-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaTRY-5 Is a Sperm-Activating Protease in Seminal Fluid
Seminal fluid proteins have been shown to play important roles in male reproductive success, but the mechanisms for this regulation remain largely unknown. In Caenorhabditis elegans, sperm differentiate from immature spermatids into mature, motile spermatozoa during a process termed sperm activation. For C. elegans males, sperm activation occurs during insemination of the hermaphrodite and is thought to be mediated by seminal fluid, but the molecular nature of this activity has not been previously identified. Here we show that TRY-5 is a seminal fluid protease that is required in C. elegans for male-mediated sperm activation. We observed that TRY-5::GFP is expressed in the male somatic gonad and is transferred along with sperm to hermaphrodites during mating. In the absence of TRY-5, male seminal fluid loses its potency to transactivate hermaphrodite sperm. However, TRY-5 is not required for either hermaphrodite or male fertility, suggesting that hermaphrodite sperm are normally activated by a distinct hermaphrodite-specific activator to which male sperm are also competent to respond. Within males, TRY-5::GFP localization within the seminal vesicle is antagonized by the protease inhibitor SWM-1. Together, these data suggest that TRY-5 functions as an extracellular activator of C. elegans sperm. The presence of TRY-5 within the seminal fluid couples the timing of sperm activation to that of transfer of sperm into the hermaphrodite uterus, where motility must be rapidly acquired. Our results provide insight into how C. elegans has adopted sex-specific regulation of sperm motility to accommodate its male-hermaphrodite mode of reproduction.
Published in the journal: . PLoS Genet 7(11): e32767. doi:10.1371/journal.pgen.1002375
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002375Summary
Seminal fluid proteins have been shown to play important roles in male reproductive success, but the mechanisms for this regulation remain largely unknown. In Caenorhabditis elegans, sperm differentiate from immature spermatids into mature, motile spermatozoa during a process termed sperm activation. For C. elegans males, sperm activation occurs during insemination of the hermaphrodite and is thought to be mediated by seminal fluid, but the molecular nature of this activity has not been previously identified. Here we show that TRY-5 is a seminal fluid protease that is required in C. elegans for male-mediated sperm activation. We observed that TRY-5::GFP is expressed in the male somatic gonad and is transferred along with sperm to hermaphrodites during mating. In the absence of TRY-5, male seminal fluid loses its potency to transactivate hermaphrodite sperm. However, TRY-5 is not required for either hermaphrodite or male fertility, suggesting that hermaphrodite sperm are normally activated by a distinct hermaphrodite-specific activator to which male sperm are also competent to respond. Within males, TRY-5::GFP localization within the seminal vesicle is antagonized by the protease inhibitor SWM-1. Together, these data suggest that TRY-5 functions as an extracellular activator of C. elegans sperm. The presence of TRY-5 within the seminal fluid couples the timing of sperm activation to that of transfer of sperm into the hermaphrodite uterus, where motility must be rapidly acquired. Our results provide insight into how C. elegans has adopted sex-specific regulation of sperm motility to accommodate its male-hermaphrodite mode of reproduction.
Introduction
A general feature of sexual reproduction is the generation of motile sperm that can navigate to an egg. To assist this process, males transfer their sperm along with seminal fluid, which enhances their reproductive success in a variety of ways (reviewed in [1], [2]). Seminal fluid factors promote sperm survival, motility and fertilizing ability both by directly interacting with sperm and by interacting with tissues of the female to make her reproductive tract a more permissive environment. These factors include seminal fluid-specific proteins, a variety of hormones, and energy sources [2]. In mammals, roles for seminal fluid factors include the regulation of sperm motility and capacitation and the modulation of immune function [2], [3]. Extensive analysis in Drosophila has identified many seminal fluid proteins and uncovered roles for several of these factors in sperm storage, sperm competition, female reproductive behavior and physiology, and other processes [4]. Due to their potential for influencing reproductive success, components of seminal fluid represent a forum for both conflict and cooperation between the sexes [1], [5].
The androdioecious nematode Caenorhabditis elegans provides an opportunity to analyze sperm development and function in a context where both sexes produce sperm and can differentially regulate gamete function to promote their fertility. Hermaphrodites are self-fertilizing; during development, they produce a store of “self” sperm, which can be used to fertilize their eggs. Males mate with and transfer sperm to hermaphrodites. Males are not required for reproduction to occur, and in their absence self sperm are used with extremely high efficiency; more than 99% of self sperm are used. However, if male sperm are present, then they preferentially fertilize eggs [6].
C. elegans sperm, like those of other nematodes, lack flagella; instead, they move by crawling using a pseudopod [6]–[9]. Motility is acquired during sperm activation, a process analogous to spermiogenesis in flagellate sperm, in which haploid spermatids undergo a dramatic cellular rearrangement to become competent for both directional motility and fertilization of an oocyte [6]. While most aspects of sperm development are similar in males and hermaphrodites, the timing and context of activation differ in the two sexes. In hermaphrodites, spermatids activate when they move into the spermathecae, regions of the gonad where sperm are stored and fertilization occurs. In males, sperm are stored in a non-activated form and become activated after mating and transfer to a hermaphrodite ([6] and unpublished observations). Sperm also can be activated in vitro in response to treatment with a variety of factors, including an ionophore (monensin), proteases (Pronase), a weak base (triethanolamine/TEA), and an ion channel inhibitor (4,4′-diisothiocyano-2,2′-stilbenedisulfonic acid/DIDS) [10]–[13]. This ability, together with the observation that sperm generally activate in vivo in response to a change in location, suggests that activation is controlled by extracellular signals.
Genes that regulate sperm activation show distinct requirements in hermaphrodites and males. The activity of a set of five genes termed the “spe-8 group” (spe-8, -12, -19, -27, and -29) is required specifically for hermaphrodites to activate their self sperm; hermaphrodites mutant for any one of these genes are self sterile, while mutant males are fertile [12], [14]–[19]. Mating of spe-8 group mutant hermaphrodites with males results in self-sperm activation (“transactivation”) and can restore self fertility, suggesting that males provide their own activator to which spe-8 group hermaphrodite sperm can respond [12]. spe-8 group functions are dispensable for production of this activator, since both wild-type and spe-8 group males are competent for transactivating hermaphrodite sperm. While these analyses indicate that there are differences in the intracellular pathways by which sperm are activated in the two sexes, the functions of individual activation genes are not strictly limited to a specific sex. spe-8 group mutant male sperm show some defects, failing to activate in response to Pronase in vitro [12], [15], [17], [19]. Furthermore, some spe-8 group activity is likely required for sperm to transactivate, since animals harboring spe-8 group null alleles appear to be insensitive to male activator [17], [19]. While most analysis has focused on hermaphrodite sperm activation, a gene with a male-biased effect has been identified as well. Activity of an extracellular trypsin inhibitor-like protein, SWM-1, is required in males to prevent premature activation from occurring prior to mating, and swm-1 mutant males are infertile due to failure to transfer activated sperm [20]. swm-1 activity is dispensable in hermaphrodites, though loss of swm-1 improves fertility in a sensitized spe-8 group mutant background [20]. The finding that a protease inhibitor regulates activation in males, combined with the ability of proteases to activate sperm in vitro, suggested that protease activity could signal activation in vivo. However, the endogenous activator has not been identified as yet in either sex.
Here, we report the identification of a trypsin-family serine protease, TRY-5, which has the properties expected of a male sperm activator. Loss of try-5 suppresses mutations in swm-1. Furthermore, during mating, TRY-5 is released from the somatic gonad and transferred along with sperm, thus coupling the onset of sperm motility to the time of their transfer to a hermaphrodite. Within the male gonad, TRY-5 activity must be held in check to ensure male fertility. Strikingly, TRY-5 is not required for male fertility, but strains lacking both try-5 and spe-8 group activation functions are totally sterile, confirming that while male and hermaphrodite sperm motility is induced by distinct signals, the two pathways are redundant. In summary, TRY-5 is the first factor demonstrated to be a transferred component of seminal fluid in C. elegans, where it plays a key role in male-specific regulation of sperm function.
Results
C. elegans male sperm activation is regulated by a protease
In wild-type males, sperm are stored in the inactive form within the seminal vesicle (Figure 1A) and become activated after transfer to a hermaphrodite ([6] and unpublished data). Mutations in the secreted protease inhibitor SWM-1 result in premature sperm activation within males (Figure 1B, 1A, [20]). We predicted that loss of activation-promoting factors should suppress this phenotype. To identify such factors, we performed genetic screens for suppressors of premature sperm activation caused by the partial loss-of-function alleles swm-1(me86) or swm-1(me66) (G.M.S., unpublished; [20]). Among the swm-1 suppressor mutants, we identified three alleles of the serine protease gene try-5 (Figure 1E and 1F). We subsequently obtained tm3813, a deletion affecting the 5′ end of the try-5 coding region (gift of S. Mitani, National Bioresource Project, Japan), and showed that it also suppressed swm-1(me86) (Figure 1F). Suppression of the premature activation phenotype in swm-1 try-5 double mutants was rescued by a genomic fragment containing the full-length try-5 gene (Figure 1G, Tables S1, S2, S4 and data not shown), confirming that try-5 was responsible for this effect.
Fig. 1. Mutations in the serine protease gene try-5 suppress premature sperm activation in swm-1 mutant males. 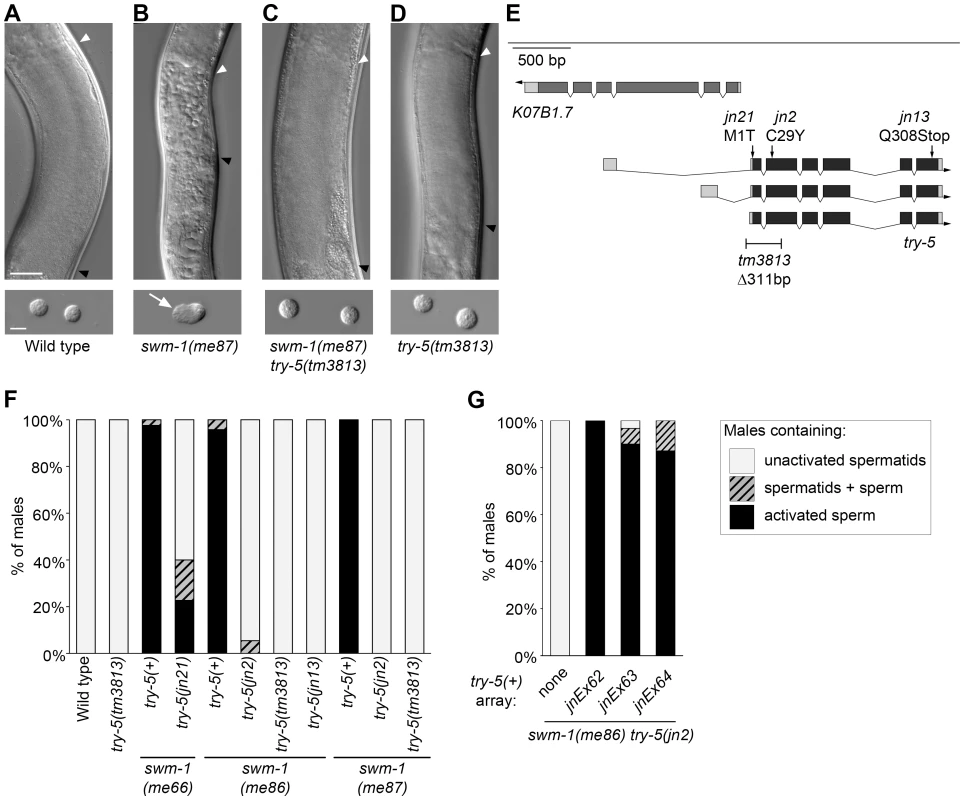
Sperm activation was examined in staged 48 hr post-L4 adult virgin males. (A–D) Differential interference contrast (DIC) images showing sperm morphology in the indicated strains. Top row: Images of intact males in the region of the seminal vesicle (SV), where sperm are stored. Strains shown in A, C and D have round spermatids, which pack together in the SV of intact animals to form a uniformly grainy appearance. The strain shown in B contains activated sperm, resulting in a rough appearance. Arrowheads indicate the anterior (white) and posterior (black) boundaries of the SV. Scale bar, 25 µm. Bottom row: DIC images of dissected sperm. Arrow indicates a pseudopod. Scale bar, 5 µm. (See Figure S1 for high-resolution versions of the images in B, C.) (E) Schematic of the try-5 region. We used RACE and RT-PCR to characterize try-5 transcripts, generating updated gene models as compared to the WormBase prediction ([59]; accession numbers JN651275, JN651276 and JN651277). On gene models, darker shading indicates predicted coding regions and arrow indicates the direction of transcription. The positions of mutations in try-5 are indicated along with their predicted effects. (See also Figure S2.) (F) Quantitation of suppression of swm-1 by mutations in try-5. Stacked columns indicate the percent of males containing either only activated sperm (black), a mixture of spermatids and activated sperm (hatched), or only non-activated spermatids (grey). At least 30 animals were scored for each genotype. (G) A try-5 transgene restores sperm activation in swm-1 mutant males. Sperm activation was assayed in unc-119; swm-1(me86) try-5(jn2) him-5 males bearing extrachromosomal arrays of pJRS14, which contains try-5(+) and C. briggsae unc-119(+) (Tables S1 and S2). Data from three independent transgenic lines are shown. Key for stacked columns as in (F). Between 22 and 31 animals were scored for each genotype. In parallel to our forward genetic screen, we also tested individual serine proteases for a role in sperm activation. We used RNA interference to reduce the function of individual protease genes in a swm-1 mutant background and screened for effects on premature activation in males. Among the tested proteases, only reduction of try-5 resulted in strong suppression (Materials and Methods and data not shown), consistent with our finding that try-5 is a regulator of sperm activation.
Based on conservation of its sequence and domain structure [21] with those of the trypsin-like superfamily, try-5 is predicted to encode a trypsin-class serine protease. This family of proteases contains numerous members in eukaryotes and regulates many processes, including blood coagulation, developmental signaling and fertilization [22]. Specific residues that form the protease active site are conserved in TRY-5, and the presence of a signal sequence on the N terminus of the protein suggests that it is secreted (Figure S2). While TRY-5 has clear orthologs in other closely related nematodes, it is divergent from serine proteases in more distantly related species (data not shown). In addition, its substrate-binding region is divergent from those of trypsin family members with characterized substrate specificities [23].
We initially identified try-5 using partial loss-of-function alleles of swm-1. To determine whether mutations in try-5 are capable of suppressing a swm-1 null, we examined animals harboring both the null allele swm-1(me87) and an allele of try-5. We found that whereas swm-1(me87) mutant males contain activated sperm [20], swm-1(me87) try-5(jn2) and swm-1(me87) try-5(tm3813) males contained non-activated sperm like those found in the wild type or in a try-5 mutant ([20], Figure 1A–1D and 1F, Figure S1). In summary, these results indicate that the protease TRY-5 is responsible for the premature sperm activation and associated loss of fertility that occur in swm-1 mutant males and suggest that the function of SWM-1 is to inhibit TRY-5 activity within the male.
Male sperm can activate in the absence of try-5
To see if try-5 is required for male sperm to activate, we assessed the ability of try-5 mutant sperm to respond to treatments that bypass normal activation signals. Wild-type sperm can be activated in vitro by treatment with any of a variety of compounds [10]–[13]. Since TRY-5 is predicted to be a protease, we first assayed the ability of try-5 mutant spermatids to activate in response to Pronase treatment. In the absence of Pronase, both wild-type and try-5 mutant sperm remained non-activated (Figure 2A and 2B). Within 5 to 10 min after addition of Pronase, the majority of sperm cells developed a pseudopod, consistent with activation (Figure 2A and 2C, Video S1). These cells were capable of motility, as they were observed crawling across the microscope slide (note altered positions of some cells in Figure 2B versus Figure 2C). There was no significant difference in either the level of activation (Figure 2A; P = 0.89, Student's t test) or the rate of activation (data not shown) of try-5 mutant sperm as compared to the wild type. We then tested the ability of try-5(tm3813) spermatids to activate in response to treatment with a second known activator, the weak base TEA. When treated with TEA, try-5 mutant spermatids activated at levels similar to wild-type sperm (data not shown). Thus, try-5 is not required for sperm activation initiated in vitro either by exogenous proteases or TEA. This result distinguishes try-5 mutants from the previously-characterized spe-8 group mutants, for which sperm activate normally when treated with TEA, but arrest at a partially-activated, “spiky” stage in response to Pronase [12], [15], [17], [19].
Fig. 2. try-5 mutant sperm are capable of activation. 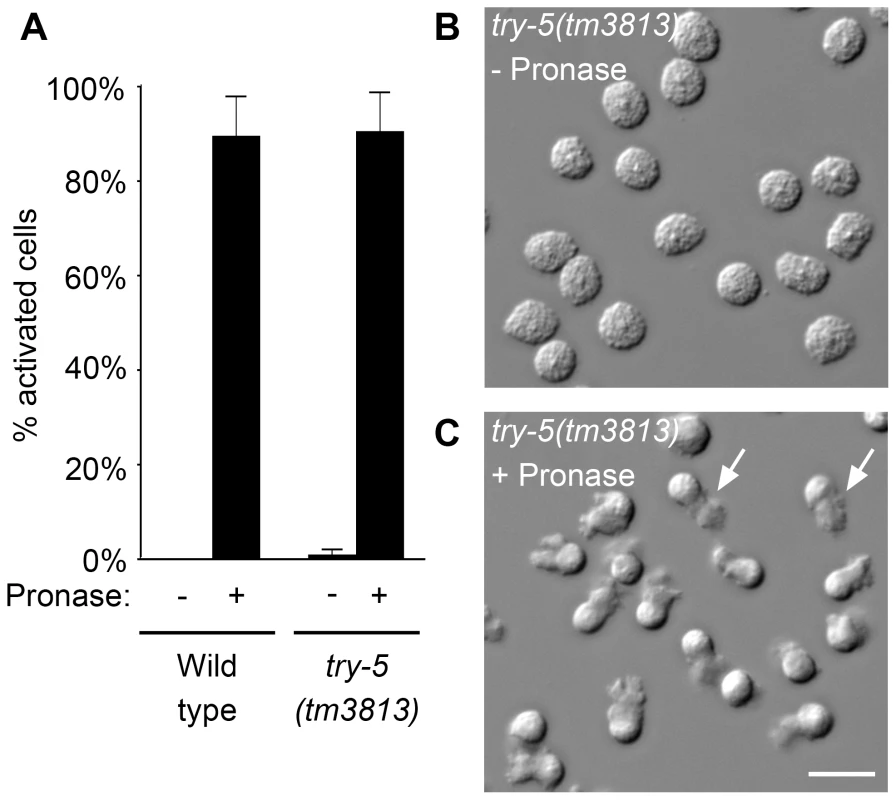
Sperm were assayed for activation in response to protease treatment. Wild-type or try-5(tm3813) mutant sperm were dissected and incubated in 200 µg/mL Pronase for 25 min; activation was scored every 5 min based on the presence or absence of a pseudopod. (A) Maximal activation observed at a single time point during the assay; average of 3 repeats. Error bars represent standard error of the mean. (B,C) DIC images of try-5(tm3813) sperm prior to Pronase treatment (B) and the same field of cells at 20 min (C). See also Video S1. Arrows indicate the pseudopodia for two of the activated cells. Scale bar, 10 µm. We next determined if try-5 is required for activation induced by loss of the intracellular activation inhibitor spe-6. SPE-6 is a sperm casein kinase 1-like protein that functions at two points during spermatogenesis: during spermatogenic cell divisions [24] and later during sperm activation [18]. Specific mutations in spe-6 allow spermatogenesis to occur but lead to premature sperm activation in males, a phenotype that is thought to be independent of extracellular signaling [18]. To determine whether try-5 function is required for the premature sperm activation phenotype of spe-6, we assayed sperm activation in spe-6(hc163); try-5(tm3813) and spe-6(hc163); try-5(jn2) mutant males. We found that, like spe-6(hc163) mutant males, spe-6(hc163); try-5 males contained activated sperm (Table 1) and their appearance was indistinguishable from that of the spe-6 mutant (data not shown). Thus, TRY-5 activity does not function downstream of the sperm protein SPE-6. Together, the ability of try-5 sperm to activate in response to either in vitro activators or loss of an intracellular inhibitor indicates that TRY-5 is not required for the subcellular rearrangements of sperm activation. Rather, these data suggest a regulatory role for this protease in signaling sperm to initiate the activation process.
Tab. 1. TRY-5 is not required for activation in spe-6 animals. 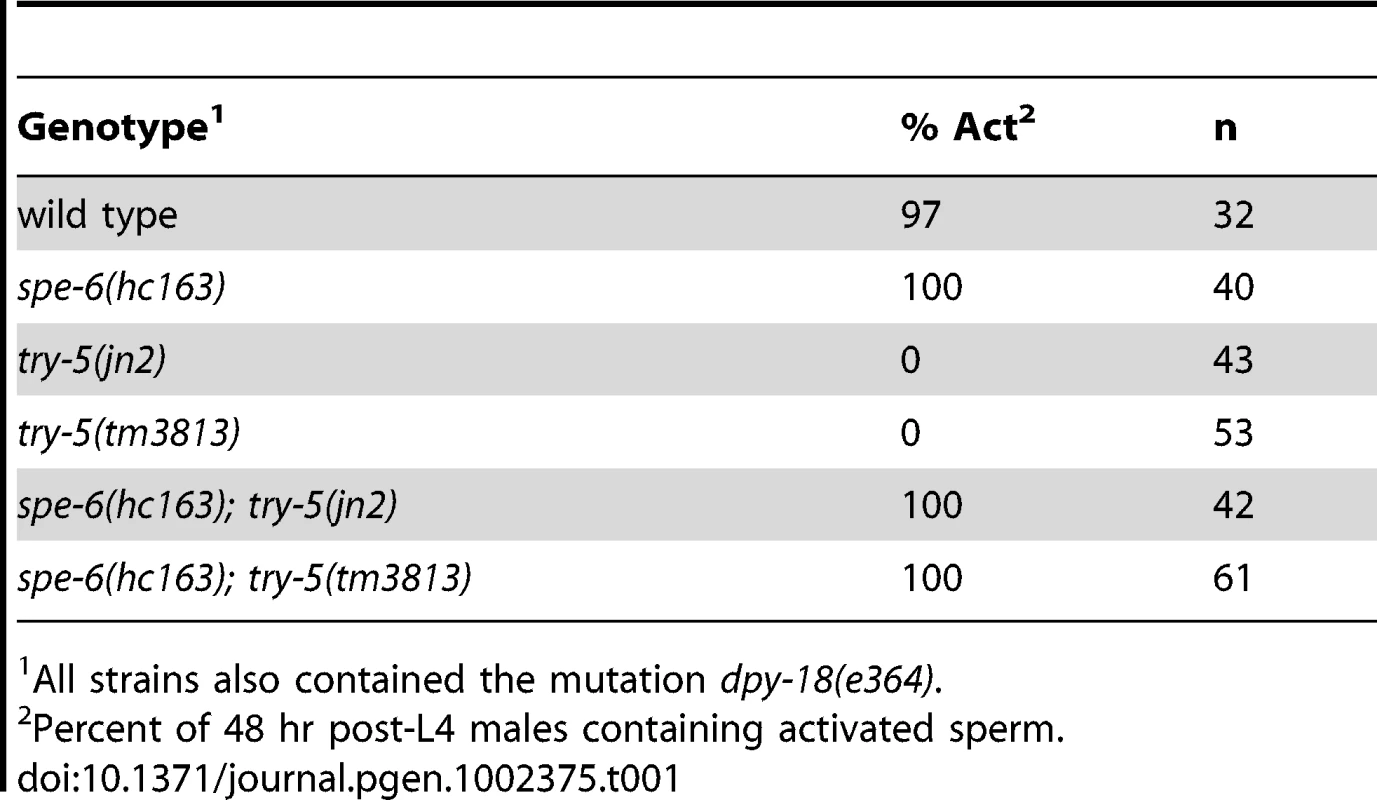
All strains also contained the mutation dpy-18(e364). try-5 is not required for fertility
Since activation is necessary to generate mature, motile spermatozoa that are competent for fertilization, failure to activate results in infertility. If try-5 is required for sperm activation, then loss of try-5 should result in decreased fertility. To test this idea, we assayed fertility in try-5 and swm-1(me87) try-5 hermaphrodites and males, using the try-5 alleles jn2 and tm3813. In self-fertilizing hermaphrodites, sperm is the limiting gamete for offspring production; nearly every self sperm in a hermaphrodite will fertilize an oocyte [6], so the total self brood size is a sensitive measure of the number of functional, activated sperm produced. We found no significant difference between the number of progeny produced by try-5 or swm-1 try-5 mutant hermaphrodites as compared to wild-type and swm-1 controls (Figure 3A, Figure S3A). Thus, try-5 is not required for hermaphrodite sperm activation or fertility.
Fig. 3. try-5 is not required for fertility and functions in parallel to the spe-8 group. 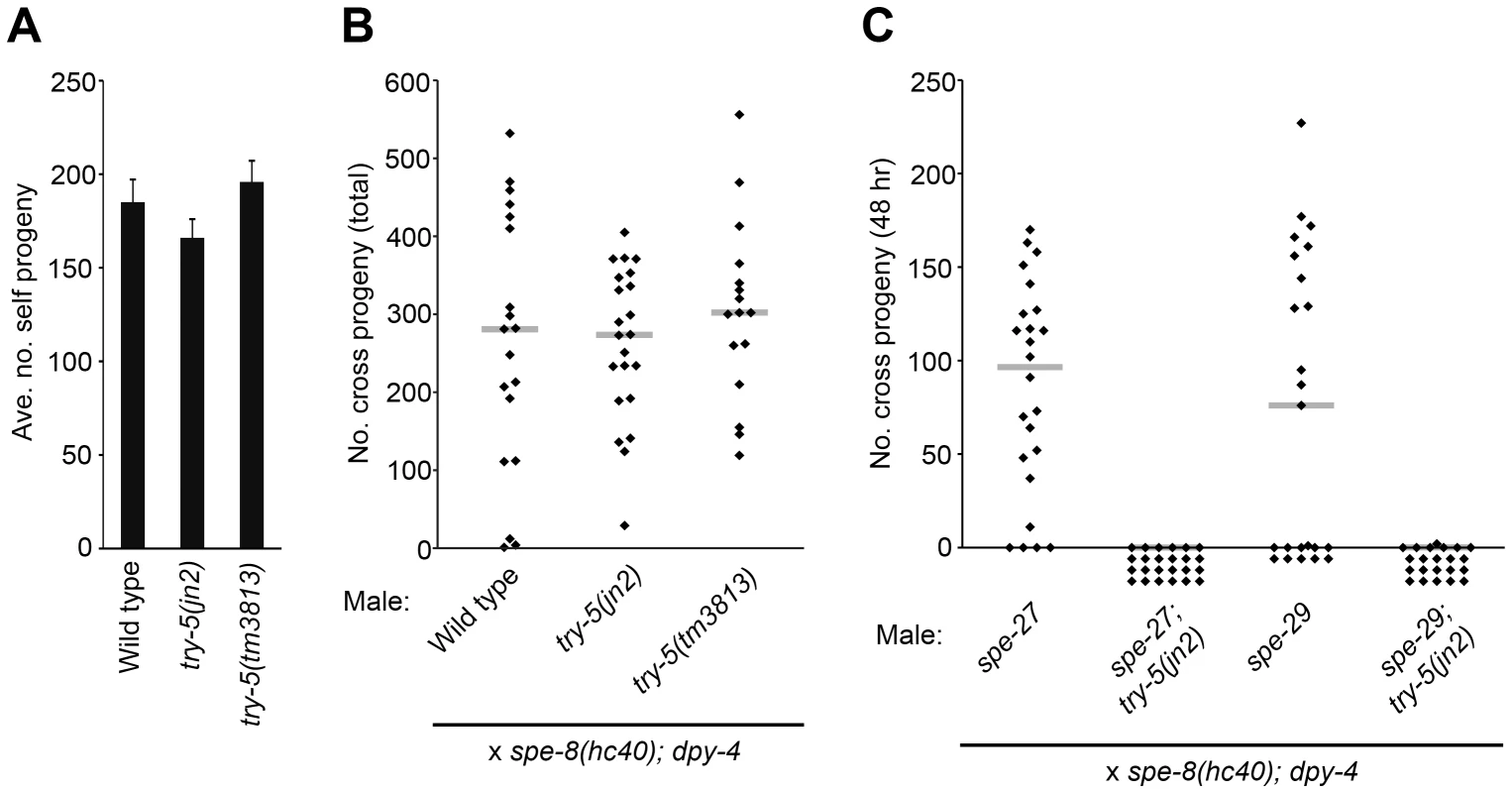
Hermaphrodite and/or male fertility was measured for try-5 mutant strains. (A) try-5 hermaphrodites show normal fertility. Columns indicate average brood size of self-fertilizing hermaphrodites. Error bars represent standard error of the mean. (B) try-5 males show normal fertility. Males were placed with spe-8(hc40); dpy-4 hermaphrodites in 1∶1 crosses for 48 hr and the entire brood size was measured by counting the total number of non-Dumpy cross progeny. try-5 mutants were not significantly different from the control (try-5(jn2), p = 0.95; try-5(tm3813), p = 0.40; Mann-Whitney U Test). (C) try-5 is required for fertility in spe-8 group mutant males. Males were placed with spe-8(hc40); dpy-4 hermaphrodites in 1∶1 crosses for 48 hr and the number of non-Dumpy cross progeny produced during the mating period was counted. Infertility of spe-8 group; try-5 males was due to failure of sperm to activate or migrate (Table S3). (B,C) Each point represents the result of an individual cross; gray lines represent medians. Sets of crosses with each genotype were repeated at least twice and a representative set of data is shown. (See also Figure S3.) We next measured male fertility in crosses of individual males to spe-8(hc40); dpy-4 recipient hermaphrodites. While there was a great deal of variation in the number of cross progeny produced even by wild-type males, as observed previously [25], try-5 mutant males showed a high level of fertility and no significant difference with the wild type was observed (Figure 3B). In addition, swm-1 try-5 males showed high levels of fertility, in some cases equivalent to that of the wild type (Figure S3B), along with suppression of the swm-1 transfer defect (data not shown). While our assays detected no obvious fertility defects in try-5 animals, it is possible that they might exhibit reduced fertility in other situations, e.g., outside the laboratory or under conditions of sperm competition. However, these results suggest that try-5 is not required for sperm activation or other aspects of fertility in either sex.
try-5 and the spe-8 group define two pathways for sperm activation
Although try-5 is not required for either male or hermaphrodite fertility, there is previous evidence for distinct pathways of sperm activation in males vs. hermaphrodites [12], [14], raising the possibility that the effect of try-5 loss is masked by functional redundancy. Therefore, we tested genes in the hermaphrodite pathway for redundancy with try-5. The activities of a set of five genes termed the “spe-8 group” (spe-8, -12, -19, -27, and -29) are required for self-sperm activation in the hermaphrodite but not for activation of male sperm (reviewed in [26]). To test whether try-5 and the spe-8 group function in independent, redundant activation pathways, we assayed sperm activation and male fertility in worms lacking both try-5 and spe-8 group activity, using the spe-8 group mutations spe-27(it110) and spe-29(it127). While spe-27 mutant males are fertile and capable of generating cross progeny, we found that spe-27; try-5 mutant males were completely infertile (Figure 3C). Similarly, while spe-29 mutant males are fertile, spe-29; try-5 fertility was greatly reduced as compared to the wild type (Figure 3C).
To investigate the cause of this infertility, we labeled males with MitoTracker [27] and crossed them to unlabeled recipient hermaphrodites to assay sperm transfer and migration [20]. We found that spe-27; try-5 males were able to transfer sperm to hermaphrodites, but the transferred sperm did not migrate. Similarly, for spe-29; try-5 males, we observed only rare instances of successful migration (Table S3 and data not shown). To determine if the migration defect was due to improper activation or a defect in migration after sperm activation, we dissected hermaphrodites immediately after their mating to spe-27; try-5 males and examined transferred, MitoTracker-labeled sperm. We found that whereas spe-27 sperm activate within fifteen minutes after transfer to a hermaphrodite, spe-27; try-5 sperm fail to activate (data not shown). Thus, spe-27; try-5 males are infertile due to failure to activate sperm upon transfer to hermaphrodites. Our findings of residual fertility and sperm migration in spe-29; try-5 males are consistent with previous observations [17] that the single known mutation in spe-29 leads to a weaker phenotype as compared to known null mutations in other spe-8 group genes. These results suggest that try-5 activity is the source of fertility in spe-8 group mutant males; i.e., the spe-8 group and try-5 function in two separate pathways for sperm activation, and either pathway is normally sufficient for full male fertility.
try-5 mutant males do not transfer activator to hermaphrodites
To determine whether try-5 indeed functions in the male-derived activation pathway, we used a specific assay to measure transfer of functional male activator. Wild-type male seminal fluid is capable of activating spe-8-group mutant hermaphrodite sperm during mating; this process is termed “transactivation” and is generally assayed using fer-1 mutant males, which are defective for producing functional sperm, to prevent cross-progeny production [12]. We crossed either fer-1 [25] or fer-1; try-5 males to spe-8(hc53); dpy-4 hermaphrodites and counted the number of self progeny generated. Crosses with fer-1 control males resulted in transactivation approximately 58% of the time. However, fer-1; try-5 males were rarely if ever capable of transactivating hermaphrodite sperm (Figure 4). To exclude the possibility that fer-1; try-5 males simply might harbor a behavioral defect that reduced their mating frequency, we used MitoTracker to label males and assessed their ability to transfer sperm. We observed similar frequencies of hermaphrodites containing labeled sperm after incubation with fer-1 males, fer-1; try-5(jn2) males, or fer-1; try-5(tm3813) males (43%, 57%, or 63%, respectively). These data indicate that fer-1; try-5 mutants mate and transfer sperm with similar success rates as compared to the control. Thus, try-5 mutant males are defective in transfer of the male activator responsible for transactivation of spe-8 group hermaphrodite sperm.
Fig. 4. TRY-5 is required for the activation of hermaphrodite sperm by male seminal fluid. 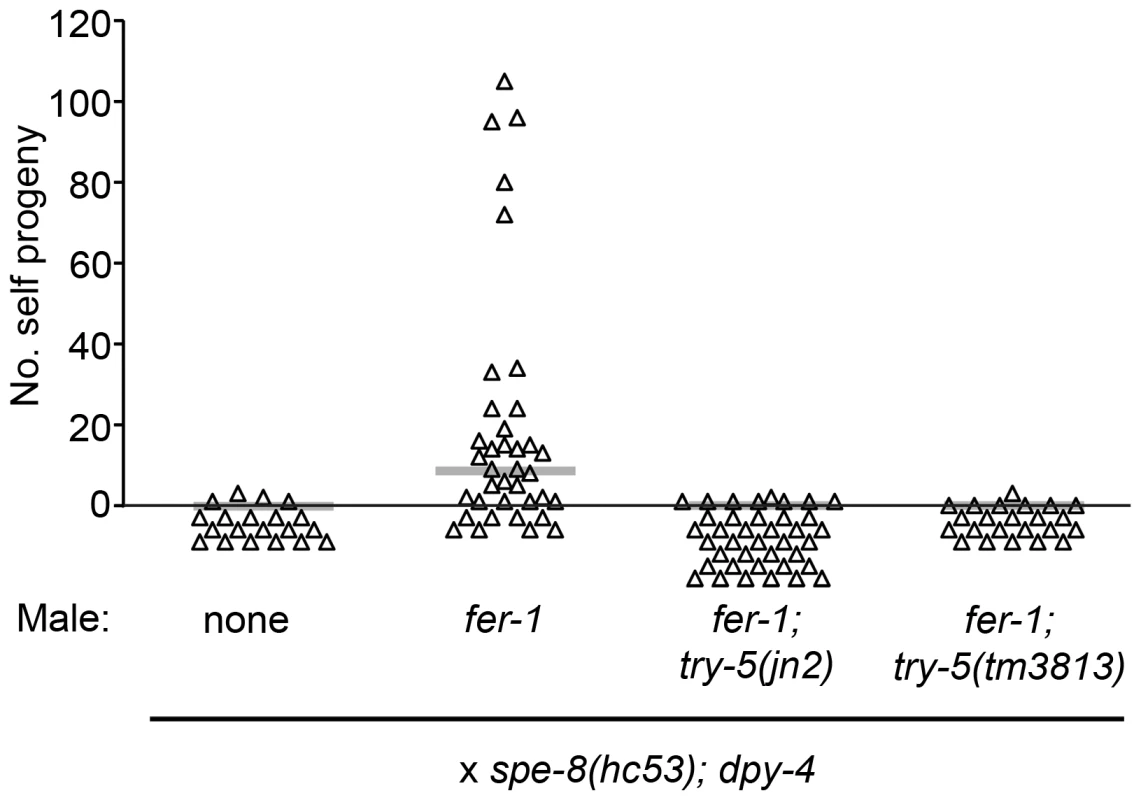
Transactivation was assayed for try-5 males. L4 males were mated to L4 spe-8(hc53); dpy-4 hermaphrodites in 4∶1 crosses for 48 hr, and the number of Dumpy self progeny produced during the mating period was counted. Each point represents the result from one cross. Gray bars represent medians. TRY-5 is expressed in and secreted from the male somatic gonad
To determine how TRY-5 functions in male sperm activation, we sought to determine where it is expressed and localized. Since we predicted that TRY-5 protein is secreted, we generated both a Ptry-5::GFP::H2B transcriptional reporter, a histone-H2B fusion that localizes to cell nuclei and facilitates identification of cells, and a Ptry-5::TRY-5::GFP translational reporter for assessing TRY-5 protein localization and function. We created stable transgenic worm strains using MosSCI (Mos1-mediated Single Copy gene Insertion [28], Tables S1 and S2) and confirmed that the Ptry-5::TRY-5::GFP transgene restored a premature sperm activation phenotype to swm-1 try-5 mutants (Materials and Methods, Tables S1, S2, S4).
Using the Ptry-5::GFP::H2B reporter, we found that the primary site of try-5 expression was in the male somatic gonad, in particular within tissues involved in storing sperm and tissues through which sperm pass during transfer to a hermaphrodite. The C. elegans male gonad is essentially a long tube. At the distal end of this tube, germline stem cells reside and proliferate, and as they move proximally, they undergo meiosis and differentiate into spermatids. A subset of somatic gonadal cells surround spermatids to form a storage organ, the seminal vesicle; a more proximal set forms a channel, the vas deferens, through which sperm move during transfer. A valve structure regulates movement of sperm between the seminal vesicle and vas deferens. The vas deferens contains at least two distinct cell types, based on shape: cuboidal and elongated cells [29]. Beyond an obvious structural role, other functions of these different cell types are not known, although some of them appear to be involved in secretion [29]. Starting at the L4 larval stage, when sperm production initiates, we observed Ptry-5::GFP::H2B expression in several regions of the male gonad (Figure 5A): the seminal vesicle (up to seven of the twenty-three total cells in this tissue [30]), the valve region (four cells), and the twelve cuboidal cells of the vas deferens [29]. This overall pattern persisted into adulthood until at least 72 hr post L4; highest expression levels were present consistently in the valve region. We observed no expression in the hermaphrodite gonad, so gonadal expression of try-5 is sexually dimorphic. However, we also observed low levels of expression in a few cells within the head and tail of both males and hermaphrodites (data not shown).
Fig. 5. try-5 is expressed in the male somatic gonad. 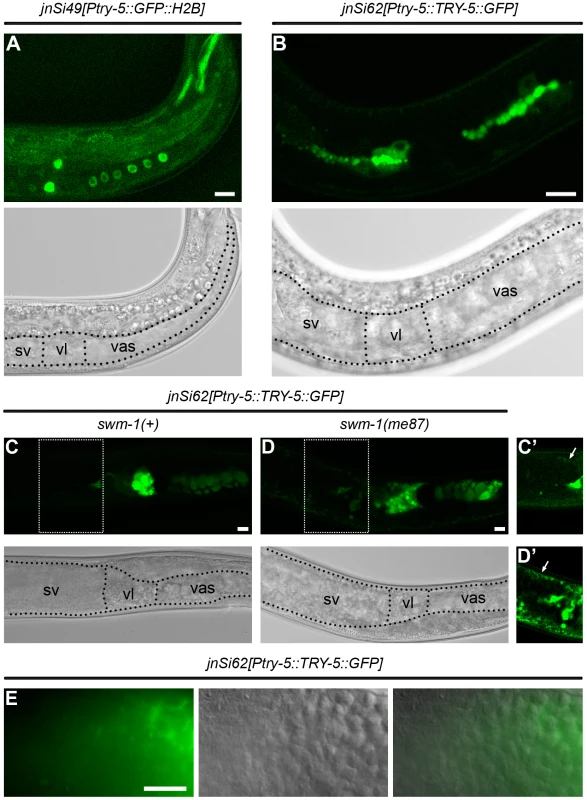
Confocal and transmitted-light (TL) images of transgenic males bearing try-5 reporter insertions. In TL images, boundaries of regions of the somatic gonad are labeled as sv, seminal vesicle; vl, valve; and vas, vas deferens. Scale bars, 10 µm. (A) jnSi49[Ptry-5::GFP::H2B]; try-5(tm3813) L4 male. In focal planes shown, GFP is visible in the four GFP-positive seminal vesicle cells, two of four GFP-positive valve cells, and six of twelve GFP-positive cuboidal cells. (B) jnSi62[Ptry-5::TRY-5::GFP]; try-5(tm3813) L4 male. TRY-5::GFP is concentrated at the apical side of seminal vesicle, valve and cuboidal cells. (C) jnSi62[Ptry-5::TRY-5::GFP]; try-5(tm3813) 48 hr adult male. Both large and small TRY-5::GFP globules are present in the cuboidal cells and valve region. In the seminal vesicle, TRY-5::GFP is present in the proximal region near the valve. (D) jnSi62[Ptry-5::TRY-5::GFP]; swm-1(me87) try-5(tm3813) 48 hr adult male. TRY-5::GFP has expanded into the seminal vesicle lumen and sperm are activated. (C′, D′) Images of seminal vesicle regions outlined in C and D with intensity levels optimized for the fainter GFP signal in these tissues. Arrows indicate GFP in the distal sheath-like cells of the seminal vesicle. (E) Paired DIC, epifluorescence and merged images of a jnSi62[Ptry-5::TRY-5::GFP]; try-5(tm3813) 72 hr adult male in the region of the seminal vesicle. Localization of TRY-5::GFP is correlated with activated sperm (also see Table S4 and S5). In worms carrying the Ptry-5::TRY-5::GFP reporter, the TRY-5::GFP fusion protein exhibited a localization pattern consistent with secretion from the vas deferens. Within the valve and cuboidal cells, TRY-5::GFP was localized to globular foci. In L4 larvae, most globules aligned with the apical domain that lines the developing sperm channel (Figure 5B). In mature adults, very large globules were present that tended to cluster apically, and additional small globules were present throughout the cytoplasm (Figure 5C and 5D). Such large globular structures are generally visible in adult males by DIC microscopy and diagnostic of vas deferens tissue, including within wild-type animals lacking a transgene. Based on their size and location, these large globules are likely to represent the “secretory globules” observed by electron microscopy [29].
We sometimes observed TRY-5::GFP within the lumen of the seminal vesicle, likely as a result of release from the adjacent, highly-expressing valve cells (Figure 5D and 5E, Table S5). The timing and extent of TRY-5::GFP expansion into the seminal vesicle was dependent on activity of the protease inhibitor SWM-1, the level of expression, and male age. In animals wild-type for swm-1, TRY-5::GFP was usually restricted to the valve cells or regions close by; when present near sperm cells, TRY-5::GFP was usually localized to a few discrete foci (data not shown). However, in animals lacking swm-1 activity, we often observed large zones of TRY-5::GFP extending from the valve and surrounding sperm in the seminal vesicle (compare Figure 5C and 5D; see Table S5). Even in swm-1(+) animals, when high levels of TRY-5::GFP were present in the seminal vesicle, we almost always observed that sperm were activated (Figure 5E, Tables S4 and S5). Together, these data suggest that TRY-5 is produced by cells of the male somatic gonad and can induce sperm activation within males if it is released into the seminal vesicle. It has been observed previously that older wild-type males sometimes contain activated sperm [20], and the finding that TRY-5::GFP is released into the seminal vesicle in older males provides a basis for this phenotype. Thus, these results support a model in which TRY-5 acts locally on sperm, either to signal their activation or to generate such a signal, and SWM-1 acts to inhibit the accumulation and/or activity of TRY-5 in the seminal vesicle.
TRY-5 is transferred to hermaphrodites during mating
Since TRY-5 localization is consistent with secretion from the male gonad, we sought to determine whether TRY-5 is transferred during mating. We placed individual MitoTracker-labeled Ptry-5::TRY-5::GFP; try-5(tm3813) males with unc-52 hermaphrodites, monitored the males for mating behavior [31], and acquired fluorescence images starting at or just before spicule insertion. We observed that TRY-5::GFP was transferred to hermaphrodites during mating (Figure 6 and Video S2). Shortly after spicule insertion, TRY-5::GFP was released from the vas deferens and transferred to the hermaphrodite (Figure 6A and 6B). A brief pause without obvious transfer then occurred (Figure 6C). Next, TRY-5::GFP was released from the valve cells and travelled rapidly through the vas deferens into the hermaphrodite (Figure 6D and 6E). Movement of this valve pool was immediately followed by transfer of sperm (data not shown). After transfer, the TRY-5::GFP signal dispersed throughout the uterus (Figure 6F) and remained visible near the vulva for several minutes, if eggs were not laid immediately. This stereotypical series of events occurred for all cases (n = 5) in which the entire process was observed from spicule insertion to sperm transfer. We also observed a partial time course of five other matings, all of which were consistent with this sequence of events.
Fig. 6. TRY-5 is transferred to hermaphrodites during mating. 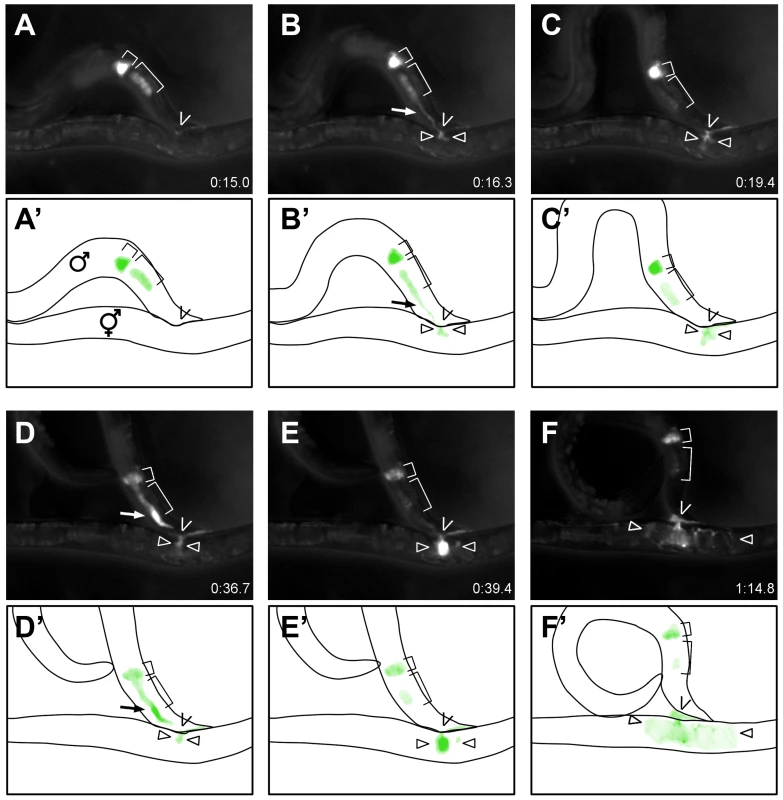
Selected still images (A–F) and schematics (A′–F′) depicting the time course of TRY-5::GFP transfer to an unc-52 hermaphrodite (see Video S2). (A) TRY-5::GFP is visible within its source cells in the valve (small bracket) and vas deferens (large bracket). (B) Transfer of TRY-5::GFP from the vas deferens into the hermaphrodite. (C) Pause between transfer of the two pools. (D, E) Transfer of TRY-5::GFP from the valve into the hermaphrodite. (F) Spread of TRY-5::GFP within the uterus concomitant with transfer of sperm (data not shown). Arrows indicate TRY-5::GFP during transfer, “V” symbols indicate position of the hermaphrodite vulva, and arrowheads indicate TRY-5::GFP within the uterus. Time shown is relative to the beginning of Video S2. To confirm that this behavioral sequence is not unique to this specific hermaphrodite genotype, we mated Ptry-5::TRY-5::GFP; try-5(tm3813) males to either unc-31 (n = 4) or him-5 unc-76 (n = 3) hermaphrodites. We were unable to observe vas deferens TRY-5::GFP transfer in these cases due to excess hermaphrodite movement. However, we did observe that valve TRY-5::GFP transfer initiated approximately 15–55 sec after spicule insertion, which is similar to the time observed for mating with unc-52 hermaphrodites (Figure 6, Video S2 and data not shown) and consistent with the reported timing for sperm transfer from 14.4 to 90.2 sec after spicule insertion as determined by Schindelman [32]. In summary, our data suggest that TRY-5 is a seminal fluid protein that is transferred to the hermaphrodite during copulation. Furthermore, our observations indicate that seminal fluid is released in discrete pools from specific tissues of the male gonad and that these events occur largely prior to and coincident with transfer of sperm.
Discussion
Activation of C. elegans sperm motility by a protease signal
We have identified a serine protease, TRY-5, which functions in C. elegans male sperm activation, the process by which amoeboid sperm cells become motile and competent to fertilize an egg. Based on our analysis of the defects of try-5 mutants and the dynamic localization of a TRY-5 reporter, we propose that TRY-5 is a sperm activating signal (Figure 7A and 7C). TRY-5 function is required for premature activation of stored sperm in males lacking the protease inhibitor SWM-1. TRY-5::GFP is expressed by the male somatic gonad within secretory cells. When observed outside these cells, localization of TRY-5::GFP protein strongly correlates with the localization of activated sperm. We have directly observed the transfer of TRY-5::GFP to hermaphrodites during copulation, and try-5 mutant males are incapable of transferring sperm activator to hermaphrodites. Together, these data strongly support a model in which TRY-5 is a component of seminal fluid that is transferred during copulation to signal sperm activation. Coupling the exposure of sperm to TRY-5 to the timing of transfer serves to ensure that sperm motility is rapidly induced at the time of - but not before - entry into a hermaphrodite's reproductive tract, thereby promoting male fertility.
Fig. 7. Models for the protease regulation of sperm activation in males and hermaphrodites. 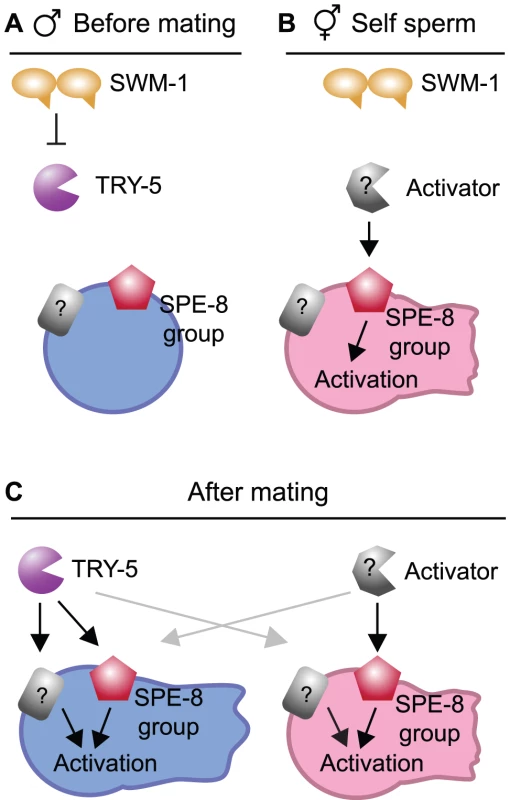
(A) Model: Male pathway for sperm activation, shown prior to mating when SWM-1 inhibits TRY-5 activity within the male gonad to prevent premature activation. In the absence of SWM-1, activation is signaled through interaction between TRY-5 and spermatids, likely through cleaving a target(s) on the cell surface (black arrows). Potential targets of TRY-5 include both members of the SPE-8 group and one or more additional sperm surface proteins. (B) Model: Hermaphrodite pathway for self-sperm activation. Activation is signaled through interaction between hermaphrodite activator and spermatids in the spermathecae (black arrow). This process is dependent on activity of the SPE-8 group. SWM-1 weakly antagonizes self sperm activation, suggesting that the hermaphrodite activator may be a protease. (C) Model: Pathways for activation after mating and seminal fluid transfer. When mating occurs, components of male and hermaphrodite pathways are both present and can promote sperm activation. Either pathway can activate both male and hermaphrodite sperm (grey arrows). However, hermaphrodite activator may function solely through the SPE-8 group. Our discovery of a seminal fluid serine protease provides a mechanistic explanation for previous results linking extracellular protease activity with sperm activation in C. elegans and in other nematodes. C. elegans sperm can be activated in vitro by incubation with Pronase, a protease preparation that primarily contains trypsin-like activity at the pH used for these assays [11]. In C. elegans males, loss of the SWM-1 protease inhibitor, which should result in increased protease activity, results in increased activation [20]. Recent studies of sex determination in C. remanei, a male-female species, showed that females could be transformed into sperm-producing “pseudohermaphrodites,” but their sperm were not motile; production of functional, activated sperm could be achieved through additional inhibition of the C. remanei orthologue of swm-1 [33]. Finally, the somatic gonad of males from the related nematode Ascaris suum contains a protease activity, which can activate sperm [34], [35]. Thus, a role for protease activity in promoting sperm motility appears to be conserved among nematodes.
Here we describe a novel role for a protease as a signaling molecule for differentiation of sperm to a motile form. Why would a protease be used in this context? The onset of motility in C. elegans sperm, as in flagellate sperm, occurs at a stage subsequent to meiotic cell division and the compaction of the haploid genome. At this stage, C. elegans sperm cells no longer express new protein products [26]. Therefore, to alter their behavior they must either reorganize their cellular contents in response to their environment or take in external factors. In addition, the timing of activation must be tightly controlled: C. elegans sperm must become motile rapidly upon entry into the hermaphrodite to avoid being lost due to the continuous outward passage of eggs [6], but early activation of motility precludes transfer of sperm from the male [20]. A protease activator provides a mechanism to trigger irreversible changes in the sperm cell surface that is readily coupled to mixing of sperm with seminal fluid. This type of activator also provides a simple mechanism to hold activation in check: the use of specific protease inhibitors such as SWM-1. We propose that the balance of TRY-5 and SWM-1 activities controls the likelihood of activation in specific locations and times within the male and hermaphrodite (Figure 7). For example, within the male gonad, SWM-1 may directly inhibit TRY-5 activity to prevent activation, allowing for sperm transfer and maintaining male fertility (Figure 7A). It is likely that additional proteases and/or inhibitors also function in this process. try-5 mutant hermaphrodites are fertile, suggesting that hermaphrodites have an activator that is independent of TRY-5 (Figure 7B). This activator could be a protease, though its identity is not known. Male sperm sometimes activate prematurely in try-5 mutants, suggesting that males could contain a second activator. However, if it exists, such a secondary male activator must not be competent to activate male or hermaphrodite spe-8 group sperm.
Genetic analysis of swm-1 had suggested that it functions to inhibit two distinct protease activities that act in parallel to promote sperm activation within males [20]. This model was based on the result that partial loss-of-function mutations affecting each of the two TIL domains of SWM-1 partially complement one another. By this model, loss of a single protease would not be expected to block sperm activation. However, we find that all SWM-1 activity works through TRY-5 in males, suggesting that both domains of SWM-1 inhibit TRY-5. The apparently separable activities of the SWM-1 TIL domains could arise from interactions with factors other than proteases. Alternatively, these results can be reconciled by a regulatory model in which SWM-1 inhibits two distinct proteases, both of which act upstream of TRY-5. It is also possible that SWM-1 might inhibit both TRY-5 and a second, TRY-5-activating protease. Consistent with these ideas, many well-known protease pathways consist of sequential cascades of activator and effector functions (e.g., [36], [37]).
As an extracellular protease, TRY-5 likely signals activation by cleaving sperm cell surface proteins and altering their activity. Some of the targets of TRY-5 may be SPE-8 group proteins, based on the fact that TRY-5 is required for transactivation, a process dependent on having some spe-8 group activity (sperm from hermaphrodites harboring null alleles of these genes are essentially incapable of being transactivated [16], [19]). However, spe-8 group mutant males are fertile, suggesting that SPE-8 group proteins are not essential for activation in all contexts. Thus, other targets may not be members of the SPE-8 group. The existence of such targets is further supported by our finding of additional swm-1 suppressors (distinct from try-5) that show full fertility in hermaphrodites and so do not fall into the spe-8 phenotypic class (G.M.S., unpublished data).
Could TRY-5 be functioning in some role other than as a direct activator? Sperm from try-5 mutant males can be activated within hermaphrodites after mating, in spe-6 mutants, or by exogenous activators in vitro. Thus, other activators can bypass TRY-5, and try-5 is not required for the cellular rearrangements that occur after activation is triggered. These data support the idea that TRY-5 functions in a regulatory step of the activation process. It is clear that TRY-5 is essential for transfer of sperm activator by C. elegans males and its localization correlates strongly with that of activated sperm. These data strongly suggest that if TRY-5 is not the signaling molecule per se, its activity is intimately associated with generation of the sperm activation signal.
TRY-5 as a component of seminal fluid
Production and transfer of seminal fluid is an important aspect of male reproduction [2], [38], [39]. TRY-5 is one of the first seminal fluid proteins identified in C. elegans. Indeed, it is the first directly demonstrated to be transferred at mating, and the first with a specific role in promoting gamete function. Previously, plg-1 was identified as a seminal fluid factor required for production of a copulatory plug [40] and shown to encode a mucin-like protein with a function in male mate guarding [41]. plg-1 is expressed within the male somatic gonad in a subset of cells that express try-5; interestingly, plg-1 is not expressed within the valve region [41], from which most TRY-5 appears to be released during mating (Video S2). Thus, as in other animals [42], [43], different regions of the C. elegans male gonad appear to be specialized to produce specific components of seminal fluid. Furthermore, our data reveal considerable complexity in the timing of release of seminal fluid from specific tissues during the mating behavioral program.
Regulatory logic of sperm activation in a male-hermaphrodite species
We have found that try-5 is functionally redundant for fertility in C. elegans. Although try-5 mutant males fail to transfer activator, they are fertile; however, loss of both try-5 and spe-8-group function leads to complete infertility for both hermaphrodites and males (tested here with mutations in two of the spe-8-group genes, spe-27 and spe-29). These data can be explained by the following model: spe-27; try-5 and spe-29; try-5 animals (1) make sperm that do not respond to hermaphrodite activator (due to loss of spe-8-group function) and (2) do not produce male activator (due to loss of try-5). In other words, try-5 males may be fertile due not to the presence of additional activators provided by the male, but rather due to rescue of male sperm activation by a signal within the hermaphrodite (Figure 7C).
These findings of redundancy raise the question: why does C. elegans have try-5? At least part of the answer might lie in the evolutionary history of this species, which evolved from a gonochoristic (male-female) ancestor [44], [45]. As the male activator, try-5 may represent the ancestral mode of activating sperm. Baldi et al. [33] have shown that the transition from gonochorism to androdioecy in the related species C. remanei requires only two steps: making sperm and activating it. Acquisition of the ability to make sperm could be advantageous, even in the initial absence of a robust self-sperm activation mechanism, as long as it tended to increase fertility. Chance encounters with a male would potentially activate hermaphrodite self sperm, as long as hermaphrodite sperm remained capable of responding to male activator. In turn, the male may have developed mechanisms to ensure his sperm were used preferentially; indeed, C. elegans male sperm show strong precedence over those of the hermaphrodite [6], [46]. Eventually, the hermaphrodite might evolve her own mechanism for activating sperm. The self-sperm activator in C. elegans is not known, but it may be a serine protease. Indirect evidence for this idea is provided by data indicating that the inhibitor SWM-1 functions in hermaphrodites: while animals mutant for the spe-8 class gene spe-29 have very low levels of self sperm activation and fertility, this phenotype is partially suppressed by mutations in swm-1 [20]. However, this protease is likely distinct from TRY-5, since we have found that try-5 is not required for either normal hermaphrodite fertility or increased activation in spe-29; swm-1 hermaphrodites (Figure 3A, Figure S4).
Alternatively, production of TRY-5 would be advantageous for males if it is a more efficient activator than that of hermaphrodites. While our fertility assays revealed no difference between fertility of wild-type and try-5 males, those assays were performed under highly permissive conditions: young adult animals were provided with many opportunities for mating to occur under conditions of unlimited food resources. TRY-5 might be important to increase reproductive fitness in less-than-ideal conditions. For example, activation by TRY-5 might occur more rapidly than that mediated by the hermaphrodite activator. If so, its transfer would decrease the chance that transferred sperm would be lost before they have the opportunity to migrate away from the vulva.
In summary, our work has identified a serine protease in C. elegans male seminal fluid that regulates the timing of sperm activation to promote male fertility. TRY-5 is transferred along with sperm during mating to couple sperm motility to entry into the hermaphrodite reproductive tract. While TRY-5 appears to be necessary for males to signal activation, hermaphrodites contain their own activator. Interestingly, these redundant pathways are competent to activate sperm from either sex, providing insight into the strategies used by C. elegans to adopt a male-hermaphrodite mode of reproduction. Further dissection of these signaling pathways will require identifying targets of TRY-5 and determining the nature of the hermaphrodite activator.
Materials and Methods
C. elegans genetics
C. elegans strains were grown as described by Brenner [47] at 20°C, except where otherwise noted. All strains were derived from the wild-type isolate Bristol N2. To ensure a ready supply of males, a strain harboring the mutation him-5(e1490) [48] was used as the wild type and him-5(e1490) was present in all other strains unless explicitly noted. The try-5 alleles jn2 and jn13 were isolated as suppressors of swm-1(me86) and jn21 was isolated as a suppressor of swm-1(me66) (G.M.S., unpublished results). Ethyl methanesulfonate (EMS) mutagenesis was performed as in [49]. try-5(tm3813) was a gift of S. Mitani (National Bioresource Project, Japan). Other alleles (described in Wood [49] unless otherwise noted) were: spe-8(hc40, hc53) I, fer-1(hc1ts) I, ttTi5605 II [28], unc-52(e444) II, dpy-18(e364) III, spe-6(hc163) III [18], unc-119(ed3, ed9) III [50], spe-27(it110) IV [15], spe-29(it127) IV [17], dpy-20(e1282) IV, mIs11[myo-2::GFP, pes-10::GFP, gut::GFP] IV, dpy-4(e1166) IV, unc-31(e169) IV, swm-1(me66, me86, me87) V [20], unc-76(e911) V and nT1[unc-?(n754) let-? qIs50 ](IV, V).
Strains containing mutations in both a spe-8 group gene and try-5 were maintained as heterozygotes using the balancer nT1. Homozygous spe-8 group; try-5 males were generated by transactivation crosses of homozygous self-sterile hermaphrodites to swm-1 mutant males, which are competent for transferring seminal fluid but rarely transfer sperm [20]. For example, for the spe-27 dpy-20/nT1; try-5 him-5/nT1 strain, homozygous spe-27 dpy-20; try-5 him-5 hermaphrodites were selected and crossed to either swm-1(me87) him-5 or mIs11; swm-1(me87) him-5 males to induce production of self progeny, which can be recognized as being phenotypically Dumpy.
To screen C. elegans proteases for a function in sperm activation, RNAi against individual protease genes was performed on swm-1 him-5 worm strains by feeding on agar plates essentially as described by [51]. Bacteria containing inducible RNAi clones (described in [52], [53]) were obtained from Source BioScience. Genes tested by RNAi were try-1, try-2, try-3, try-5, try-6, try-7, try-8, F25E5.3, F25E5.4, F25E5.7, and F48E3.4. For each gene, swm-1(me66) him-5 and swm-1(me86) him-5 eggs were collected on RNAi plates and allowed to grow to the L4 stage; L4 males were then transferred to a fresh RNAi plate and scored either 24 hr or 48 hr later for sperm activation.
Microscopy
Sperm activation was assayed in virgin males collected as L4 larvae and incubated at 20°C for 48 hr, unless otherwise indicated. To examine individual sperm cells, males were dissected in sperm medium (SM) (5 mM HEPES sodium salt pH 7.4, 50 mM NaCl, 25 mM KCl, 5 mM CaCl2, 1 mM MgSO4) supplemented with 10 mM dextrose [10]. Samples were observed using differential interference contrast (DIC) microscopy and sperm were scored based on cell shape as either non-activated, if spherical, or activated, based on the presence of a pseudopod. Samples were observed using an AxioImager M1 equipped with an AxioCam MRm (Zeiss). Confocal imaging was performed using a TCS SP2 (Leica). Images were processed using ImageJ [54] and Photoshop (Adobe Systems).
Fertility assays
Hermaphrodite self fertility was measured by picking individual hermaphrodites, transferring them to fresh plates every 1–2 days until no more eggs were laid, and counting the total progeny after worms reached the L4 stage. Cases in which hermaphrodites failed to lay oocytes or died less than four days after adulthood were excluded from analysis.
Male fertility was measured in 1∶1 crosses to spe-8(hc40); dpy-4 hermaphrodites. L4 stage animals were placed together for 48 hr; hermaphrodites were then transferred to fresh plates without males and transferred again every 1–2 days until no more eggs were laid. All cross progeny, identifiable by their non-Dumpy phenotype, were counted after worms reached the L4 stage. Use of the spe-8 mutation in recipient hermaphrodites allows for detection of mating even in cases where functional sperm are not transferred, since transfer of seminal fluid leads to production of self progeny [14], [20]. Cases in which mating was not confirmed or the hermaphrodite died less than three days after adulthood were excluded from analysis.
For all fertility assays, wild-type broods were measured in parallel to those of the strain being assayed to control for variations in temperature, media quality and other factors that can affect progeny production or mating efficiency.
Sperm and seminal fluid transfer assays
To assay sperm transfer and migration, males were labeled with 1 µg/mL MitoTracker CMXRos (Invitrogen) as described by Chen et al. [27] and observed as described previously [20].
Seminal fluid transfer (transactivation, [12]) was assayed using males harboring the fer-1(hc1ts) mutation, which results in non-functional sperm at the restrictive temperature of 25°C [25]. L4 males were crossed in a 4∶1 ratio to L4 spe-8(hc53); dpy-4 hermaphrodites for 48 hr at 25°C. The number of self progeny (Dumpy offspring) produced during the mating period was determined after three additional days. Any crosses resulting in cross progeny (non-Dumpy offspring) were excluded from analysis. All other crosses with recipient worms surviving to the end of the mating period were included, because no marker for successful mating is available for this assay. To assess mating frequency in different fer-1 mutant strains, males were labeled with MitoTracker and incubated with hermaphrodites in 1∶1 crosses. Hermaphrodites were then examined after 5 hr for the presence of labeled sperm. This assay likely underestimates the total mating frequency in transactivation assays, since 1) fer-1 sperm can not migrate and are only retained within hermaphrodites for a short time period, and 2) a higher ratio of 4 males:1 hermaphrodite was used for transactivation assays.
Assays of in vitro sperm activation
Activation assays were performed essentially as in [12]. Adult virgin males were dissected to release sperm in a drop of SM on a glass slide; a chamber was formed over the cells using a coverslip supported by a thin layer of Vaseline; additional SM either with activator (200 µg/mL Pronase or 60 mM TEA) or without it (control) was wicked through this chamber; and the coverslip was completely sealed with Vaseline. An image was obtained immediately upon wicking through activator and subsequent images were obtained every 5 min for at least 25 min. Activation was scored at each time point based on cell shape. To obtain time-lapse videos, activation assays were performed as described except that images were obtained once per minute.
Observation of TRY-5::GFP transfer
For each trial, one to two 24 hr post-L4 Ptry-5::TRY-5::GFP; try-5(tm3813) him-5 males were placed at the center of a circle of ten unc-52, unc-31 or unc-76 him-5 virgin adult hermaphrodites. Males were observed for 10 min under transmitted light using a Leica MZ16FL microscope. Prior to or shortly after spicule insertion occurred, the light source was switched to epifluorescence and images were collected at maximum speed (an exposure time of approximately 300 msec) using an AxioCam MRm (Zeiss) until spicules were removed. If copulation was not attempted within 10 min, males were removed and replaced with fresh males.
Molecular biology
Standard molecular biology protocols were used [55]. RNA was extracted from mixed-stage him-5 worms using TRIzol (Invitrogen). Reactions for 5′ - and 3′-RACE (rapid amplification of cDNA ends) were performed using GeneRacer (Invitrogen). The MultiSite Gateway Three-fragment Vector Construction Kit (Invitrogen) with pCFJ150 as the destination vector [28] was used to generate MosSCI donor constructs (Tables S1 and S2). Plasmid pCM1.35 was a gift from G. Seydoux [56]. For TRY-5::GFP, fusion PCR was performed as in [57]. Details of Gateway plasmid construction are listed in Table S1 and Table S2. To generate pJRS17, the 279 bp KpnI-XhoI fragment from pPD95.85 was ligated into the 4855 bp KpnI-XhoI fragment from pJRS11, thereby replacing the Ser65Cys variation present in GFP derived from pPD95.75 with the Ser65Thr variation from pPD95.85.
Transgenic strains
To generate transgenic strains harboring extrachromosomal arrays, constructs were injected [58] into the strain unc-119; swm-1(me86) try-5(jn2) him-5 and transgenic lines were selected based on rescue of the Unc-119 phenotype [28], [50]. Single-copy insertion (MosSCI) strains were generated by the direct insertion technique into the Mos1 insertion site ttTi5605 as described by Frokjaer-Jensen [28]. Targeting constructs were coinjected with Pglh-2::transposase as the source of Mos transposase and coinjection markers labeling pharyngeal muscle (Pmyo-2::mCherry), body wall muscle (Pmyo-3::mCherry), and neurons (Prab-3::mCherry) [28].
Supporting Information
Zdroje
1. PitnickSWolfnerMFSuarezS 2009 Ejaculate-female and sperm-female interactions. BirkheadTRHoskenDJPitnickS Sperm Biology: An Evolutionary Perspective Amsterdam; London Elsevier/Academic Press 247 304
2. PoianiA 2006 Complexity of seminal fluid: a review. Behav Ecol Sociobiol 60 289 310
3. YoshidaMKawanoNYoshidaK 2008 Control of sperm motility and fertility: diverse factors and common mechanisms. Cell Mol Life Sci 65 3446 3457
4. AvilaFWSirotLKLaFlammeBARubinsteinCDWolfnerMF 2011 Insect seminal fluid proteins: identification and function. Annu Rev Entomol 56 21 40
5. WolfnerMF 2009 Battle and Ballet: Molecular Interactions between the Sexes in Drosophila. J Hered 100 399 410
6. WardSCarrelJS 1979 Fertilization and sperm competition in the nematode Caenorhabditis elegans. Dev Biol 73 304 321
7. WardS 1977 The use of nematode behavioral mutants for analysis of neural function and development. CowanWMFerrendelliJA Neurosciences Research Symposium II: Approaches to the cell biology of neurons Bethesda, MD Society for Neurosciences 1 61
8. JustineJ-L 2002 Male and female gametes and fertilisation. LeeDL The biology of nematodes London Taylor & Francis 162 244
9. SmithHE 2006 Sperm motility and MSP. In: WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.68.1, http://www.wormbook.org
10. NelsonGAWardS 1980 Vesicle fusion, pseudopod extension and amoeboid motility are induced in nematode spermatids by the ionophore monensin. Cell 19 457 464
11. WardSHoganENelsonGA 1983 The initiation of spermiogenesis in the nematode Caenorhabditis elegans. Dev Biol 98 70 79
12. ShakesDCWardS 1989 Initiation of spermiogenesis in C. elegans: a pharmacological and genetic analysis. Dev Biol 134 189 200
13. MachacaKDeFeliceLJL'HernaultSW 1996 A novel chloride channel localizes to Caenorhabditis elegans spermatids and chloride channel blockers induce spermatid differentiation. Dev Biol 176 1 16
14. L'HernaultSWShakesDCWardS 1988 Developmental genetics of chromosome I spermatogenesis-defective mutants in the nematode Caenorhabditis elegans. Genetics 120 435 452
15. MinnitiANSadlerCWardS 1996 Genetic and molecular analysis of spe-27, a gene required for spermiogenesis in Caenorhabditis elegans hermaphrodites. Genetics 143 213 223
16. NanceJMinnitiANSadlerCWardS 1999 spe-12 encodes a sperm cell surface protein that promotes spermiogenesis in Caenorhabditis elegans. Genetics 152 209 220
17. NanceJDavisEBWardS 2000 spe-29 encodes a small predicted membrane protein required for the initiation of sperm activation in Caenorhabditis elegans. Genetics 156 1623 1633
18. MuhlradPJWardS 2002 Spermiogenesis initiation in Caenorhabditis elegans involves a casein kinase 1 encoded by the spe-6 gene. Genetics 161 143 155
19. GeldzilerBChatterjeeISingsonA 2005 The genetic and molecular analysis of spe-19, a gene required for sperm activation in Caenorhabditis elegans. Dev Biol 283 424 436
20. StanfieldGMVilleneuveAM 2006 Regulation of sperm activation by SWM-1 is required for reproductive success of C. elegans males. Curr Biol 16 252 263
21. Marchler-BauerAAndersonJBChitsazFDerbyshireMKDeWeese-ScottC 2009 CDD: specific functional annotation with the Conserved Domain Database. Nucleic Acids Res 37 D205 210
22. Di CeraE 2009 Serine proteases. IUBMB Life 61 510 515
23. PeronaJJCraikCS 1995 Structural basis of substrate specificity in the serine proteases. Protein Sci 4 337 360
24. VarkeyJPJansmaPLMinnitiANWardS 1993 The Caenorhabditis elegans spe-6 gene is required for major sperm protein assembly and shows second site non-complementation with an unlinked deficiency. Genetics 133 79 86
25. WardSMiwaJ 1978 Characterization of temperature-sensitive, fertilization-defective mutants of the nematode Caenorhabditis elegans. Genetics 88 285 303
26. L'HernaultSW 2006 Spermatogenesis. In: WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.85.1, http://www.wormbook.org
27. ChenFHershBMConradtBZhouZRiemerD 2000 Translocation of C. elegans CED-4 to nuclear membranes during programmed cell death. Science 287 1485 1489
28. Frokjaer-JensenCDavisMWHopkinsCENewmanBJThummelJM 2008 Single-copy insertion of transgenes in Caenorhabditis elegans. Nat Genet 40 1375 1383
29. LintsRHallDH 2009 Male reproductive system, somatic gonad. WormAtlas doi:10.3908/wormatlas.2.15. Accessed 1 May 2011
30. KimbleJHirshD 1979 The postembryonic cell lineages of the hermaphrodite and male gonads in Caenorhabditis elegans. Dev Biol 70 396 417
31. BarrMMGarciaLR 2006 Male mating behavior. In: WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.78.1, http://www.wormbook.org
32. SchindelmanGWhittakerAJThumJYGharibSSternbergPW 2006 Initiation of male sperm-transfer behavior in Caenorhabditis elegans requires input from the ventral nerve cord. BMC Biol 4 26
33. BaldiCChoSEllisRE 2009 Mutations in two independent pathways are sufficient to create hermaphroditic nematodes. Science 326 1002 1005
34. FoorWEMcMahonJT 1973 Role of the glandular vas deferens in the development of Ascaris spermatozoa. J Parasitol 59 753 758
35. BurghardtRCFoorWE 1978 Membrane fusion during spermiogenesis in Ascaris. J Ultrastruct Res 62 190 202
36. PampalakisGSotiropoulouG 2007 Tissue kallikrein proteolytic cascade pathways in normal physiology and cancer. Biochim Biophys Acta 1776 22 31
37. OvaerePLippensSVandenabeelePDeclercqW 2009 The emerging roles of serine protease cascades in the epidermis. Trends Biochem Sci 34 453 463
38. GillottC 2003 Male accessory gland secretions: Modulators of female reproductive physiology and behavior. Annu Rev Entomol 48 163 184
39. WolfnerMF 2007 “S.P.E.R.M.” (seminal proteins (are) essential reproductive modulators): the view from Drosophila. Soc Reprod Fertil Suppl 65 183 199
40. HodgkinJDoniachT 1997 Natural variation and copulatory plug formation in Caenorhabditis elegans. Genetics 146 149 164
41. PalopoliMFRockmanMVTinMaungARamsayCCurwenS 2008 Molecular basis of the copulatory plug polymorphism in Caenorhabditis elegans. Nature 454 1019 1022
42. GattiJLCastellaSDacheuxFEcroydHMetayerS 2004 Post-testicular sperm environment and fertility. Anim Reprod Sci 82-83 321 339
43. CornwallGAvan HorstenHH 2007 Sperm maturation in the epididymis: Role of segment-specific microenvironments. CarrellDT The genetics of male infertility Totowa, N.J. Humana Press 211 231
44. KiontkeKGavinNPRaynesYRoehrigCPianoF 2004 Caenorhabditis phylogeny predicts convergence of hermaphroditism and extensive intron loss. Proc Natl Acad Sci U S A 101 9003 9008
45. ChoSJinSWCohenAEllisRE 2004 A phylogeny of Caenorhabditis reveals frequent loss of introns during nematode evolution. Genome Res 14 1207 1220
46. LaMunyonCWWardS 1995 Sperm precedence in a hermaphroditic nematode (Caenorhabditis elegans) is due to competitive superiority of male sperm. Experientia 51 817 823
47. BrennerS 1974 The genetics of Caenorhabditis elegans. Genetics 77 71 94
48. HodgkinJHorvitzHRBrennerS 1979 Nondisjunction mutants of the nematode Caenorhabditis elegans. Genetics 91 67 94
49. Wood and the Community of C. elegans Researchers 1988 The nematode Caenorhabditis elegans Cold Spring Harbor, N.Y. Cold Spring Harbor Laboratory
50. MaduroMPilgrimD 1995 Identification and cloning of unc-119, a gene expressed in the Caenorhabditis elegans nervous system. Genetics 141 977 988
51. AhringerJ 2006 Reverse genetics. In: WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.47.1, http://www.wormbook.org
52. FraserAGKamathRSZipperlenPMartinez-CamposMSohrmannM 2000 Functional genomic analysis of C. elegans chromosome I by systematic RNA interference. Nature 408 325 330
53. KamathRSFraserAGDongYPoulinGDurbinR 2003 Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421 231 237
54. RasbandWS 1997–2009 ImageJ Bethesda, Maryland, US U.S. National Institutes of Health
55. SambrookJFritschEFManiatisT 1989 Molecular Cloning: A Laboratory Manual Cold Spring Harbor, New York Cold Spring Harbor Laboratory Press
56. MerrittCRasolosonDKoDSeydouxG 2008 3′ UTRs are the primary regulators of gene expression in the C. elegans germline. Curr Biol 18 1476 1482
57. HobertO 2002 PCR fusion-based approach to create reporter gene constructs for expression analysis in transgenic C. elegans. Biotechniques 32 728 730
58. MelloCCKramerJMStinchcombDAmbrosV 1991 Efficient gene transfer in C.elegans: extrachromosomal maintenance and integration of transforming sequences. EMBO J 10 3959 3970
59. WormBase http://www.wormbase.org, release WS224. Accessed 6 June 2011
60. BendtsenJDNielsenHvon HeijneGBrunakS 2004 Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340 783 795
Štítky
Genetika Reprodukční medicína
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 11- Akutní intermitentní porfyrie
- Růst a vývoj dětí narozených pomocí IVF
- Farmakogenetické testování pomáhá předcházet nežádoucím efektům léčiv
- Pilotní studie: stres a úzkost v průběhu IVF cyklu
- IVF a rakovina prsu – zvyšují hormony riziko vzniku rakoviny?
-
Všechny články tohoto čísla
- De Novo Origins of Human Genes
- Duplication Hotspots Are Associated with Late-Replicating Regions of the Genome
- De Novo Origin of Human Protein-Coding Genes
- Cyclin D/CDK4 and Cyclin E/CDK2 Induce Distinct Cell Cycle Re-Entry Programs in Differentiated Muscle Cells
- Short Day–Mediated Cessation of Growth Requires the Downregulation of AINTEGUMENTALIKE1 Transcription Factor in Hybrid Aspen
- Physiological IRE-1-XBP-1 and PEK-1 Signaling in Larval Development and Immunity
- Role of Pirh2 in Mediating the Regulation of p53 and c-Myc
- Signatures of Environmental Genetic Adaptation Pinpoint Pathogens as the Main Selective Pressure through Human Evolution
- FOXO Regulates Organ-Specific Phenotypic Plasticity In
- Heritable Epigenetic Variation among Maize Inbreds
- Foxn1 Regulates Lineage Progression in Cortical and Medullary Thymic Epithelial Cells But Is Dispensable for Medullary Sublineage Divergence
- Attenuation of the Sensing Capabilities of PhoQ in Transition to Obligate Insect–Bacterial Association
- A Novel Protein LZTFL1 Regulates Ciliary Trafficking of the BBSome and Smoothened
- Activation of Bmp2-Smad1 Signal and Its Regulation by Coordinated Alteration of H3K27 Trimethylation in -Induced Senescence
- Histone H3K56 Acetylation, CAF1, and Rtt106 Coordinate Nucleosome Assembly and Stability of Advancing Replication Forks
- The SUN Protein Mps3 Is Required for Spindle Pole Body Insertion into the Nuclear Membrane and Nuclear Envelope Homeostasis
- Evidence-Based Annotation of Gene Function in MR-1 Using Genome-Wide Fitness Profiling across 121 Conditions
- Effect of Host Species on the Distribution of Mutational Fitness Effects for an RNA Virus
- Pch2 Acts through Xrs2 and Tel1/ATM to Modulate Interhomolog Bias and Checkpoint Function during Meiosis
- SOX9 Governs Differentiation Stage-Specific Gene Expression in Growth Plate Chondrocytes via Direct Concomitant Transactivation and Repression
- from the Aphid : A Missing Link from Facultative to Obligate Insect Endosymbiont
- Recessive Antimorphic Alleles Overcome Functionally Redundant Loci to Reveal Function in Flowers and Meristems
- Over-Expression of DSCAM and COL6A2 Cooperatively Generates Congenital Heart Defects
- Consequences of Eukaryotic Enhancer Architecture for Gene Expression Dynamics, Development, and Fitness
- Distinct Genetic Architectures for Male and Female Inflorescence Traits of Maize
- Capture of MicroRNA–Bound mRNAs Identifies the Tumor Suppressor miR-34a as a Regulator of Growth Factor Signaling
- For Male , Sperm Activation Is a “Just-in-Time” Event
- PcG Complexes Set the Stage for Epigenetic Inheritance of Gene Silencing in Early S Phase before Replication
- The Gene Contains Hotspots for L1 Endonuclease-Dependent Insertion
- Relative Burden of Large CNVs on a Range of Neurodevelopmental Phenotypes
- Multiple Means to the Same End: The Genetic Basis of Acquired Stress Resistance in Yeast
- Genome-Wide Crossover Distribution in Meiosis Reveals Sex-Specific Patterns along Chromosomes
- TRY-5 Is a Sperm-Activating Protease in Seminal Fluid
- Homologs of Retinoblastoma-Associated Protein 46/48 Associate with a Histone Deacetylase to Act Redundantly in Chromatin Silencing
- Genetic Interaction Maps in Reveal Functional Crosstalk among Cell Envelope Biogenesis Pathways
- The ERI-6/7 Helicase Acts at the First Stage of an siRNA Amplification Pathway That Targets Recent Gene Duplications
- PBX1 Genomic Pioneer Function Drives ERα Signaling Underlying Progression in Breast Cancer
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Evidence-Based Annotation of Gene Function in MR-1 Using Genome-Wide Fitness Profiling across 121 Conditions
- De Novo Origins of Human Genes
- Capture of MicroRNA–Bound mRNAs Identifies the Tumor Suppressor miR-34a as a Regulator of Growth Factor Signaling
- TRY-5 Is a Sperm-Activating Protease in Seminal Fluid
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



