-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Concocting Cholinergy
article has not abstract
Published in the journal: . PLoS Genet 10(4): e32767. doi:10.1371/journal.pgen.1004313
Category: Perspective
doi: https://doi.org/10.1371/journal.pgen.1004313Summary
article has not abstract
The neuronal diversity in our brains is staggering. Understandably, uncovering the molecular rules that govern it is a very difficult pursuit. Its starting point should most certainly be a catalogue raisonné of this diversity, ordered perhaps according to the relatively few neurotransmitters employed in the nervous system. Historically, this has been achieved in descriptive studies; first using histochemical approaches and later antibody staining for the neurotransmitters, their synthetic enzymes, and the transporters that load them into secretory vesicles as well as for their receptors on target cells. How the expression of such neurotransmitter machinery is coordinated to endow a neuron with a “neurotransmitter identity” (NtI) is a big piece of the molecular puzzle of neuronal diversity. Cho et al. provide a glimpse into this question by uncovering how an acetylcholine NtI gene transcription program is established in two very different vertebrate neuronal populations [1].
Perhaps the simplest way to achieve this might be through the ancestral “regulon” strategy employed in bacteria to coordinate biosynthetic pathway enzyme expression [2]. Neurons of a particular NtI would thus express a master regulatory transcription factor that is able to turn on the expression of all genes encoding a particular NtI neurotransmitter identity through its ability to bind a common cis-regulatory element present in all loci. A more complex strategy might involve neuron-subpopulation–specific NtI transcription factors in which different neuron classes use different transcription factors or their combinations to turn on the expression of the same NtI genes. Some recent experiments in the worm Caenorhabditis elegans have confirmed the existence of NtI master regulatory transcription factors [3]–[5], raising the question of whether a similar strategy is used in vertebrates. Cho et al. show in vertebrates that such regulatory factors exist but work in a combinatorial manner. The study focuses on two cholinergic neuronal populations: spinal motor neurons (MNs) and forebrain cholinergic neurons (FCNs) [6]. MNs are an ideal starting point for this analysis as much is known about how their genesis is controlled at the transcriptional level [7], [8]. One of the key inducers of MN fate, including their cholinergic NtI, is a hexameric DNA-binding complex containing the LIM-homeodomain transcription factors Lhx3 and Isl1 joined together by the LIM-interactor protein NLI. Such “Isl1-Lhx3 hexamers” are sufficient to induce ectopic MNs exhibiting cholinergic features in the spinal cord and from undifferentiated precursors [9].
The authors first considered the DNA sequences surrounding “cholinergic genes” such as those encoding choline transporters and choline synthesis enzymes. Chromatin immunoprecipitation sequence analysis revealed that the Isl1-Lhx3 hexamer binds conserved regions of these genes, both in embryonic stem (ES) cells and spinal cords. Overexpression of the Isl1-Lhx3 hexamer results in induction of expression of cholinergic genes in the spinal cord, and loss of Isl1 function leads to their down-regulation. Next, using a transcription reporter, the authors showed that the conserved sequences in the cholinergic genes are sufficient for expression in the presence of the Isl1-Lhx3 hexamer and can be further narrowed down to a core response element (HxRE). When multimerized, the HxRE can selectively drive green fluorescent protein (GFP) expression in motor neurons. Next, a developmental time course analysis revealed that FCNs originating from the medial ganglionic eminence express Isl1, NLI, and a distinct LIM-homeodomain (LIM-HD) protein, Lhx8. Notably, deletion of Isl1 from this region results in a depletion of FCNs, reminiscent of the loss of MNs in the spinal cord [10]. Biochemical manipulations next showed that Lhx8 forms a complex with NLI and Isl1 that is analogous to the Isl1-Lhx3 hexamer and that the Isl1-Lhx8 complex binds a specific DNA motif remarkably similar to that recognized by the Isl1-Lhx3 hexamer. This motif is also present in many cholinergic enhancers, and its multimerization is able to drive GFP reporter expression selectively within the embryonic forebrain.
Are these hexamers interchangeable? Overexpression experiments argue that only Isl1-Lhx8 hexamer is able to activate cholinergic features in the forebrain, while Isl1-Lhx3 can only induce motor-neuron–specific genes such as Hb9 and Isl2 in the spinal cord, arguing that the cellular context is critical for the function of the Isl1-LhxX hexamers. Extending this conclusion, the production of the Isl1-Lhx8 hexamer in an ES cell line can induce the expression of cholinergic genes but not motor neuron genes. All these data point to the conclusion that Isl1 controls cholinergic NtI in two different regions of the central nervous system through neuron-subtype–specific transcription factor complexes (see Figure 1).
Fig. 1. LIM-homeodomain proteins Lhx3 and Lhx8 induce the cholinergic neurotransmitter identity in spinal motor neurons and forebrain cholinergic neurons through Isl1 and NLI hexamer complex transcriptional activity. 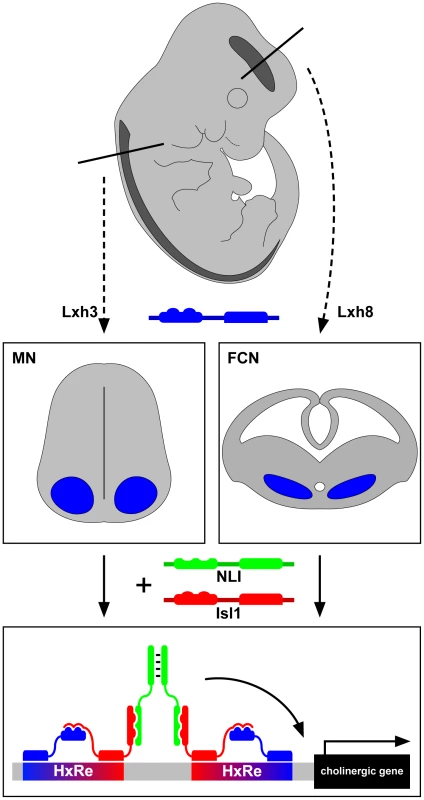
What does it all mean? Clearly, Isl1 is a central player in vertebrate cholinergic NtI whose molecular logic is more complex than that of the simple bacterial regulon. Isl1 works in concert with at least two other neuron-subtype–specific LIM-homeodomain proteins, Lhx1 and Lhx8, which apparently target common enhancer regions near “cholinergic genes.” This is also different from the C. elegans strategy in which the UNC-3 transcription factor is sufficient to coregulate all “cholinergic genes” [4]. The most pressing question stemming from this study is the identity of the sequences bound by Isl1-Lhx8 and Isl1-Lhx3. Some preliminary and unpublished observations suggest that these are distinct. One corollary would be that there are other cholinergic neuron-subtype–specific Isl1-Lhx complexes and that each of these has a binding sequence near cholinergic genes. This would make for quite a baroque model of vertebrate cholinergic NtI. Another possibility could be that there are neuron-type–specific Isl1-Lhx hexamers but that these bind to the same DNA sequences. They would only be able to turn on cholinergic genes through the interaction of the Isl1 partner (Lhx, Lhx8, etc) with another cell-type–specific transcription factor binding to nearby sequences.
Arguably, NtI must be tightly regulated so that neurons do not express inappropriate neurotransmitters and adhere (mostly) to Dale's principle that neurons use a single mode of transmission [11]. One scheme could involve active suppression of “competing” NtIs, in which, for example, Isl1-Lhx8 could bind promoters of glutamatergic and GABAergic genes and actively shut them down. This does not appear to be a strategy in C. elegans, but vertebrate NtI could be more tortuous [12]. In the broader context, this study provides some attractive models in which to examine the intersection between NtI and other aspects of neuronal identity, such as dendrite morphology or soma location. To what extent are all these linked? Could they really be thought of as separate modules or would a selection of a particular NtI favour some morphological features? This could be regulated either directly at a transcriptional level, in which Isl1-Lhx8, for example, might influence the expression of some FCN “structural” genes, or through homeostatic mechanisms in which the use of a particular neurotransmitter has global consequences on the electrical properties of a neuron. Finally, another important general question is that of the maintenance of neuronal identity, including NtI. Could the same transcriptional complexes that are turning on cholinergy also be required for maintaining it? Could Alzheimer disease and amyotrophic lateral sclerosis, two human pathologies affecting cholinergic neurons, involve a loss of Isl1-Lhx3 and Isl1-Lhx8 function? Some answers to this question will certainly come from a deeper understanding of molecular strategies controlling the intersection of NtI with other aspects of neuronal identity.
Zdroje
1. ChoH-H, CargninF, KimY, LeeB, KwonR-J, et al. (2014) Isl1 directly controls a cholinergic neuronal identity in the developing forebrain and spinal cord by forming cell type-specific complexes. PLoS Genet 10: e1004280 doi:10.1371/journal.pgen.1004280
2. EpsteinW, BeckwithJR (1968) Regulation of Gene Expression. Ann Rev Biochemistry 37 : 411–436.
3. DoitsidouM, FlamesN, TopalidouI, AbeN, FeltonT, et al. (2013) A combinatorial regulatory signature controls terminal differentiation of the dopaminergic nervous system in C. elegans. Genes Dev 27 : 1391–1405.
4. KratsiosP, StolfiA, LevineM, HobertO (2011) Coordinated regulation of cholinergic motor neuron traits through a conserved terminal selector gene. Nat Neurosci 15 : 205–214.
5. Serrano-SaizE, PooleRJ, FeltonT, ZhangF, De La CruzED, et al. (2013) Modular control of glutamatergic neuronal identity in C. elegans by distinct homeodomain proteins. Cell 155 : 659–673.
6. WoolfNJ (1991) Cholinergic systems in mammalian brain and spinal cord. Prog Neurobiol 37 : 475–524.
7. AlaynickWA, JessellTM, PfaffSL (2011) SnapShot: spinal cord development. Cell 146 : 178–178.e1.
8. Dalla Torre di SanguinettoSA, DasenJS, ArberS (2008) Transcriptional mechanisms controlling motor neuron diversity and connectivity. Curr Opin Neurobiol 18 : 36–43.
9. ThalerJP, LeeS-K, JurataLW, GillGN, PfaffSL (2002) LIM factor Lhx3 contributes to the specification of motor neuron and interneuron identity through cell-type-specific protein-protein interactions. Cell 110 : 237–249.
10. PfaffSL, MendelsohnM, StewartCL, EdlundT, JessellTM (1996) Requirement for LIM homeobox gene Isl1 in motor neuron generation reveals a motor neuron-dependent step in interneuron differentiation. Cell 84 : 309–320.
11. BurnstockG (2004) Cotransmission. Curr Opin Pharmacol 4 : 47–52.
12. HobertO, CarreraI, StefanakisN (2010) The molecular and gene regulatory signature of a neuron. Trends Neurosci 33 : 435–445.
Štítky
Genetika Reprodukční medicína
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2014 Číslo 4- Akutní intermitentní porfyrie
- Farmakogenetické testování pomáhá předcházet nežádoucím efektům léčiv
- Hypogonadotropní hypogonadismus u žen a vliv na výsledky reprodukce po IVF
- Molekulární vyšetření pro stanovení prognózy pacientů s chronickou lymfocytární leukémií
- Prof. Petr Urbánek: Potřebujeme najít pacienty s nediagnostikovanou akutní intermitentní porfyrií
-
Všechny články tohoto čísla
- The Challenges of Mitochondrial Replacement
- Concocting Cholinergy
- Genome-Wide Diet-Gene Interaction Analyses for Risk of Colorectal Cancer
- Statistical Power to Detect Genetic (Co)Variance of Complex Traits Using SNP Data in Unrelated Samples
- Mouse Pulmonary Adenoma Susceptibility 1 Locus Is an Expression QTL Modulating -4A
- Transcription-Associated R-Loop Formation across the Human CGG-Repeat Region
- Epigenetic Regulation by Heritable RNA
- Protein Quantitative Trait Loci Identify Novel Candidates Modulating Cellular Response to Chemotherapy
- Genome-Wide Profiling of Yeast DNA:RNA Hybrid Prone Sites with DRIP-Chip
- The Mechanism of Gene Targeting in Human Somatic Cells
- A LINE-1 Insertion in DLX6 Is Responsible for Cleft Palate and Mandibular Abnormalities in a Canine Model of Pierre Robin Sequence
- Interaction between Two Timing MicroRNAs Controls Trichome Distribution in
- DNA Glycosylases Involved in Base Excision Repair May Be Associated with Cancer Risk in and Mutation Carriers
- The Myc-Mondo/Mad Complexes Integrate Diverse Longevity Signals
- Evolutionarily Diverged Regulation of X-chromosomal Genes as a Primal Event in Mouse Reproductive Isolation
- Mutations in Conserved Residues of the microRNA Argonaute ALG-1 Identify Separable Functions in ALG-1 miRISC Loading and Target Repression
- Genetic Predisposition to In Situ and Invasive Lobular Carcinoma of the Breast
- Isl1 Directly Controls a Cholinergic Neuronal Identity in the Developing Forebrain and Spinal Cord by Forming Cell Type-Specific Complexes
- A Synthetic Community Approach Reveals Plant Genotypes Affecting the Phyllosphere Microbiota
- The Sequence-Specific Transcription Factor c-Jun Targets Cockayne Syndrome Protein B to Regulate Transcription and Chromatin Structure
- Determining the Control Circuitry of Redox Metabolism at the Genome-Scale
- DNA Repair Pathway Selection Caused by Defects in , , and Telomere Addition Generates Specific Chromosomal Rearrangement Signatures
- Methylome Diversification through Changes in DNA Methyltransferase Sequence Specificity
- Folliculin Regulates Ampk-Dependent Autophagy and Metabolic Stress Survival
- Fine Mapping of Dominant -Linked Incompatibility Alleles in Hybrids
- Unexpected Role of the Steroid-Deficiency Protein Ecdysoneless in Pre-mRNA Splicing
- Three Groups of Transposable Elements with Contrasting Copy Number Dynamics and Host Responses in the Maize ( ssp. ) Genome
- Sox5 Functions as a Fate Switch in Medaka Pigment Cell Development
- Synergistic Interactions between the Molecular and Neuronal Circadian Networks Drive Robust Behavioral Circadian Rhythms in
- Chromatin Landscapes of Retroviral and Transposon Integration Profiles
- Widespread Use of Non-productive Alternative Splice Sites in
- Ras GTPase-Like Protein MglA, a Controller of Bacterial Social-Motility in Myxobacteria, Has Evolved to Control Bacterial Predation by
- Cell Type-Specific Functions of Genes Revealed by Novel Adipocyte and Hepatocyte Circadian Clock Models
- Phenotype Ontologies and Cross-Species Analysis for Translational Research
- Embryogenesis Scales Uniformly across Temperature in Developmentally Diverse Species
- In Pursuit of the Gene: An Interview with James Schwartz
- Molecular Mechanisms of Hypoxic Responses via Unique Roles of Ras1, Cdc24 and Ptp3 in a Human Fungal Pathogen
- Analysis of the Genome and Transcriptome of var. Reveals Complex RNA Expression and Microevolution Leading to Virulence Attenuation
- Genotypic and Functional Impact of HIV-1 Adaptation to Its Host Population during the North American Epidemic
- RNA Editome in Rhesus Macaque Shaped by Purifying Selection
- Proper Actin Ring Formation and Septum Constriction Requires Coordinated Regulation of SIN and MOR Pathways through the Germinal Centre Kinase MST-1
- Interplay of the Serine/Threonine-Kinase StkP and the Paralogs DivIVA and GpsB in Pneumococcal Cell Elongation and Division
- A Quality Control Mechanism Coordinates Meiotic Prophase Events to Promote Crossover Assurance
- CNNM2 Mutations Cause Impaired Brain Development and Seizures in Patients with Hypomagnesemia
- The RNA-Binding Protein QKI Suppresses Cancer-Associated Aberrant Splicing
- Uncoupling Transcription from Covalent Histone Modification
- Rad51–Rad52 Mediated Maintenance of Centromeric Chromatin in
- FRA2A Is a CGG Repeat Expansion Associated with Silencing of
- A General Approach for Haplotype Phasing across the Full Spectrum of Relatedness
- A Novel Highly Divergent Protein Family Identified from a Viviparous Insect by RNA-seq Analysis: A Potential Target for Tsetse Fly-Specific Abortifacients
- A Central Role for in Regulation of Islet Function in Man
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- The Sequence-Specific Transcription Factor c-Jun Targets Cockayne Syndrome Protein B to Regulate Transcription and Chromatin Structure
- The Mechanism of Gene Targeting in Human Somatic Cells
- Genetic Predisposition to In Situ and Invasive Lobular Carcinoma of the Breast
- Widespread Use of Non-productive Alternative Splice Sites in
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



