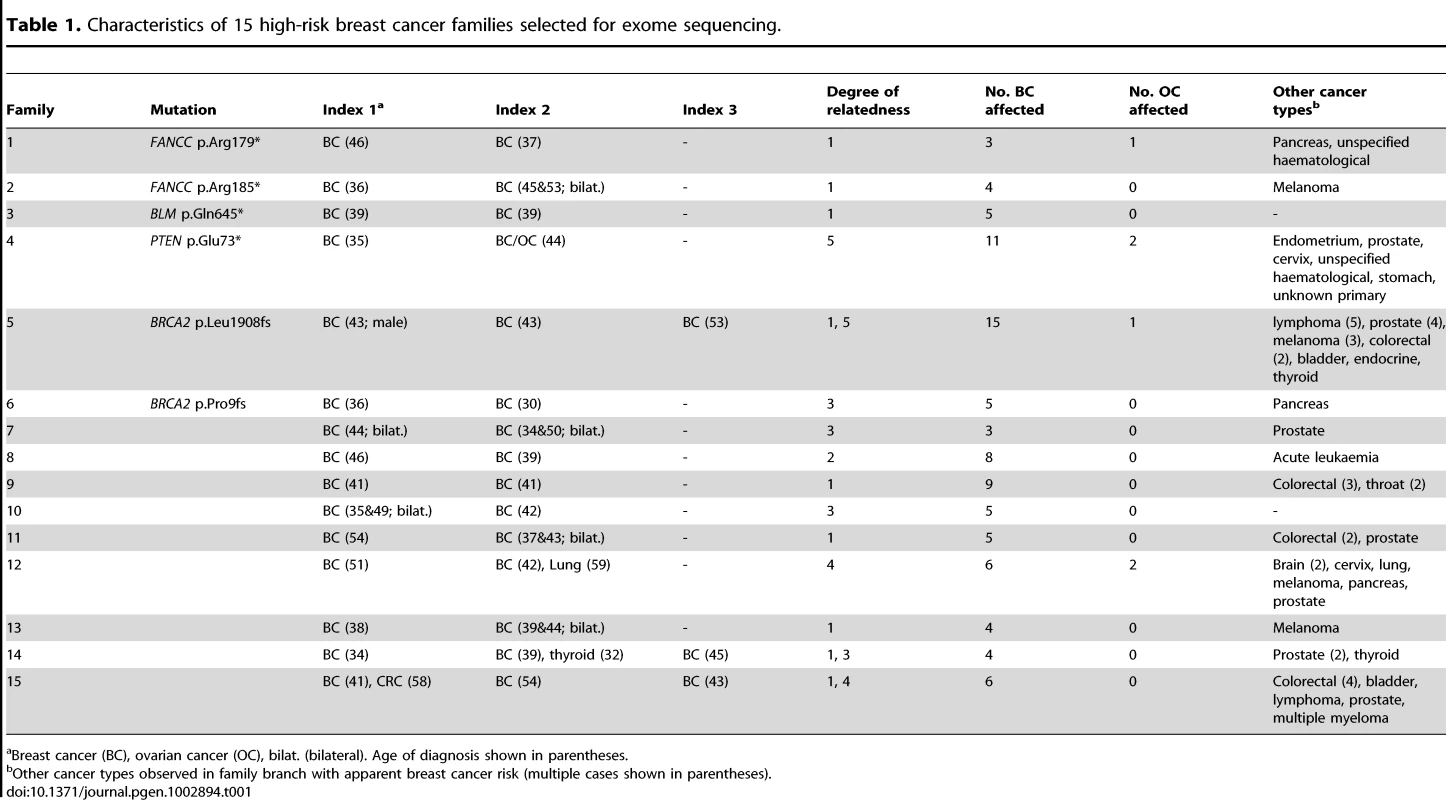
Breast cancer (BC), ovarian cancer (OC), bilat. (bilateral). Age of diagnosis shown in parentheses. To identify candidate predisposition genes we only considered those with overtly deleterious mutations that were shared by multiple affected relatives and/or were targeted in more than one family and further priority was given to genes with a role in mechanistically well-established breast cancer–associated DNA repair. A list of all overtly deleterious mutations identified in among the 33 individuals sequenced is provided in Table S2. Two of the fifteen families were found to carry independent heterozygous truncating mutations in the Fanconi Anemia (FA) gene, FANCC. Neither family was reported to be of Ashkenazi Jewish ancestry and the mutations are different to those commonly reported among this ethnic group. Family 1 carried a novel nonsense mutation (FANCC c.535C>T, p.Arg179*) that was present in the youngest affected individual (breast cancer at age 37) and in her mother who had ovarian cancer at age 66, but not in her breast cancer-affected sister who was diagnosed at age 46 (Figure 1). Family 2 was found to harbor a known pathogenic FA mutation (FANCC c.553C>T, p.Arg185*) [10] which was present in two sisters who developed breast cancer aged 36, and bilateral breast cancer aged 46 and 53, respectively. A third family analyzed by exome sequencing was found to carry a heterozygous c.1993C>T mutation in the BLM gene which is predicted to truncate the protein at codon 645 (p.Gln645*). This known pathogenic Bloom syndrome mutation [11] co-segregated with cancer in the family (Figure 1), being present in all three sisters diagnosed with breast cancer aged 39, 39 and 41 years respectively and absent in the two unaffected sisters. Although retrospective likelihood segregation analysis of these limited pedigrees did not reach significance (see Text S1), overall, co-segregation of FANCC and BLM mutations in these families appears consistent with that expected for moderately penetrant breast cancer alleles.
Fig. 1. FANCC and BLM mutations identified in familial breast cancer pedigrees. 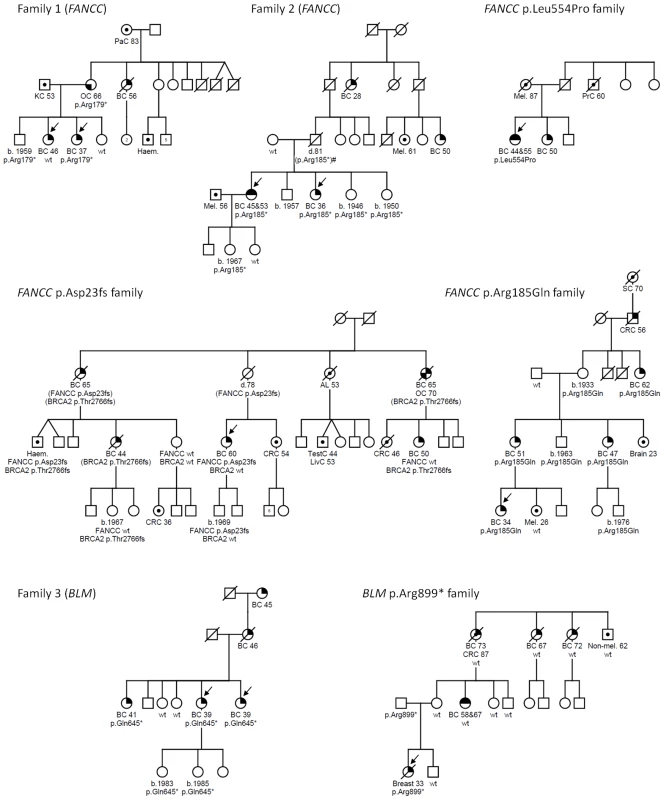
Males and females are represented by squares and circles, respectively. Arrows indicate individuals who underwent whole exome sequencing (families 1–3) or were the index case in subsequent mutation analysis (FANCC p.Asp23fs, p.Leu554Pro and p.Arg185Gln and BLM p.Arg899* families). Cancer-affected individuals are represented with the following symbols: breast cancer, top right quadrant filled in; bilateral breast cancer, top half; ovarian cancer, bottom left quadrant; or other cancers as indicated, centre circle. Mutation status is indicated with either the family specific mutation or wildtype (wt) under each tested individual. Age at cancer diagnosis or year of birth (b.) where known is shown for all mutation carriers. Breast cancer (BC), ovarian cancer (OC), acute leukaemia (AL), colorectal cancer (CRC), haematological malignancy (type unspecified) (Haem.), kidney cancer (KC), liver cancer (LivC), melanoma (Mel.), pancreatic cancer (PaC), prostate cancer (PrC), skin non-melanoma (Non-mel.) stomach cancer (SC), testicular cancer (TestC). Mutations indicated in parentheses indicate untested obligate carriers. Family 2 contains an individual (indicated by #) for whom mutation status is inferred assuming that non-paternity or gonadal mosaicism have not occurred. Mutation analysis of all coding exons of FANCC and BLM was extended to the index cases from a further 438 BRCA1/2 negative breast cancer families (from kConFab). This approach identified one further family with a heterozygous, known pathogenic FANCC mutation, (c.67delG, p.Asp23Ilefs*23, rs104886459) [12] and one with a heterozygous pathogenic BLM mutation (c.2695C>T, p.Arg899*) [11]. For FANCC, mutation hotspot exons 2, 5, 7, 14 and 15 were screened in the index cases from an additional 957 BRCA1/2 uninformative breast cancer families attending familial cancer services (including 561 obtained from the Peter MacCallum Cancer Centre Familial Cancer Centre and a further 396 from kConFab). One further family with a heterozygous FANCC c.1661T>C (p.Leu554Pro, rs104886458) missense variant, which is a functionally validated pathogenic FA mutation, was identified [13].
The index case in the FANCC c.67delG family developed breast cancer at age 60 but independent clinical testing subsequently identified a deleterious mutation in BRCA2 (c.8297delC, p.Thr2766Asnfs*11) in other breast cancer-affected family members (Figure 1). Genotyping of both mutations within this family suggests that different individuals may carry risk conferred by one or both of these family mutations.
The index case of the FANCC c.1661T>C family developed bilateral breast cancer at age 44 and 55, but DNA from other family members was not available for segregation analysis. All FANCC variants detected in index cases or controls are summarized in Table S3.
The index case of the BLM c.2695C>T family developed breast cancer at age 33 but segregation analysis showed the mutation was inherited from her father rather than her mother whose reported family history of breast cancer had initiated their recruitment into kConFab (Figure 1). Interestingly, breast cancer was diagnosed much earlier in the index case compared to her maternal relatives (33 years versus 58 to 73 years) possibly indicating a different genetic etiology. Unfortunately data regarding family history on the paternal side are limited. Neither the father nor the paternal grandparents were reported to have developed cancer but no further information regarding number or cancer status of other relatives is available. All BLM variants detected in index cases or controls are summarized in Table S4.
No pathogenic BLM mutations were detected in 464 healthy controls and none have been reported in the 1000 Genomes data (20100804 release, n = 1,092) [14] compared to 2/438 breast cancer families with BLM mutations. Likewise, no known pathogenic or overtly deleterious FANCC mutations were identified among the 464 controls or the 1000 Genomes data or among 654 healthy controls examined in an independent study [15]. The Exome Variant Server (EVS), NHLBI Exome Sequencing Project, Seattle, WA, does report deleterious mutations in FANCC and BLM in 3/3,510 and 4/3,510 individuals of European decent, respectively. However, this cohort includes extreme tail sampling of traits relating to heart, lung and blood disorders. The latter group in particular may be expected to show enrichment for mutations in DNA repair machinery including FA genes. Excluding the Exome Variant Server frequency data, a total of 4/1,395 breast cancer families screened for all or at least the mutation hot spot exons carried overtly deleterious FANCC mutations compared to none among the combined control population (n = 2,210). While this is indicative that overtly deleterious mutation in FANCC and BLM are likely to be very rare in the population this must be considered a crude measure as the controls were drawn from diverse populations the majority of which were not matched to the index cases. However, it is possible that more families in our breast cancer family cohort may be explained by FANCC and BLM mutations since, for both genes, private non-synonymous variants were identified that are predicted to be damaging by in silico algorithms. One such variant, for which there was DNA available for segregation analysis, was FANCC p.Arg185Gln. This variant closely segregated with disease in this family, which included four female blood relatives with breast cancers diagnosed at ages 34, 51, 47 and 62 (Figure 1). The p.Arg185Gln variant was identified in 1/1,395 breast cancer families but not in any of 464 controls and has not been reported in the 1000 Genomes project or EVS database.
Homozygous mutations in FANCC and BLM are responsible for FA (complementation group C) and Bloom syndrome, respectively, and individuals diagnosed with these syndromes have a high risk of cancer. Functionally, the FA and Bloom syndrome pathways play important roles in homologous recombination (HR) based repair of double-stranded DNA breaks [16], [17]. Constitutional inactivating mutations in genes integral to error-free HR and responsible for FA have been clearly associated with an increased susceptibility to both breast and ovarian cancer [16], and include the genes BRCA1, BRCA2 (FANCD1), FANCN (PALB2), FANCJ (BRIP1), RAD51C (FANCO) and RAD51D. Thus, in addition to the direct genetic evidence that we have described here, FANCC and BLM are strong candidates for breast cancer susceptibility genes due to their role in the precise regulation of HR and some of its associated functions. Although there is limited data, heterozygous FANCC mutations have previously been linked to an increased incidence of breast and early onset pancreatic cancer [15], [18], [19], however, no excess breast and ovarian cancer was observed among Ashkenazi Jews carrying the FANCC c.711+4A>T mutation [20]. While another previous study failed to identify overtly pathogenic FANCC mutations in breast cancer, the study cohort size was small (n = 88) [21]. In keeping with our data, two recurrent truncating mutations in the BLM gene were shown in a case control study to be associated with increased breast cancer risk in Russia [22]. Gruber et al reported an elevated risk of colorectal cancer in Ashkenazi Jews carrying the common BLMASH mutation and a non-significant excess of breast cancer [23] although a later study failed to confirm these findings [24].
Further to the germline mutations in FANCC and BLM, exome sequencing identified mutations in the breast cancer predisposition genes, PTEN and BRCA2 in an additional three of the original 15 families (Figure S1). The truncating PTEN mutation (c.217G>T, p.Glu73*) was identified in only one branch of the family suggesting another susceptibility gene may explain the extended family history. Prior to this finding, the treating familial cancer centre reported no PTEN-associated clinical features within the family. In family 5, exome sequencing identified a deleterious BRCA2 mutation (c.5722_5723delCT, p.Leu1908Argfs*2, rs80359530) in two of the three family members tested (Figure S1). The mutation is present in a male diagnosed with breast cancer but not in the youngest affected female relative in the family, who had been offered the original clinical BRCA1 and BRCA2 mutation test in the clinic setting. Similarly in family 6, exome sequencing identified a deleterious BRCA2 mutation (c.26delC, p.Pro9Glnfs*16, rs80359343) in a female diagnosed with breast cancer at age 30, but not in her cousin who was diagnosed at age 36 and was the only family member to have undergone full diagnostic BRCA1 and BRCA2 gene sequencing (Figure S1). These families are interesting in a clinical context since they were designated as unresolved on the basis of best clinical practice and demonstrate the need for targeted sequencing of all proven breast and ovarian cancer susceptibility genes to obtain maximum information in the clinical setting (as previously demonstrated [25]). Our data also highlights the major challenge confounding genetic studies of common adult onset familial disease; the presence of ‘phenocopies’ in families with an inherited genetic predisposition and/or the convergence of pedigrees with different genetic causes (e.g. PTEN family 4). Among the remaining nine breast cancer families there were numerous genes that were recurrently targeted that warrant further investigation. It is noteworthy that in one family, one individual harbored a known FA pathogenic truncating mutation in FANCL. Mutation of this gene is responsible for a very small fraction of FA families and only three pathogenic mutations in FANCL are recorded in the Fanconi Anemia Mutation Database.
In conclusion, we describe two potential breast cancer susceptibility genes FANCC and BLM both of which have functional roles in the regulation of HR. The heterozygous mutation carrier rate in Caucasians for these genes is extremely low (for FANCC it is estimated at 1/3,000 [15], whilst the carrier frequency of BLM mutations is unknown since the syndrome is exceedingly rare) and notwithstanding the possibility of the “winners curse” [26], the exome sequencing data is strongly suggestive that FANCC and BLM represent breast cancer predisposing genes. Together with the recently identified association of RAD51 paralogues with cancer predisposition [27], [28], our findings suggest that the number of unidentified moderate to high-risk susceptibility genes is very much larger than previously expected and the number of families explained by each gene is likely to be much less than 1% (cf. RAD51C [27], [29]). Consequently, providing definitive evidence for a causative role for novel breast cancer genes will be challenging and will require validation of rare mutations in thousands rather than hundreds of families. We predict that this will be a generic problem associated with identifying causative mutations in common diseases such as breast cancer and that validation rather than the technical exercise of exome sequencing is where the real challenge lies.
Materials and Methods
Subjects
This study was approved by the Peter Mac Ethics Committee (project numbers 09/62 and 11/50). Informed consent was obtained from all participants. Fifteen high-risk breast cancer families with at least four cases of multi-generational breast cancer including at least one additional high-risk feature (such as bilateral, early onset or male breast cancer, or ovarian cancer) and at least two available blood specimens from breast cancer-affected individuals, were selected for whole exome sequencing from among approximately 800 BRCA1 and BRCA2 mutation negative families from the Kathleen Cunningham Foundation Consortium for Research into Familial Breast Cancer (kConFab), which has been collecting biospecimens and clinical and epidemiological information from families recruited through Familial Cancer Centres in Australia and New Zealand since 1997 [30]. DNA from two or three breast cancer-affected individuals were obtained from each family for analysis (as shown in Table 1), at least one of whom had previously been screened for BRCA1 and BRCA2 mutations (by sequencing of all coding exons and Multiplex Ligation-dependent Probe Amplification). Blood DNA from index cases from a further 834 mutation negative kConFab families and 561 mutation negative families obtained from the Peter MacCallum Cancer Centre Familial Cancer Centre were obtained for mutation analysis of candidate genes. Of those index cases obtained through the Familial Cancer Centre, individuals were breast cancer-affected, had a strong family history and been assessed for the probability of harboring a BRCA1 or BRCA2 mutations using BRCAPRO [31] and had been found on the basis of a verified family and personal history of having a 10% or greater probability. The index cases had undergone full diagnostic BRCA1/2 mutation search and no mutation was identified. However, it should be noted that the majority of these families did not fulfill the very stringent family history criteria that was required for recruitment to kConFab, the research cohort from which the families for the initial exome sequencing were taken [30]. Non-cancer control DNA samples were obtained from kConFab (226 age - and ethnicity-matched best friend controls) and from the Princess Anne Hospital, UK (238 Caucasian female volunteers, as described previously [32]). DNA for candidate gene mutation analysis underwent whole genome amplification (WGA) using Repli-G Phi-mediated amplification system (Qiagen) prior to mutation analysis.
Whole-Exome Sequencing
2–3 µg of DNA was fragmented to approximately 200 bp by sonication (Covaris) and used to prepare single - or paired-end libraries using the SPRIworks Fragment Library System I for Illumina Genome Analyzer on the SPRI-TE Nucleic Acid Extractor (Beckman Coulter). Exome enrichment was performed using the NimbleGen Sequence Capture 2.1 M Exome Array, EZ Exome Library (Roche NimbleGen) or SureSelect Human All Exon version 2 or 50 Mb libraries (Agilent Technologies) according to the recommended protocols. Sequencing was performed on GAIIx or HiSeq instruments (Illumina). Library preparation and sequencing details for each sample are provided in Table S1. We did not observe any significant differences in performance of the different exome capture platforms.
Sequencing Alignment and Variant Calling
Paired-end sequence reads were aligned to the human genome (hg19 assembly) using the Burrows–Wheeler Aligner (BWA) program [33]. Local realignment around indels was performed using the Genome Analysis Tool Kit (GATK) software [34]. Subsequently, duplicate reads were removed using Picard and base quality score recalibration performed using GATK software. Single nucleotide variants (SNVs) and indels were identified using the GATK Unified Genotyper and variant quality score recalibration. Variants were annotated with information from Ensembl release 62 using Ensembl Perl Application Program Interface (API) including SNP Effect Predictor [35], [36]. Single-end sequence reads were aligned as above except duplicate reads were flagged prior to base quality score recalibration and included in variant calling.
Candidate Variant Identification
Variants were first filtered for confident calls originating from bidirectional sequence reads using a quality threshold of ≥30, read depth of ≥10 and allele frequency ≥0.15. Prior to further filtering, variants were assessed for overtly deleterious mutation in known breast cancer associated genes [25]. Then, all variants present in the dbSNP database v132, except those also reported in the public version of the Human Gene Mutation Database (HGMD) [37] were removed, as were all common variants detected in >10 out of 33 exomes. Next, variants with functionally deleterious consequences (nonsense SNVs, frameshift indels, essential splice variants and complex indels) were identified for evaluation [35]. Functionally deleterious variants were evaluated in each individual as well as pairwise between relatives.
Variant Validation Using Sanger Sequencing
Primers flanking the BRCA2, PTEN, FANCC and BLM mutations identified by whole exome sequence analysis were used to amplify germline DNA from affected index cases and all available relatives. The purified products were directly sequenced using BigDye terminator v3.1 chemistry on a 3130 Genetic Analyzer (Applied Biosystems).
Mutation Analysis of FANCC and BLM
High resolution melt (HRM) analysis was performed on duplicate PCR products amplified from 15 ng WGA DNA. Primer sequences and PCR conditions are provided in Table S5. Melt analyses were performed on a LightCycler 480 Instrument using Gene Scanning Software (Roche). Duplicate PCR products exhibiting variant DNA melt curves were Sanger sequenced to identify sequence variations. All novel sequence variants were confirmed by Sanger sequencing an independent PCR amplified from non-WGA DNA. The functional effect of missense variants were evaluated using in silico prediction tools SIFT and PolyPhen-2 [38], [39].
Accession Codes
The following GenBank reference sequences were used for variant annotation: FANCC, NM_000136 BLM, NM_000057; PTEN, NM_000314 and BRCA2, NM_000059.
Web Resources
1000 Genomes Browser, http://browser.1000genomes.org/; Ensembl, http://www.ensembl.org/index.html; The Genome Analysis Toolkit, http://www.broadinstitute.org/gsa/wiki/index.php/The_Genome_Analysis_Toolkit; HGMD, http://www.hgmd.org/; Picard, http://picard.sourceforge.net; HGVS nomenclature for the description of sequence variants, http://www.hgvs.org/mutnomen/; NCBI SNP database, http://www.ncbi.nlm.nih.gov/projects/SNP/; The Fanconi Anemia Mutation Database, http://www.rockefeller.edu/fanconi/; BLMbase mutation registry, http://bioinf.uta.fi/BLMbase/; SIFT, http://sift.jcvi.org/; PolyPhen-2, http://genetics.bwh.harvard.edu/pph2/. Exome Variant Server, http://evs.gs.washington.edu/EVS/.
Supporting Information
Zdroje
1. PharoahPD, LipscombeJM, RedmanKL, DayNE, EastonDF, et al. (2000) Familial predisposition to breast cancer in a British population: implications for prevention. Eur J Cancer 36 : 773–779.
2. MikiY, SwensenJ, Shattuck-EidensD, FutrealPA, HarshmanK, et al. (1994) A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266 : 66–71.
3. WoosterR, BignellG, LancasterJ, SwiftS, SealS, et al. (1995) Identification of the breast cancer susceptibility gene BRCA2. Nature 378 : 789–792.
4. FordD, EastonDF, StrattonM, NarodS, GoldgarD, et al. (1998) Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet 62 : 676–689.
5. MavaddatN, PharoahPD, BlowsF, DriverKE, ProvenzanoE, et al. (2010) Familial relative risks for breast cancer by pathological subtype: a population-based cohort study. Breast Cancer Res 12: R10.
6. ThompsonD, EastonD (2004) The genetic epidemiology of breast cancer genes. J Mammary Gland Biol Neoplasia 9 : 221–236.
7. DomchekSM, FriebelTM, SingerCF, EvansDG, LynchHT, et al. (2010) Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA 304 : 967–975.
8. Comino-MendezI, Gracia-AznarezFJ, SchiaviF, LandaI, Leandro-GarciaLJ, et al. (2011) Exome sequencing identifies MAX mutations as a cause of hereditary pheochromocytoma. Nat Genet 43 : 663–667.
9. JonesS, HrubanRH, KamiyamaM, BorgesM, ZhangX, et al. (2009) Exomic sequencing identifies PALB2 as a pancreatic cancer susceptibility gene. Science 324 : 217.
10. GibsonRA, HajianpourA, Murer-OrlandoM, BuchwaldM, MathewCG (1993) A nonsense mutation and exon skipping in the Fanconi anaemia group C gene. Human Molecular Genetics 2 : 797–799.
11. GermanJ, SanzMM, CiocciS, YeTZ, EllisNA (2007) Syndrome-causing mutations of the BLM gene in persons in the Bloom's Syndrome Registry. Hum Mutat 28 : 743–753.
12. StrathdeeCA, GavishH, ShannonWR, BuchwaldM (1992) Cloning of cDNAs for Fanconi's anaemia by functional complementation. Nature 356 : 763–767.
13. GavishH, dos SantosCC, BuchwaldM (1993) A Leu554-to-Pro substitution completely abolishes the functional complementing activity of the Fanconi anemia (FACC) protein. Human Molecular Genetics 2 : 123–126.
14. The 1000 Genomes Project Consortium (2010) A map of human genome variation from population-scale sequencing. Nature 467 : 1061–1073.
15. CouchFJ, JohnsonMR, RabeK, BoardmanL, McWilliamsR, et al. (2005) Germ line Fanconi anemia complementation group C mutations and pancreatic cancer. Cancer Research 65 : 383–386.
16. WangW (2007) Emergence of a DNA-damage response network consisting of Fanconi anaemia and BRCA proteins. Nat Rev Genet 8 : 735–748.
17. HeyerWD, EhmsenKT, LiuJ (2010) Regulation of homologous recombination in eukaryotes. Annu Rev Genet 44 : 113–139.
18. van der HeijdenMS, YeoCJ, HrubanRH, KernSE (2003) Fanconi anemia gene mutations in young-onset pancreatic cancer. Cancer Research 63 : 2585–2588.
19. BerwickM, SatagopanJM, Ben-PoratL, CarlsonA, MahK, et al. (2007) Genetic heterogeneity among Fanconi anemia heterozygotes and risk of cancer. Cancer Research 67 : 9591–9596.
20. BarisHN, KedarI, HalpernGJ, ShohatT, MagalN, et al. (2007) Prevalence of breast and colorectal cancer in Ashkenazi Jewish carriers of Fanconi anemia and Bloom syndrome. Isr Med Assoc J 9 : 847–850.
21. SealS, ThompsonD, RenwickA, ElliottA, KellyP, et al. (2006) Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nature Genetics 38 : 1239–1241.
22. SokolenkoAP, IyevlevaAG, PreobrazhenskayaEV, MitiushkinaNV, AbyshevaSN, et al. (2011) High prevalence and breast cancer predisposing role of the BLMc.1642 C>T (Q548X) mutation in Russia. International Journal of Cancer doi: 10.1002/ijc.26342.
23. GruberSB, EllisNA, ScottKK, AlmogR, KolachanaP, et al. (2002) BLM heterozygosity and the risk of colorectal cancer. Science 297 : 2013.
24. ClearySP, ZhangW, Di NicolaN, AronsonM, AubeJ, et al. (2003) Heterozygosity for the BLM(Ash) mutation and cancer risk. Cancer Res 63 : 1769–1771.
25. WalshT, LeeMK, CasadeiS, ThorntonAM, StraySM, et al. (2010) Detection of inherited mutations for breast and ovarian cancer using genomic capture and massively parallel sequencing. Proceedings of the National Academy of Sciences of the United States of America 107 : 12629–12633.
26. ZollnerS, PritchardJK (2007) Overcoming the winner's curse: estimating penetrance parameters from case-control data. Am J Hum Genet 80 : 605–615.
27. MeindlA, HellebrandH, WiekC, ErvenV, WappenschmidtB, et al. (2010) Germline mutations in breast and ovarian cancer pedigrees establish RAD51C as a human cancer susceptibility gene. Nature Genetics 42 : 410–416.
28. LovedayC, TurnbullC, RamsayE, HughesD, RuarkE, et al. (2011) Germline mutations in RAD51D confer susceptibility to ovarian cancer. Nature Genetics 43 : 879–882.
29. ThompsonER, BoyleSE, JohnsonJ, RylandGL, SawyerS, et al. (2012) Analysis of RAD51C germline mutations in high-risk breast and ovarian cancer families and ovarian cancer patients. Human Mutation 33 : 95–99.
30. OsborneRH, HopperJL, KirkJA, Chenevix-TrenchG, ThorneHJ, et al. (2000) kConFab: a research resource of Australasian breast cancer families. Kathleen Cuningham Foundation Consortium for Research into Familial Breast Cancer [letter]. med j aust 172 : 463–464.
31. BerryDA, IversenESJr, GudbjartssonDF, HillerEH, GarberJE, et al. (2002) BRCAPRO validation, sensitivity of genetic testing of BRCA1/BRCA2, and prevalence of other breast cancer susceptibility genes. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology 20 : 2701–2712.
32. BaxterSW, ChoongDY, EcclesDM, CampbellIG (2001) Polymorphic variation in CYP19 and the risk of breast cancer. Carcinogenesis 22 : 347–349.
33. LiH, DurbinR (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25 : 1754–1760.
34. McKennaA, HannaM, BanksE, SivachenkoA, CibulskisK, et al. (2010) The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Research 20 : 1297–1303.
35. McLarenW, PritchardB, RiosD, ChenY, FlicekP, et al. (2010) Deriving the consequences of genomic variants with the Ensembl API and SNP Effect Predictor. Bioinformatics 26 : 2069–2070.
36. RiosD, McLarenWM, ChenY, BirneyE, StabenauA, et al. (2010) A database and API for variation, dense genotyping and resequencing data. BMC Bioinformatics 11 : 238.
37. StensonPD, BallEV, HowellsK, PhillipsAD, MortM, et al. (2009) The Human Gene Mutation Database: providing a comprehensive central mutation database for molecular diagnostics and personalized genomics. Hum Genomics 4 : 69–72.
38. NgPC, HenikoffS (2001) Predicting deleterious amino acid substitutions. Genome Research 11 : 863–874.
39. AdzhubeiIA, SchmidtS, PeshkinL, RamenskyVE, GerasimovaA, et al. (2010) A method and server for predicting damaging missense mutations. Nat Methods 7 : 248–249.
Štítky
Genetika Reprodukční medicína
Článek Genome-Wide Association Study for Serum Complement C3 and C4 Levels in Healthy Chinese SubjectsČlánek Role of Transposon-Derived Small RNAs in the Interplay between Genomes and Parasitic DNA in RiceČlánek Tetraspanin Is Required for Generation of Reactive Oxygen Species by the Dual Oxidase System inČlánek An Essential Role of Variant Histone H3.3 for Ectomesenchyme Potential of the Cranial Neural CrestČlánek A Mimicking-of-DNA-Methylation-Patterns Pipeline for Overcoming the Restriction Barrier of BacteriaČlánek A Genome-Wide Association Study Identifies Five Loci Influencing Facial Morphology in Europeans
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2012 Číslo 9- Akutní intermitentní porfyrie
- Růst a vývoj dětí narozených pomocí IVF
- Vliv melatoninu a cirkadiálního rytmu na ženskou reprodukci
- Intrauterinní inseminace a její úspěšnost
- Délka menstruačního cyklu jako marker ženské plodnosti
-
Všechny články tohoto čísla
- Heterozygous Mutations in DNA Repair Genes and Hereditary Breast Cancer: A Question of Power
- GWAS of Diabetic Nephropathy: Is the GENIE out of the Bottle?
- The Conflict within and the Escalating War between the Sex Chromosomes
- Proteome-Wide Analysis of Disease-Associated SNPs That Show Allele-Specific Transcription Factor Binding
- Exome Sequencing Identifies Rare Deleterious Mutations in DNA Repair Genes and as Potential Breast Cancer Susceptibility Alleles
- A Gene Family Derived from Transposable Elements during Early Angiosperm Evolution Has Reproductive Fitness Benefits in
- Genome-Wide Association Study for Serum Complement C3 and C4 Levels in Healthy Chinese Subjects
- Role of Transposon-Derived Small RNAs in the Interplay between Genomes and Parasitic DNA in Rice
- Co-Evolution of Mitochondrial tRNA Import and Codon Usage Determines Translational Efficiency in the Green Alga
- SIRT6/7 Homolog SIR-2.4 Promotes DAF-16 Relocalization and Function during Stress
- CNV Formation in Mouse Embryonic Stem Cells Occurs in the Absence of Xrcc4-Dependent Nonhomologous End Joining
- Tetraspanin Is Required for Generation of Reactive Oxygen Species by the Dual Oxidase System in
- Citrullination of Histone H3 Interferes with HP1-Mediated Transcriptional Repression
- Variation in Genes Related to Cochlear Biology Is Strongly Associated with Adult-Onset Deafness in Border Collies
- The Long Non-Coding RNA Affects Chromatin Conformation and Expression of , but Does Not Regulate Its Imprinting in the Developing Heart
- Rif2 Promotes a Telomere Fold-Back Structure through Rpd3L Recruitment in Budding Yeast
- Is a Metastasis Susceptibility Gene That Suppresses Metastasis by Modifying Tumor Interaction with the Cell-Mediated Immunity
- The p38/MK2-Driven Exchange between Tristetraprolin and HuR Regulates AU–Rich Element–Dependent Translation
- Rare Copy Number Variants Contribute to Congenital Left-Sided Heart Disease
- A Genetic Basis for a Postmeiotic X Versus Y Chromosome Intragenomic Conflict in the Mouse
- An Essential Role of Variant Histone H3.3 for Ectomesenchyme Potential of the Cranial Neural Crest
- Characterization of Inducible Models of Tay-Sachs and Related Disease
- Hominoid-Specific Protein-Coding Genes Originating from Long Non-Coding RNAs
- Transcriptional Repression of Hox Genes by HP1/HPL and H1/HIS-24
- Integrative Genomic Analysis Identifies Isoleucine and CodY as Regulators of Virulence
- Convergence of the Transcriptional Responses to Heat Shock and Singlet Oxygen Stresses
- Genomics of Adaptation during Experimental Evolution of the Opportunistic Pathogen
- Enrichment of HP1a on Drosophila Chromosome 4 Genes Creates an Alternate Chromatin Structure Critical for Regulation in this Heterochromatic Domain
- Vsx2 Controls Eye Organogenesis and Retinal Progenitor Identity Via Homeodomain and Non-Homeodomain Residues Required for High Affinity DNA Binding
- The Long Path from QTL to Gene
- TCF7L2 Modulates Glucose Homeostasis by Regulating CREB- and FoxO1-Dependent Transcriptional Pathway in the Liver
- The Non-Flagellar Type III Secretion System Evolved from the Bacterial Flagellum and Diversified into Host-Cell Adapted Systems
- Complex Chromosomal Rearrangements Mediated by Break-Induced Replication Involve Structure-Selective Endonucleases
- Factors That Promote H3 Chromatin Integrity during Transcription Prevent Promiscuous Deposition of CENP-A in Fission Yeast
- A Mimicking-of-DNA-Methylation-Patterns Pipeline for Overcoming the Restriction Barrier of Bacteria
- Determinants of Human Adipose Tissue Gene Expression: Impact of Diet, Sex, Metabolic Status, and Genetic Regulation
- Genome-Wide Association Studies Identify Heavy Metal ATPase3 as the Primary Determinant of Natural Variation in Leaf Cadmium in
- Tethering of the Conserved piggyBac Transposase Fusion Protein CSB-PGBD3 to Chromosomal AP-1 Proteins Regulates Expression of Nearby Genes in Humans
- A Genome-Wide Association Study Identifies Five Loci Influencing Facial Morphology in Europeans
- Normal DNA Methylation Dynamics in DICER1-Deficient Mouse Embryonic Stem Cells
- H4K20me1 Contributes to Downregulation of X-Linked Genes for Dosage Compensation
- The NDR Kinase Scaffold HYM1/MO25 Is Essential for MAK2 MAP Kinase Signaling in
- Coevolution within and between Regulatory Loci Can Preserve Promoter Function Despite Evolutionary Rate Acceleration
- New Susceptibility Loci Associated with Kidney Disease in Type 1 Diabetes
- SWI/SNF-Like Chromatin Remodeling Factor Fun30 Supports Point Centromere Function in
- A Response Regulator Interfaces between the Frz Chemosensory System and the MglA/MglB GTPase/GAP Module to Regulate Polarity in
- Functional Variants in and Involved in Activation of the NF-κB Pathway Are Associated with Rheumatoid Arthritis in Japanese
- Two Distinct Repressive Mechanisms for Histone 3 Lysine 4 Methylation through Promoting 3′-End Antisense Transcription
- Genetic Modifiers of Chromatin Acetylation Antagonize the Reprogramming of Epi-Polymorphisms
- UTX and UTY Demonstrate Histone Demethylase-Independent Function in Mouse Embryonic Development
- A Comparison of Brain Gene Expression Levels in Domesticated and Wild Animals
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Enrichment of HP1a on Drosophila Chromosome 4 Genes Creates an Alternate Chromatin Structure Critical for Regulation in this Heterochromatic Domain
- Normal DNA Methylation Dynamics in DICER1-Deficient Mouse Embryonic Stem Cells
- The NDR Kinase Scaffold HYM1/MO25 Is Essential for MAK2 MAP Kinase Signaling in
- Functional Variants in and Involved in Activation of the NF-κB Pathway Are Associated with Rheumatoid Arthritis in Japanese
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



