-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaFunctional Analysis of the Interdependence between DNA Uptake Sequence and Its Cognate ComP Receptor during Natural Transformation in Species
Natural transformation is the widespread biological process by which “competent” bacteria take up free DNA, incorporate it into their genomes, and become genetically altered or “transformed”. To curb often deleterious transformation by foreign DNA, several competent species preferentially take up their own DNA that contains specific DUS (DNA uptake sequence) watermarks. Our recent finding that ComP is the long sought DUS receptor in Neisseria species paves the way for the functional analysis of the DUS-ComP interdependence which is reported here. By abolishing/modulating ComP levels in Neisseria meningitidis, we show that the enhancement of transformation seen in the presence of DUS is entirely dependent on ComP, which also controls transformation in the absence of DUS. While peripheral bases in the DUS were found to be less important, inner bases are essential since single base mutations led to dramatically impaired interaction with ComP and transformation. Strikingly, naturally occurring DUS variants in the genomes of human Neisseria commensals differing from DUS by only one or two bases were found to be similarly impaired for transformation of N. meningitidis. By showing that ComPsub from the N. subflava commensal specifically binds its cognate DUS variant and mediates DUS-enhanced transformation when expressed in a comP mutant of N. meningitidis, we confirm that a similar mechanism is used by all Neisseria species to promote transformation by their own, or closely related DNA. Together, these findings shed new light on the molecular events involved in the earliest step in natural transformation, and reveal an elegant mechanism for modulating horizontal gene transfer between competent species sharing the same niche.
Published in the journal: . PLoS Genet 9(12): e32767. doi:10.1371/journal.pgen.1004014
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1004014Summary
Natural transformation is the widespread biological process by which “competent” bacteria take up free DNA, incorporate it into their genomes, and become genetically altered or “transformed”. To curb often deleterious transformation by foreign DNA, several competent species preferentially take up their own DNA that contains specific DUS (DNA uptake sequence) watermarks. Our recent finding that ComP is the long sought DUS receptor in Neisseria species paves the way for the functional analysis of the DUS-ComP interdependence which is reported here. By abolishing/modulating ComP levels in Neisseria meningitidis, we show that the enhancement of transformation seen in the presence of DUS is entirely dependent on ComP, which also controls transformation in the absence of DUS. While peripheral bases in the DUS were found to be less important, inner bases are essential since single base mutations led to dramatically impaired interaction with ComP and transformation. Strikingly, naturally occurring DUS variants in the genomes of human Neisseria commensals differing from DUS by only one or two bases were found to be similarly impaired for transformation of N. meningitidis. By showing that ComPsub from the N. subflava commensal specifically binds its cognate DUS variant and mediates DUS-enhanced transformation when expressed in a comP mutant of N. meningitidis, we confirm that a similar mechanism is used by all Neisseria species to promote transformation by their own, or closely related DNA. Together, these findings shed new light on the molecular events involved in the earliest step in natural transformation, and reveal an elegant mechanism for modulating horizontal gene transfer between competent species sharing the same niche.
Introduction
Natural transformation is a widespread biological property shared by dozens of bacterial species which plays a key role in evolution by generating genetic diversity through horizontal gene transfer [1]. In the human pathogen Neisseria meningitidis, natural transformation promotes gene reassortment on a massive scale with up to 200 genes frequently differing between isolates [2]. The meningococcus acquires DNA mainly from other meningococci but there is ample evidence for horizontal gene transfer from other bacteria sharing the same ecological niche, i.e. the human nasopharynx, such as other Neisseria species [3] or Haemophilus species [4]. This astonishing genome plasticity is a key virulence property of N. meningitidis, which contributes markedly to its success as a pathogen [5]. The same is true in other competent species where horizontal gene transfer has been implicated in the emergence of more virulent clones, and/or “superbugs” resistant to a wide variety of antibiotics.
Natural transformation, which has been studied for almost a century [6] and was key to the discovery that DNA carries genetic information [7], is thought to function as follows [8]. Free DNA is first bound by surface-exposed receptors that are likely to be components of type IV pili (Tfp) (or evolutionarily related competence pseudopili), such as the minor pilin ComP in Neisseria species [9]. Tfp are filamentous structures found in hundreds of species [10], which generate remarkable force upon retraction powered by the PilT ATPase [11]. Pilus/pseudopilus retraction promotes DNA translocation (or “uptake”) across formidable barriers, i.e. the outer membrane in Gram negative competent species [12], or the thick layer of peptidoglycan in Gram positive species [13]. Once in the periplasm/pseudoperiplasm, DNA interacts with a second widely conserved machinery that further transports it across the cytoplasmic membrane [8]. This machinery consists of another DNA receptor named ComE/ComEA [14], [15] which delivers the DNA to the permease ComEC [16] that transports it to the cytoplasm during a process powered by ATP hydrolysis [17]. Finally, bacteria become transformed upon stable, RecA-mediated incorporation of the new DNA into their genome. However, incoming DNA can also be used as a template for the repair of DNA damage (when donor is identical), or as a source of food (when donor is too different) [8].
Natural transformation by foreign DNA is a continuum between stable inheritance of a few useful new genes, and eventually loss of fitness and/or species structure, which explains why it is tightly controlled. In most competent species, transformation by foreign DNA is limited by rapid degradation by restriction enzymes and/or extensive sequence divergence that prevents RecA action [18]. However some competent species, including N. meningitidis, protect themselves by preferentially taking up their own DNA. It has been known for decades that uptake selectivity in N. meningitidis, and the closely related human pathogen N. gonorrhoeae, is dependent on short sequence motifs called DUS [19] that are 12 bp (ATGCCGTCTGAA) repeats representing 1% of the genome [20], [21]. In contrast, the identification of ComP as the DUS receptor is extremely recent [22]. We found that ComP, which was known to be crucial for natural transformation without affecting Tfp biogenesis [23], [24], (i) has a structure typical of type IV pilins, (ii) is required for efficient DNA binding by purified Tfp, (iii) is the only meningococcal pilin capable of binding DNA in vitro, and (iv) although it can bind any DNA has a marked preference for DUS [22]. Consistent with sequence conservation of DUS in N. meningitidis and N. gonorrhoeae, ComP was found to be virtually identical in these species, as well as in the human commensals N. lactamica and N. polysaccharea [22]. More distant ComP homologs were found in other Neisseria species and in many other members of the Neisseriaceae family, suggesting that ComP-mediated selective uptake of homotypic DNA is a widespread phenomenon [22]. Since canonical DUS are scarce in these latter species, many of which share the same ecological niche with N. meningitidis [25], it is tempting to speculate that co-evolution of DUS variants and cognate ComP receptors has occurred as a mechanism for modulating horizontal gene transfer between competent species living in the same environment.
The identification of ComP as the DUS receptor in Neisseria species paves the way for a detailed functional analysis of the DUS-ComP interdependence during natural transformation. Therefore, we have carried out and report here a series of experiments in which we have tested (i) the impact of ComP on transformation both in the presence and in the absence of DUS, (ii) the importance of DUS, and each of its bases, for transformation and efficient recognition by ComP, (iii) the ability of DUS variants naturally occuring in human Neisseria commensals to transform N. meningitidis, and (iv) the ability of a ComP homolog in one of these commensals (N. subflava) to bind its cognate DUS and mediate DUS-enhanced transformation.
Results
DUS enhancement of transformation in N. meningitidis 8013 is significant and ComP-dependent
Natural transformation has been extensively studied in multiple strains of N. meningitidis and N. gonorrhoeae. Competence has been shown to be dependent on the strain, type of DNA, and antibiotic resistance used for selection of transformants [18], [21], [26], [27]. In order to perform an in depth analysis of the functional interdependence between DUS and its ComP receptor, we designed a protocol for measuring competence in which all relevant parameters could be controlled. We focused on N. meningitidis clinical isolate 8013 because its genome sequence is available [2], and it is a model strain for studying Tfp biology [24], [28]. Rather than using genomic DNA for the transformation experiments, whose length and DUS composition cannot be controlled, we used a PCR fragment (1,934 bp) corresponding to a ΔpilN mutation in which the pilN gene has been replaced by an aphA3 cassette encoding resistance to kanamycin [29]. Critically, in this PCR fragment (Figure 1) (i) the aphA3 gene is flanked by genomic regions long enough for allelic exchange to occur efficiently and transformants to be selected [29], (ii) the GC content is similar to that of the genome [2], and (iii) no DUS with as many as two mismatches is found. Therefore, canonical or variant DUS can be added in 3′ of the transforming fragments simply by including them as overhangs (Figure 1) on the reverse primers used for PCR (Table 1).
Fig. 1. Schematic cartoon, drawn to scale, of transforming DNAs used in this study. 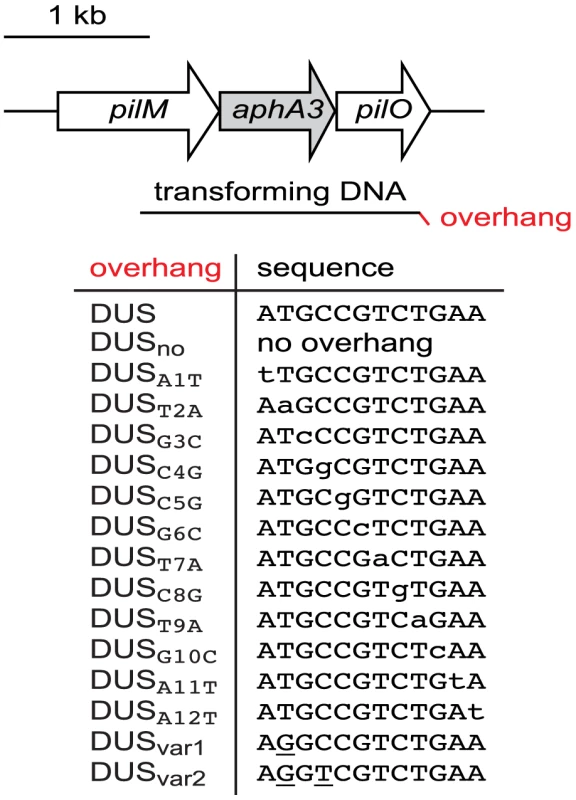
PCR fragments (1.9 kbp) corresponding to a ΔpilN mutation were used for transformations. In ΔpilN, an aphA3 cassette encoding resistance to kanamycin replaces the pilN gene and is flanked by 542 and 597 bp of pilM and pilO, respectively [29]. This fragment contains no canonical DUS (ATGCCGTCTGAA), hence DUS, or DUS mutants/variants whose base composition can be precisely controlled, can be added in 3′ by using reverse primers containing the listed overhangs. Tab. 1. Primers used in this study. 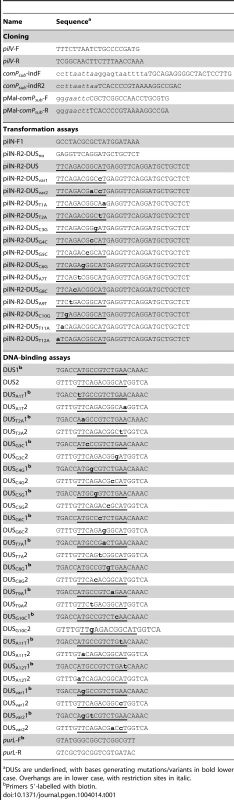
a DUSs are underlined, with bases generating mutations/variants in bold lower case. Overhangs are in lower case, with restriction sites in italic. We first quantified competence of strain 8013 in the presence and in the absence of DUS (DUSno) by counting the number of kanamycin-resistant (KmR) colonies obtained after transforming equivalent numbers of colony-forming units (CFU) with 100 ng of purified PCR fragments obtained with suitable primers (Figure 2). KmR colonies were never obtained in the absence of DNA, suggesting that all the clones obtained in this assay are genuine transformants. While 0.003% recipient cells were transformed with DUS-containing DNA, mean competence was reduced almost 25 times when no DUS was present (Figure 2). This finding, which is statistically significant (P<0.01) as assessed by a two-tailed Student's t test (P = 0.0048), indicates that DUS enhancement of transformation in 8013 is similar to what has been described previously for other strains [26], and validates our experimental approach. We next assessed transformation frequencies in isogenic comP and pilT mutants that are dramatically impaired for competence despite being piliated [24]. In the comP mutant, although there was more than a 1,000-fold decrease in competence (P = 0.0038), few transformants were reproducibly obtained (Figure 2). Importantly, there was no DUS enhancement of transformation in this mutant since similar low frequencies of transformation were obtained regardless of the presence of DUS (Figure 2). These findings are consistent with ComP being the main DNA receptor during transformation both in the presence and in the absence of DUS. When the pilT mutant was tested, we found that competence was abolished (P = 0.0037) since no transformants could be detected (Figure 2). This finding confirms that PilT which powers pilus retraction is critical for DNA transformation [12], [24], and suggests that the background level of transformation observed in the comP mutant was dependent on pilus retraction, and thus Tfp-dependent.
Fig. 2. DUS enhancement of transformation in N. meningitidis 8013 is significant and ComP-dependent. 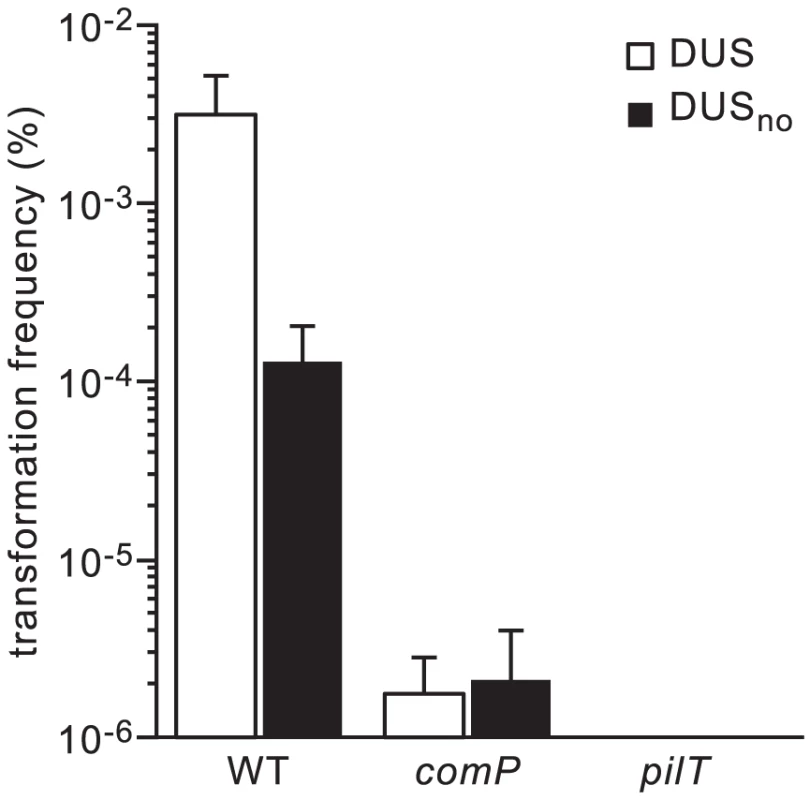
Competence for DNA transformation in strain 8013 and its isogenic comP and pilT mutants was quantified by transforming equivalent numbers of recipient cells with 100 ng of ΔpilN PCR fragments obtained using primers containing the canonical DUS (white bars), or no DUS (black bars). Recipient cells and KmR transformants were counted by plating. Results are expressed as percentages of recipient cells transformed, and are the mean ± standard deviation of six independent experiments. ComP controls transformation both in the presence and in the absence of DUS
Next, we assessed the impact of increasing levels of ComP on transformation by quantifying competence in a strain in which comP expression can be precisely controlled. In this strain comP/comPind, a wild-type (WT) copy of comP under the control of an isopropyl-β-D-thiogalactopyranoside (IPTG)-inducible promoter is integrated ectopically in the genome of a comP mutant [24]. As assessed by immunoblotting, ComP levels increased steadily with increasing concentrations of IPTG, from undetectable levels in the absence of inducer to massive over-production above 30 µM IPTG (Figure 3A). We then quantified competence in comP/comPind in the presence of increasing concentrations of IPTG, and in the presence or absence of DUS (Figure 3B). In the presence of DUS, transformation frequencies mirrored the results of the immunoblot analysis, with an increase in competence with increasing levels of ComP. However, WT-like competence in the comP/comPind strain was achieved at ComP levels slightly higher than in the WT strain, which suggests that not all ComP that is produced from comPind is functional. In the absence of IPTG, competence was low but 10-fold higher than in a comP mutant (see Figure 2) suggesting that some ComP was produced although it could not be detected by immunoblotting. At high concentrations of inducer, when ComP is massively over-produced, competence in the presence of DUS is significantly higher than in the WT strain (P = 0.0048 and 0.0016 at 125 and 500 µM IPTG, respectively) as described previously [24], [27]. These findings suggest that ComP concentration is a limiting factor for competence in a WT genetic background. Unexpectedly, when competence was quantified in the absence of DUS, a similar trend was seen (Figure 3B). There was an increase in transformation with increasing levels of ComP, with similar differences in competence between DUS and DUSno at each IPTG concentration. When ComP is massively over-produced, competence in the absence of DUS is almost as high as competence in the WT in the presence of DUS (Figure 3B). Taken together, these findings show that ComP controls transformation both in the presence and in the absence of DUS, which is consistent with our finding that although ComP binds DUS preferentially, it is capable of binding any DNA efficiently [22].
Fig. 3. ComP controls transformation both in the presence and in the absence of DUS. 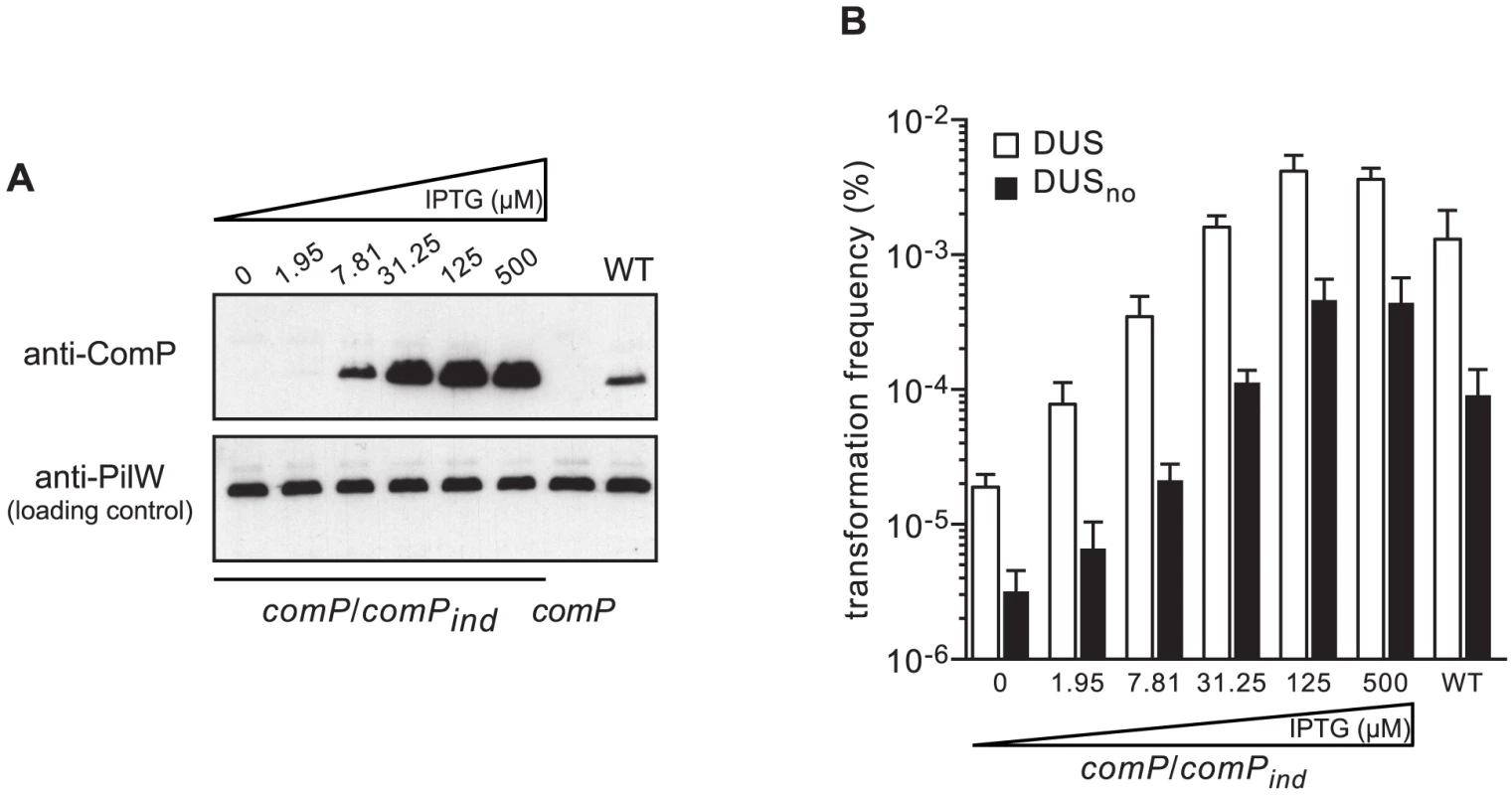
(A) Immunoblot analysis of ComP production (upper panel) in strain comP/comPind in the presence of increasing concentrations of IPTG. Strain comP/comPind is a comP mutant complemented by an ectopic WT copy of comP under the control of an IPTG-inducible promoter. The WT strain was included as a control. Equal amounts of whole-cell protein extracts were loaded in each lane, which was confirmed by detecting PilW (lower panel). (B) Quantification of competence for DNA transformation in strain comP/comPind in the presence of increasing concentrations of IPTG using ΔpilN PCR fragments obtained with primers containing the canonical DUS (white bars), or no DUS (black bars). The WT strain was included as a control. Results are expressed as percentages of recipient cells transformed, and are the mean ± standard deviation of four to six independent experiments. Peripheral bases of DUS are less important for recognition by ComP and transformation
As mentioned above, DUS is highly over-represented and conserved in the genomes of N. meningitidis and closely related species [21]. In the genome of strain 8013 there are 1,474 copies of the canonical 12 bp DUS [2]. Interestingly, there are also 682 copies of DUS differing only by one base. It is unknown whether these mutant DUS are functional. Since the relative importance of the different bases of the DUS has never been formally assessed, we used the above method to produce transforming PCR fragments that differed only by a single base of the DUS (Figure 1). To maintain overall GC content, transversion mutations were introduced in each of the 12 bases of the DUS (Table 1), and competence was quantified (Figure 4). Perhaps unexpectedly, these experiments revealed that not all bases in the DUS are functionally equivalent. First, it confirmed that the first and second bases, which were identified only recently as being part of the DUS [20], [21], are functionally important since transformation frequencies were 5-fold (P = 2.06×10−4) and 7-fold (P = 1.25×10−4) lower when these were mutated. Surprisingly, three bases (3, 11, and 12) appeared to be dispensable since transformation frequencies with the corresponding mutants were similar to WT DUS. In marked contrast, all the other inner bases (4 to 10) were critical for DUS enhancement of transformation (Figure 4) since frequencies with the corresponding mutants were down to background DUSno levels (P<5×10−5).
Fig. 4. Seven inner bases of DUS are critical for enhancement of transformation. 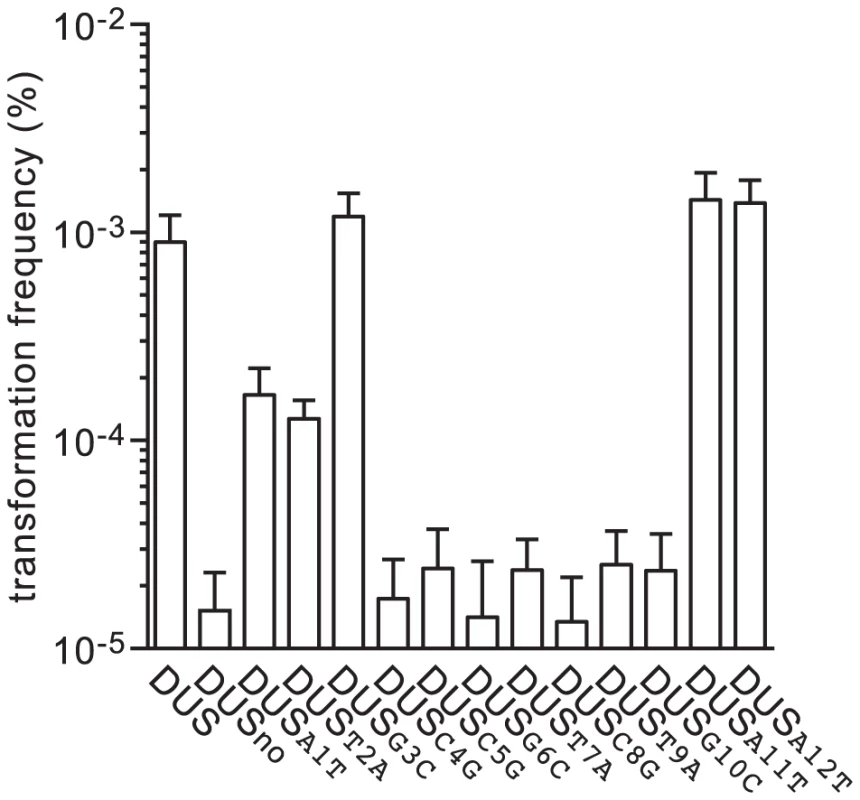
Competence for DNA transformation in N. meningitidis 8013 was quantified after transformation with 100 ng of ΔpilN PCR fragments containing a series of DUS mutants in which each base of the DUS was mutated. The specific transversion mutations are indicated in each case. As a control, the WT strain transformed with DUS and no DUS was included. Results are expressed as percentages of recipient cells transformed, and are the mean ± standard deviation of six independent experiments. In order to assess whether impaired recognition by ComP might be the reason for the observed transformation defects, we used surface plasmon resonance (SPR) as recently described [22]. We measured binding of purified ComP to double-stranded (ds) primers corresponding to each mutant DUS and compared it to binding to DUS and SDU (a scrambled primer in which every second base of DUS is changed) [22]. In brief, a neutravidin-coated sensor chip was used for immobilizing equivalent amounts of biotinylated ds primers, before 30 µM pure ComP was injected and the responses at equilibrium (Req) were measured (Figure S1). This assay revealed that the inner bases of DUS are indeed important for recognition by ComP (Figure 5), with a binding as low as binding to SDU for mutants in T7, C8, T9 and G10 (P<5×10−29). Curiously, binding was less affected for mutants in C5 and G6, and apparently not affected for mutant in C4, although these bases were crucial for transformation. Together, these findings suggest that the inner bases of DUS (7 to 10) are critical for recognition by ComP and hence transformation.
Fig. 5. Most inner bases of DUS critical for enhancement of transformation are also critical for recognition by ComP. 
Similar amounts (as assessed by measuring RU) of biotinylated ds primers corresponding to DUS, SDU (a primer in which every second base of DUS is changed) and the various DUS mutants were immobilized on sensor chips. For each DNA, Req values were determined with 30 µM of pure ComP and normalized to bound RU. Results are expressed in % as ratios to DUS Req, and are the mean ± standard deviation of 12 independent experiments. Transformation of N. meningitidis by naturally occuring DUS variants in human Neisseria commensals is dramatically impaired
Recent sequencing efforts have revealed the occurrence of gene transfers between human Neisseria species and, intriguingly, that at least some of these species harbour DUS variants [3]. When the genomes of the Neisseria species found in humans were scanned for the most frequent 12 bp repeat they contain, three variants of DUS were found. The “classical” DUS ATGCCGTCTGAA was found in N. cinerea, N. gonorrhoeae, N. lactamica, N. meningitidis and N. polysaccharea. A first variant differing by one base, AGGCCGTCTGAA (called DUSvar1), was found in N. bacilliformis, N. elongata, N. flavescens, and N. subflava (and also in strain C102 which may have been erroneously identified as N. mucosa). A second variant differing by two bases, AGGTCGTCTGAA (called DUSvar2), was found in N. mucosa and N. sicca. The above findings that DUS enhancement of transformation could be dramatically altered even by single base differences (see Figure 4) suggested that these minor naturally occuring variations in DUS could have a similar impact. We therefore used the above method to produce transforming ΔpilN PCR fragments harbouring DUSvar1 and DUSvar2 (Figure 1), and we tested their ability to transform N. meningitidis (Figure 6). The ability of DUSvar1 (which differs from DUS by only one base) to transform strain 8013 was 10-fold lower than that of DUS, but still significantly higher than DUSno. In contrast, transformation by the more variable DUSvar2 (which differs from DUS by two bases) was essentially down to levels observed in the absence of DUS (Figure 6). Intriguingly, as for some of the DUS mutants that were affected for competence but showed apparently normal binding by purified ComP (see Figure 5), SPR experiments did not reveal measurable differences in ComP binding to DUS, DUSvar1 and DUSvar2 (Figure S2). These results show that naturally occuring variations in DUS are expected to limit gene transfer between Neisseria species living in the same environment.
Fig. 6. Naturally occuring DUS variants in human Neisseria commensals are impaired for enhancement of transformation in N. meningitidis. 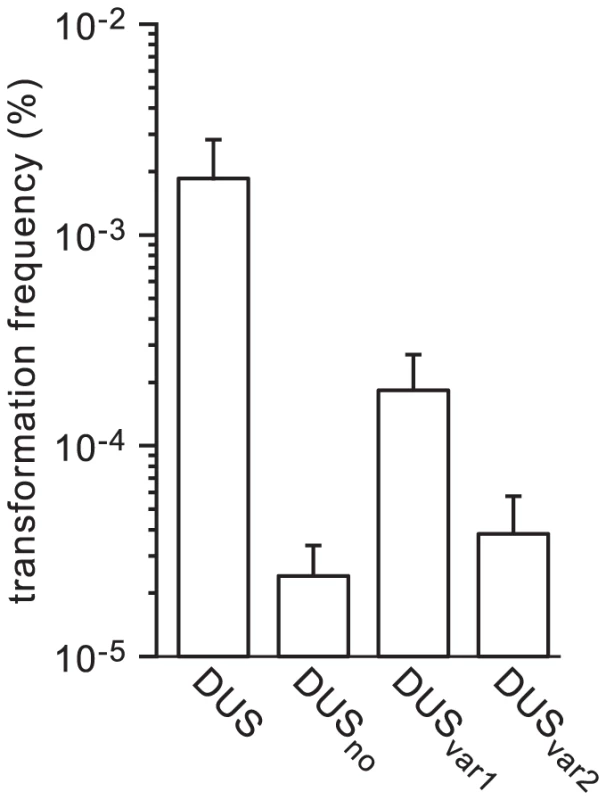
The ability of DUSvar1 and DUSvar2 to transform N. meningitidis 8013 was quantified using 100 ng of ΔpilN PCR fragments containing these DUS variants and compared to that of DUS and DUSno. Results are expressed as percentages of recipient cells transformed, and are the mean ± standard deviation of six to seven independent experiments. The ComP homolog in the N. subflava commensal preferentially binds its cognate DUS and mediates DUS-enhanced transformation when expressed in N. meningitidis
Trees derived from multiple gene phylogenies show that species harbouring the same DUS cluster together in clades [3], [25], [30], except for the species harbouring DUSvar1 that form two distinct groups. Strikingly, a very similar tree was derived from ComP sequences only (Figure 7), showing that species habouring the same DUS have highly conserved, often virtually identical, ComPs, which extends our previous observations [22]. It was therefore tempting to speculate that these ComP homologs are DNA receptors adapted to specific recognition of their cognate DUS variants, and would play a role in competence similar to meningococcal ComP. We therefore purified ComPsub from N. subflava, which is predicted to recognize DUSvar1, and tested its ability to bind DNA in vitro using electrophoretic mobility shift assays (EMSA) as previously done for meningococcal ComP [22]. We found that ComPsub has intrinsic DNA-binding ability since a shift was observed when using a biotinylated PCR fragment centered on one copy of DUSvar1 (Figure 8A). Competition assays, during which we assessed the effect of an excess of unlabelled ds primer on the formation of ComPsub-DNA complex, showed that DUSvar1 competed better than SDU as demonstrated by the gradual reappearance of the free from of biotinylated DNA (Figure 8A). This important finding confirms that ComPsub has indeed a DNA-binding preference for its cognate DUSvar1, like the one meningococcal ComP exhibits for DUS [22].
Fig. 7. Human Neisseria species harbouring different DUS variants have different cognate ComPs. 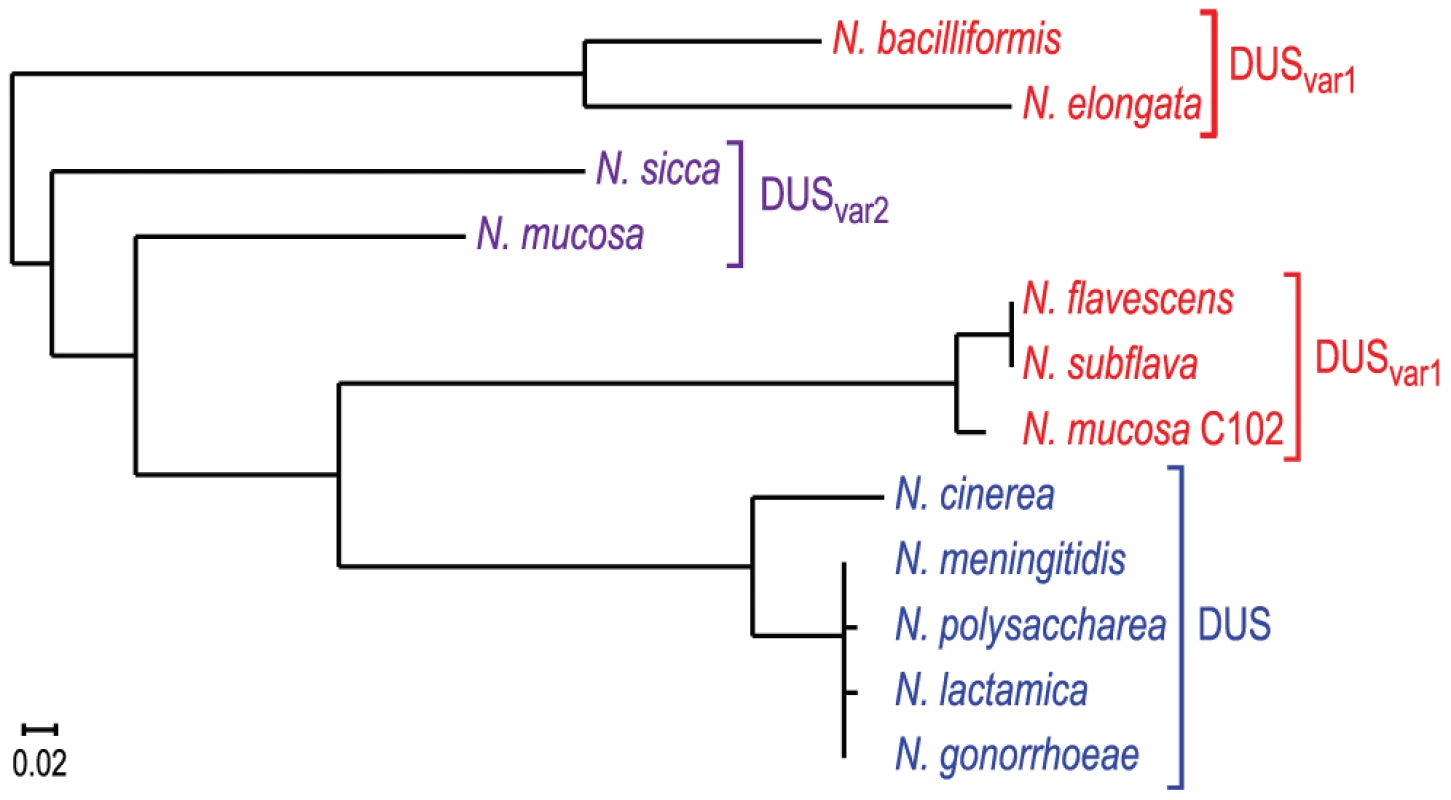
A maximum likelihood phylogenetic tree [36] based on the sequences of mature ComP found in human Neisseria species shows that species harbouring the same DUS (highlighted in the same colour) have highly conserved ComPs. Fig. 8. ComP homologs in other human Neisseria species are DNA receptors playing a similar role in transformation to meningococcal ComP. 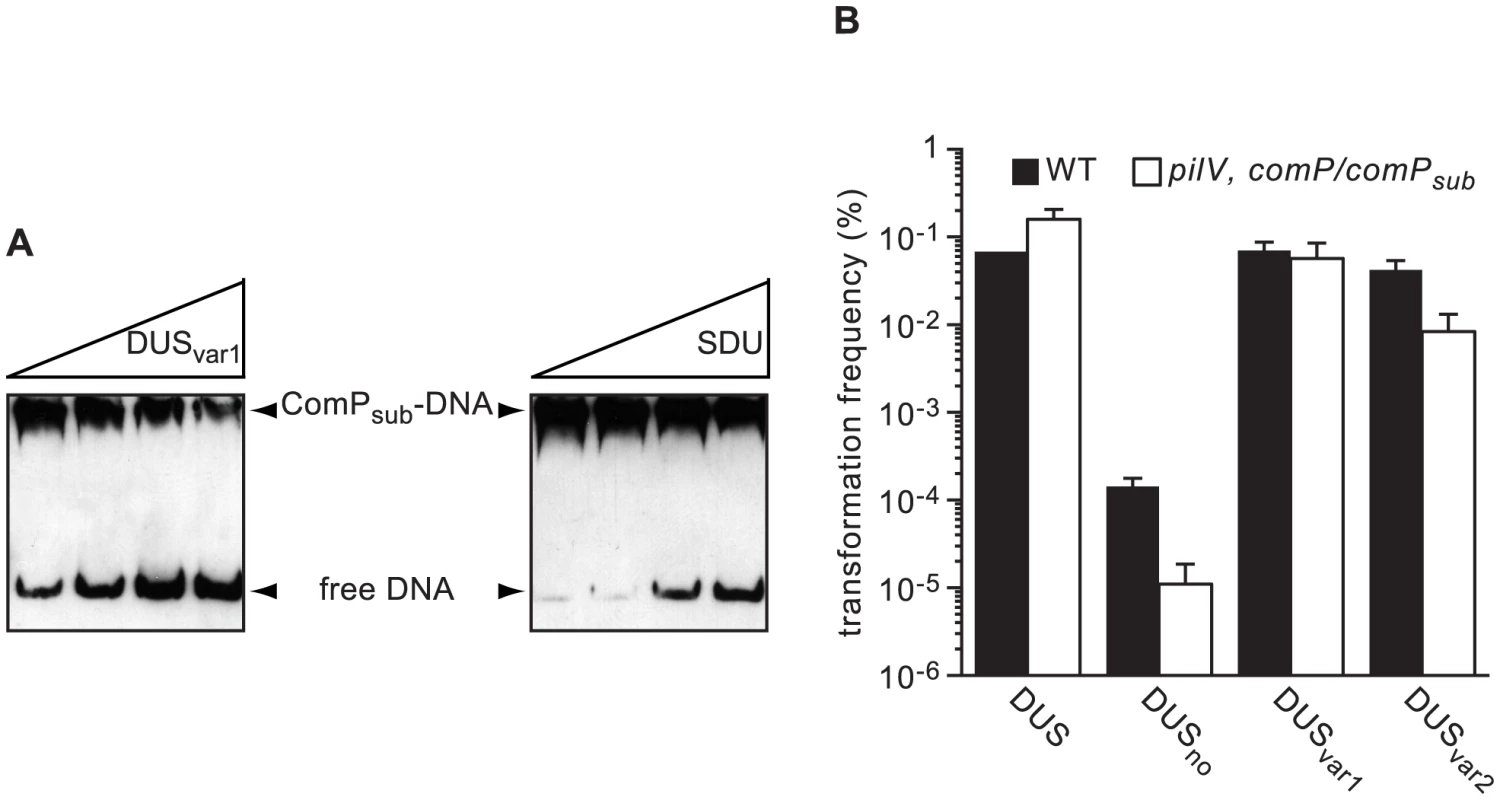
(A) Effect on the ComPsub-DNA complex of the addition of increasing amounts of unlabelled competitor as assessed by acrylamide EMSA. ComPsub and biotin-labelled PCR fragment containing one copy of DUSvar1 were incubated with two-fold increasing concentrations of unlabelled ds DUSvar1 or SDU primers. DNA was then resolved by electrophoresis on an acrylamide native gel, transferred to a positive nylon membrane, UV-crosslinked and detected using a streptavidin-HRP conjugate. (B) Quantification of competence for DNA transformation in the strain 8013 (black bars) and an engineered derivative expressing ComPsub (white bars). Strains were transformed in the presence of 500 µM IPTG using 10 ng of plasmids in which ΔpilN mutant PCR fragments containing DUS, DUSno, DUSvar1 and DUSvar2 have been cloned. Results are expressed as percentages of recipient cells transformed, and are the mean ± standard deviation of five independent experiments. Next, we tested the functionality of ComPsub in vivo by integrating the corresponding gene under the control of an IPTG-inducible promoter ectopically in the genome of a N. meningitidis comP mutant. When genomic DNA from a N. meningitidis ΔpilN mutant was used for transforming comP/comPsub, there was a 56-fold increase in competence (P = 0.003) when compared to the comP mutant (Figure S3), which confirms that ComP and ComPsub play a similar role in competence. When a pilV mutation was introduced in comP/comPsub, competence was even as high as in the WT strain, which is consistent with PilV's role as an antagonist of ComP [31]. Although pilV, comP/comPsub could not be transformed by ΔpilN PCR fragments (data not shown), it could be transformed by plasmids in which these fragments PCR fragments had been cloned, which allowed us to test the effect of various DUS on transformation (Figure 8B). Surprisingly, and for an unknown reason, when plasmids were used for transformation of strain 8013 although there was a massive reduction of competence when no DUS was present (almost 500-fold), the specificity toward the various DUS was relaxed since the ability of DUSvar1 to transform this strain was similar to that of DUS, while transformation by DUSvar2 was only 2-fold lower. Importantly, the pilV, comP/comPsub strain behaved similarly (Figure 8B), with a dramatic reduction of competence when no DUS was present, similar transformation frequencies by DUS and DUSvar1, and a significantly impaired transformation by DUSvar2. These results confirm that DUS-specific enhancement of transformation is a property conserved among ComP orthologs.
Discussion
Although most, if not all [32], genes involved in competence have now been identified in naturally transformable model species, our understanding of this multi-step and complex biological property is still fragmentary. Until recently, possibly the least well understood steps of natural transformation were the very first one (how the free DNA is detected and bound by Tfp/competence pseudopili) and the last one (how competent bacteria manage to preserve species structure by avoiding uncontrolled transformation by foreign DNA). The recent identification of ComP as the Tfp-exposed DNA receptor in pathogenic Neisseria species [22], has provided some answers to these questions. We found that N. meningitidis Tfp bind DNA through ComP, and that this minor pilin is at the basis of the trademark selective uptake of homotypic DNA by Neisseria species because of its marked preference for the signature DUS found in their genomes [21]. These findings paved the way to previously unfeasible experiments aimed at understanding the interdependence between DUS and its cognate receptor, and its impact on natural transformation.
The first important finding in this study is that ComP is a key player in natural transformation by controlling both DUS-mediated transformation and, unexpectedly, transformation in the absence of DUS. As expected, when DUS was present in the transforming DNA, there was a marked enhancement of transformation when compared to DNA without DUS. It is important to note, however, that transformation in the absence of DUS remains fairly efficient, and much higher (100-fold) than in a non-competent mutant such as pilT. In the absence of ComP, competence was very low and the presence of DUS did not make any difference, strengthening the notion that DUS enhancement is mediated by ComP, and suggesting that ComP is responsible for DUSno transformation as well. This latter hypothesis was confirmed by the finding that overproduction of ComP resulted in increased transformation not only by DUS-containing DNA, but also by DNA devoid of DUS. Therefore, together with our previous results [22], [24], these findings suggest that ComP is the primary DNA receptor during natural transformation in Neisseria species, and are consistent with the following scenario. When free DNA is present, and as a result of ComP's demonstrated ability to bind any DNA [22], it is bound by Neisseria Tfp through ComP irrespective of the presence of DUS. Upon Tfp retraction, bound DNA is taken up and later incorporated in the genome (provided that it is similar enough to Neisseria DNA), as indicated by the significant transformation seen both in the presence and in the absence of DUS. However, ComP's higher affinity for DUS makes this process much more efficient with DUS-containing DNA, resulting in the well-known enhancement of transformation [21], [26] also observed here. In the absence of ComP, DNA-binding by Tfp (the mechanism for this residual binding remains to be identified) and subsequent uptake are inefficient, resulting in dramatically reduced transformation, irrespective of the presence of DUS.
The second major finding in this study is that evolution has finely tailored DUSs to their cognate ComP receptors. A mutational analysis showed that several single base substitutions in DUS (especially bases 7, 8, 9, and 10) resulted in impaired interaction with ComP and a total loss of enhancement of transformation. Nevertheless, one of the DUS mutants that was dramatically affected for competence (DUSC4G) was bound by ComP as efficiently as WT DUS. However, it is not impossible to imagine (among other things) that DNA binding is much tighter when ComP is within Tfp and that a minor binding defect of DUSC4G undetectable in vitro might be significant enough in vivo for the bound DNA not to be taken up efficiently, which is supported by a very recent study [33] (see Note added in proof). In contrast, the peripheral bases in the DUS were found to be less important for transformation, but they are likely to play an important role (still to be identified) explaining why they are highly conserved in Neisseria genomes. Interestingly, a majority (72%) of the 682 copies of DUS with one base substitution that were identified in the genome of strain 8013 harbour mutations in one of the five peripheral positions and could therefore be, at least partly, functional variants. Similarly, DUSvar1 and DUSvar2 naturally occuring in human Neisseria commensals that are part of the normal flora in the nasopharynx [25], were found to be impaired for enhancement of transformation in N. meningitidis. Consistent with our mutational analysis, which showed that DUST2A was still partly functional for competence while DUSC4G was not, DUSvar1 that differs from DUS at base 2 was still capable of enhancing transformation significantly (although much less than DUS) while DUSvar2 that harbours a second change at base 4 was not. However, as for DUST2A and DUSC4G, no ComP binding defect could be detected for DUSvar1 and DUSvar2 in vitro, but again a minor defect undetectable in vitro with significant consequences in vivo [33] remains possible. Finally, we provide evidence that ComP homologs in human Neisseria commensals play a similar role in transformation as indicated by the findings that ComPsub from N. subflava binds DNA in vitro with a preference for its cognate DUSvar1, and is capable of fully complementing the competence defect in a N. meningitidis comP mutant with a dramatic (>10,000-fold) enhancement of transformation in the presence of DUS. Surprisingly, the use of plasmids (since for an unknown reason ComPsub-expressing N. meningitidis strains are not transformable by PCR fragments) led to relaxed DUS-specificity, preventing us from demonstrating that expression of a different comP allele in the meningococcus leads to a switch in DUS specificity.
In conclusion, this study reveals a previously unrecognized mechanism that likely plays an important role in helping the many competent species that inhabit the human nasopharynx curb transformation by foreign DNA and preserve species structure, i.e. through co-evolution of DUSs and cognate pilin receptors. Although transforming DNA identity will also play an important part, it is possible to predict that N. meningitidis will preferably acquire genes from (in the following order) meningococci, species that share the same DUS (N. cinerea, N. lactamica, and N. polysaccharea), species containing DUSvar1, species containing DUSvar2, and then other species. Likewise, other competent Neisseriaceae will use their ComPs (or other unrelated pilin receptors in the Pasteurellaceae) in a similar fashion to promote intra-species gene transfer in a “crowded” environment such as the human nasopharynx.
Note added in proof
While the work presented here was under review, Frye et al. following a DUS-centered approach reported that a variety of DUSs they nicely nicknamed “dialects” are present not only in Neisseria species but in most members of the Neisseriaceae family [33], which we previously showed harboured ComP homologs [22]. They confirmed that Kingella denitrificans and Eikenella corrodens are transformed better by DNA containing their own DUS dialect, although no selective DNA uptake could be demonstrated. Using a mutational analysis similar to ours, they found that the inner bases of the DUS are important for both DNA uptake and transformation, which we show here is due to impaired interaction with ComP. Finally, they show that different DUS dialects transform the meningococcus to various extents, which was importantly correlated to differences in DNA uptake. These findings strengthen the notion that the mechanism we have identified here is widespread.
Materials and Methods
Bacterial strains, plasmids and primers
N. meningitidis was grown on GC medium base (GCB) plates (Difco) containing Kellogg's supplements and, when required, 100 µg/ml kanamycin, 40 µg/ml spectinomycin, 3 µg/ml erythromycin, 5 µg/ml chloramphenicol (all antibiotics were from Sigma), or various concentrations of IPTG (Merck Chemicals). Plates were incubated overnight in a moist atmosphere containing 5% CO2. N. meningitidis strains used in this study have all been described. The WT strain is a derivative of the sequenced serogroup C clinical isolate 8013 [2]. In the comP and pilT isogenic mutants the corresponding genes were interrupted by the cloning of cassettes encoding resistance to spectinomycin and erythromycin, respectively [24]. Similarly, a pilV isogenic mutant was constructed by transforming 8013 with a pCR8/GW/TOPO-derived vector (Invitrogen) into which the pilV gene, amplified with pilV-F and pilV-R primers (Table 1) (all primers were from Invitrogen), had been interrupted by the cloning, in a unique NcoI site, of a chloramphenicol cassette extracted from pT1-Cm1 [34]. In the ΔpilN mutant, constructed by splicing PCR, the pilN gene is cleanly replaced by a kanamycin resistance cassette [29]. The comP/comPind strain was obtained by transforming the above comP mutation in comPind strain that contains an ectopic copy of comP under an IPTG-inducible promoter [24]. Using a similar approach, the comPsub allele from N. subflava was amplified with comPsub-indF and comPsub-indR2, cloned under an IPTG-inducible promoter and integrated in the genome of 8013, generating strain comPsub. The comP/comPsub and pilV, comP/comPsub strains were obtained by transforming comPsub with the above comP and/or pilV mutations.
E. coli DH5α was used for cloning experiments, while E. coli BL21(DE3) was used for protein expression and purification experiments. E. coli was routinely grown in liquid or solid Luria-Bertani (LB) medium (Difco) containing, when required, 100 µg/ml spectinomycin or 100 µg/ml ampicillin. Ultra-competent cells were prepared as described elsewhere [35]. The derivative of the pMAL-p2X expression vector (New England Biolabs) encoding a fusion between MBP and the soluble portion of ComP has been described previously [22]. A pMAL-p2X derivative encoding a fusion between MBP and the soluble portion of N. subflava ComP (ComPsub) was constructed in the same way. We generated pCR8/GW/TOPO-derived vectors into which various ΔpilN PCR fragments have been directly cloned. When doing competence assays with PCR fragments, we used the pCR8/GW/TOPO-derived vector with no DUS as a template for generating all the transforming PCR fragments. This was done to avoid “contamination” by using DUS-containing genomic DNA of the ΔpilN mutant as a template for PCRs. PCRs were done using the high-fidelity Herculase II Fusion DNA polymerase (Agilent). PCR fragments were purified using QIAquick PCR purification kit (QIAGEN) and quantified using a Nanodrop Lite spectrophotometer (Labtech International). When required, overhangs corresponding to DUS, mutant DUS or naturally occuring variants of DUS were added to the R2 primers (Table 1).
Competence assays
Natural competence for DNA transformation was quantified as previously described [24]. In brief, bacteria grown overnight on agar plates were resuspended at an OD600 of 1 in liquid GCB containing 5 mM MgCl2 (called GCB transfo). When the assays were done with comP/comPind, comP/comPsub and pilV, comP/comPsub, IPTG at desired concentrations was added into the GCB transfo. Input CFU were determined by plating serial dilutions on GCB plates containing no antibiotic. Two hundred µl of the bacterial suspension were aliquoted in 24-well plates and mixed with either 100 ng of purified ΔpilN PCR fragments, 200 ng of genomic DNA extracted from a ΔpilN mutant, or 10 ng of pCR8/GW/TOPO-derived vectors into which the various ΔpilN PCR fragments had been cloned. After incubating bacteria together with DNA for 30 min at 37°C on an orbital shaker (140 rpm), 0.8 ml GCB transfo was added to each well and plates were further incubated 3 h at 37°C, without shaking. Appropriate dilutions were spread on GCB plates containing kanamycin and the number of KmR transformants obtained were counted the next day. Results were expressed as percentage of recipient cells transformed.
Protein purification and DNA binding assays
ComP without its highly conserved and hydrophobic N-terminal α-helix was purified as previously described [22]. First, a MBP-trap HP column (GE Healthcare) was used to purify MBP-ComP in 50 mM sodium phosphate pH 7.5, 50 mM NaCl. After cleavage of the MBP moiety with factor Xa (New England Biolabs), the buffer was exchanged with 50 mM sodium phosphate pH 6.7, and free ComP was recovered in the same buffer containing 1 M NaCl on a HiTrap CM FF ion exchange column (GE Healthcare). ComPsub was purified in a similar way.
The binding of purified ComP to various DUS sequences was measured by SPR as described previously [22], with minor modifications. Double-stranded primers corresponding to the various motifs (Table 1) were prepared by mixing complementary primers (one of which was 5′-labeled with biotin) with a 10-fold excess of the unlabelled primer, incubating the mixture at 100°C during 5 min, and slowly cooling to room temperature. Biotin-labelled double-stranded primers corresponding to the various motifs (Table 1) were coupled to neutravidin on the surface of ProteOn NLC sensor chips (BioRad), resulting in ∼230 RU increase. SPR was then performed on a ProteOn XPR36 protein interaction array system instrument (BioRad) operated at 25°C. ComP protein at 30 µM (in 25 mM HEPES pH 7.9, 150 mM NaCl, 2.5 mM MgCl2, 0.05% Tween-20) was then flowed over the chip surface at a flow-rate of 40 µl/min and Req were recorded. A control trace was collected using an uncoupled channel. For each DNA, we did 12 independent measurements using two separate injections of ComP over six channels.
The ability of purified ComPsub to bind DNA was assessed by acrylamide EMSA as previously done for ComP [22]. In brief, we used as a target 1 fmole a biotin-labelled PCR fragment (152 bp) centered on one copy of DUSvar1, which corresponds to an inner portion of the purL gene from 8013 [2]. For competition assays, two-fold increasing concentrations of unlabelled DUSvar1 or SDU ds primers (from 22.5 to 180 pmoles) were added in a reaction containing 0.8 µM ComPsub prior to the addition of biotin-labelled PCR fragment. DNA was then separated by electrophoresis on native 10% acrylamide gels, transferred to positively charged membranes and detected using the LightShift chemiluminescent EMSA kit (Pierce).
SDS-PAGE and immunoblotting
Separation of proteins by SDS-PAGE and immunoblotting was done as described previously [29]. Gels were blotted to Amersham Hybond ECL membranes and detection was performed using Amersham ECL Prime (both from GE Healthcare).
Bioinformatics
To identify the most over-represented sequence motifs in different Neisseria genomes we used the wordcount application from the EMBOSS suite (http://emboss.bioinformatics.nl). To count the number of occurrences of defined motifs in a genome, with or without mismatches, we used the fuzznuc program from the Mobyle portal (http://mobyle.pasteur.fr/). To construct a phylogenetic tree from ComPs found in human Neisseria species, we used the protein sequences corresponding to mature ComP (obtained by removing the short leader sequence of the corresponding pre-proteins) and the Phylogeny.fr platform (http://www.phylogeny.fr) in “One Click” mode [36].
Supporting Information
Zdroje
1. JohnsborgO, EldholmV, HåvarsteinLS (2007) Natural genetic transformation: prevalence, mechanisms and function. Res Microbiol 158 : 767–778.
2. RusniokC, VallenetD, FloquetS, EwlesH, Mouzé-SoulamaC, et al. (2009) NeMeSys: a resource for narrowing the gap between sequence and function in the human pathogen Neisseria meningitidis. Genome Biol 10: R110.
3. MarriPR, PaniscusM, WeyandNJ, RendonMA, CaltonCM, et al. (2010) Genome sequencing reveals widespread virulence gene exchange among human Neisseria species. PLoS One 5: e11835.
4. KrollJS, WilksKE, FarrantJL, LangfordPR (1998) Natural genetic exchange between Haemophilus and Neisseria: intergeneric transfer of chromosomal genes between major human pahogens. Proc Natl Acad Sci USA 65 : 12381–12385.
5. VirjiM (2009) Pathogenic Neisseriae: surface modulation, pathogenesis and infection control. Nat Rev Microbiol 7 : 274–286.
6. GriffithF (1928) The significance of pneumococcal types. J Hyg (Lond) 27 : 113–159.
7. AveryOT, MacLeodCM, McCartyM (1944) Studies on the chemical nature of the substance inducing transformation of pneumococcal types: induction of transformation by a desoxyribonucleic acid fraction isolated from pneumococcus type III. J Exp Med 79 : 137–158.
8. ChenI, DubnauD (2004) DNA uptake during bacterial transformation. Nat Rev Microbiol 2 : 241–249.
9. CehovinA, KrollJS, PelicicV (2011) Testing the vaccine potential of PilV, PilX and ComP, minor subunits of Neisseria meningitidis type IV pili. Vaccine 29 : 6858–6865.
10. PelicicV (2008) Type IV pili: e pluribus unum? Mol Microbiol 68 : 827–837.
11. MaierB, PotterL, SoM, LongCD, SeifertHS, et al. (2002) Single pilus motor forces exceed 100 pN. Proc Natl Acad Sci USA 99 : 16012–16017.
12. WolfgangM, LauerP, ParkHS, BrossayL, HébertJ, et al. (1998) PilT mutations lead to simultaneous defects in competence for natural transformation and twitching motility in piliated Neisseria gonorrhoeae. Mol Microbiol 29 : 321–330.
13. MaierB, ChenI, DubnauD, SheetzMP (2004) DNA transport into Bacillus subtilis requires proton motive force to generate large molecular forces. Nat Struct Mol Biol 11 : 643–649.
14. InamineGS, DubnauD (1995) ComEA, a Bacillus subtilis integral membrane protein required for genetic transformation, is needed for both DNA binding and transport. J Bacteriol 177 : 3045–3051.
15. ChenI, GotschlichEC (2001) ComE, a competence protein from Neisseria gonorrhoeae with DNA-binding activity. J Bacteriol 183 : 3160–3168.
16. DraskovicI, DubnauD (2005) Biogenesis of a putative channel protein, ComEC, required for DNA uptake: membrane topology, oligomerization and formation of disulphide bonds. Mol Microbiol 55 : 881–896.
17. Londoño-VallejoJA, DubnauD (1994) Mutation of the putative nucleotide binding site of the Bacillus subtilis membrane protein ComFA abolishes the uptake of DNA during transformation. J Bacteriol 176 : 4642–4645.
18. AmburOH, FryeSA, NilsenM, HovlandE, TønjumT (2012) Restriction and sequence alterations affect DNA uptake sequence-dependent transformation in Neisseria meningitidis. PLoS One 7: e39742.
19. GoodmanSD, ScoccaJJ (1988) Identification and arrangement of the DNA sequence recognized in specific transformation of Neisseria gonorrhoeae. Proc Natl Acad Sci USA 85 : 6982–6986.
20. SmithHO, GwinnML, SalzbergSL (1999) DNA uptake signal sequences in naturally transformable bacteria. Res Microbiol 150 : 603–616.
21. AmburOH, FryeSA, TønjumT (2007) New functional identity for the DNA uptake sequence in transformation and its presence in transcriptional terminators. J Bacteriol 189 : 2077–2085.
22. CehovinA, SimpsonPJ, McDowellMA, BrownDR, NoscheseR, et al. (2013) Specific DNA recognition mediated by a type IV pilin. Proc Natl Acad Sci USA 110 : 3065–3070.
23. WolfgangM, van PuttenJP, HayesSF, KoomeyM (1999) The comP locus of Neisseria gonorrhoeae encodes a type IV prepilin that is dispensable for pilus biogenesis but essential for natural transformation. Mol Microbiol 31 : 1345–1357.
24. BrownD, HelaineS, CarbonnelleE, PelicicV (2010) Systematic functional analysis reveals that a set of 7 genes is involved in fine tuning of the multiple functions mediated by type IV pili in Neisseria meningitidis. Infect Immun 78 : 3053–3063.
25. Tønjum T (2005) Family Neisseriaceae. In: Garrity GM, editor. Bergey's Manual of Systematic Bacteriology. 2nd ed. New York Springer Verlag pp. 775–776.
26. DuffinPM, SeifertHS (2010) DNA uptake sequence-mediated enhancement of transformation in Neisseria gonorrhoeae is strain dependent. J Bacteriol 192 : 4436–4444.
27. AasFE, WolfgangM, FryeS, DunhamS, LøvoldC, et al. (2002) Competence for natural transformation in Neisseria gonorrhoeae: components of DNA binding and uptake linked to type IV pilus expression. Mol Microbiol 46 : 749–760.
28. CarbonnelleE, HelaineS, NassifX, PelicicV (2006) A systematic genetic analysis in Neisseria meningitidis defines the Pil proteins required for assembly, functionality, stabilization and export of type IV pili. Mol Microbiol 61 : 1510–1522.
29. GeorgiadouM, CastagniniM, KarimovaG, LadantD, PelicicV (2012) Large-scale study of the interactions between proteins involved in type IV pilus biology in Neisseria meningitidis: characterization of a subcomplex involved in pilus assembly. Mol Microbiol 84 : 857–873.
30. BennettJS, JolleyKA, EarleSG, CortonC, BentleySD, et al. (2012) A genomic approach to bacterial taxonomy: an examination and proposed reclassification of species within the genus Neisseria. Microbiology 158 : 1570–1580.
31. AasFE, LøvoldC, KoomeyM (2002) An inhibitor of DNA binding and uptake events dictates the proficiency of genetic transformation in Neisseria gonorrhoeae: mechanism of action and links to type IV pilus expression. Molecular Microbiology 46 : 1441–1450.
32. SinhaS, MellJC, RedfieldRJ (2012) Seventeen Sxy-dependent cyclic AMP receptor protein site-regulated genes are needed for natural transformation in Haemophilus influenzae. J Bacteriol 194 : 5245–5254.
33. FryeSA, NilsenM, TønjumT, AmburOH (2013) Dialects of the DNA uptake sequence in Neisseriaceae. PLoS Genet 9: e1003458.
34. TinsleyCR, VoulhouxR, BerettiJL, TommassenJ, NassifX (2004) Three homologues, including two membrane-bound proteins, of the disulphide oxidoreductase DsbA in Neisseria meningitidis: effects on bacterial growth and biogenesis of functional type IV pili. J Biol Chem 279 : 27078–27087.
35. InoueH, NojimaH, OkayamaH (1990) High efficiency transformation of Escherichia coli with plasmids. Gene 96 : 23–28.
36. DereeperA, GuignonV, BlancG, AudicS, BuffetS, et al. (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36: W465–469.
Štítky
Genetika Reprodukční medicína
Článek Interaction between and during Mammalian Jaw Patterning and in the Pathogenesis of SyngnathiaČlánek Clustering of Tissue-Specific Sub-TADs Accompanies the Regulation of Genes in Developing LimbsČlánek Transcription Factor Occupancy Can Mediate Active Turnover of DNA Methylation at Regulatory RegionsČlánek Tay Bridge Is a Negative Regulator of EGFR Signalling and Interacts with Erk and Mkp3 in the Wing
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2013 Číslo 12- Akutní intermitentní porfyrie
- Farmakogenetické testování pomáhá předcházet nežádoucím efektům léčiv
- Růst a vývoj dětí narozených pomocí IVF
- IVF a rakovina prsu – zvyšují hormony riziko vzniku rakoviny?
- Pilotní studie: stres a úzkost v průběhu IVF cyklu
-
Všechny články tohoto čísla
- Stressing the Importance of CHOP in Liver Cancer
- The AmAZI1ng Roles of Centriolar Satellites during Development
- Flies Get a Head Start on Meiosis
- Recommendations from Jane Gitschier's Bookshelf
- And Baby Makes Three: Genomic Imprinting in Plant Embryos
- Bugs in Transition: The Dynamic World of in Insects
- Defining the Role of ATP Hydrolysis in Mitotic Segregation of Bacterial Plasmids
- Synaptonemal Complex Components Promote Centromere Pairing in Pre-meiotic Germ Cells
- Cohesinopathies of a Feather Flock Together
- Genetic Recombination Is Targeted towards Gene Promoter Regions in Dogs
- Parathyroid-Specific Deletion of Unravels a Novel Calcineurin-Dependent FGF23 Signaling Pathway That Regulates PTH Secretion
- MAN1B1 Deficiency: An Unexpected CDG-II
- Phosphate Flow between Hybrid Histidine Kinases CheA and CheS Controls Cyst Formation
- Basolateral Mg Extrusion via CNNM4 Mediates Transcellular Mg Transport across Epithelia: A Mouse Model
- Truncation of Unsilences Paternal and Ameliorates Behavioral Defects in the Angelman Syndrome Mouse Model
- Autozygome Sequencing Expands the Horizon of Human Knockout Research and Provides Novel Insights into Human Phenotypic Variation
- Huntington's Disease Induced Cardiac Amyloidosis Is Reversed by Modulating Protein Folding and Oxidative Stress Pathways in the Heart
- Low Frequency Variants, Collapsed Based on Biological Knowledge, Uncover Complexity of Population Stratification in 1000 Genomes Project Data
- Targeted Ablation of and in Retinal Progenitor Cells Mimics Leber Congenital Amaurosis
- Genomic Imprinting in the Embryo Is Partly Regulated by PRC2
- Binary Cell Fate Decisions and Fate Transformation in the Larval Eye
- The Stress-Regulated Transcription Factor CHOP Promotes Hepatic Inflammatory Gene Expression, Fibrosis, and Oncogenesis
- A Global RNAi Screen Identifies a Key Role of Ceramide Phosphoethanolamine for Glial Ensheathment of Axons
- Functional Analysis of the Interdependence between DNA Uptake Sequence and Its Cognate ComP Receptor during Natural Transformation in Species
- Cross-Modulation of Homeostatic Responses to Temperature, Oxygen and Carbon Dioxide in
- Alcohol-Induced Histone Acetylation Reveals a Gene Network Involved in Alcohol Tolerance
- Molecular Characterization of Host-Specific Biofilm Formation in a Vertebrate Gut Symbiont
- CRIS—A Novel cAMP-Binding Protein Controlling Spermiogenesis and the Development of Flagellar Bending
- Dual Regulation of the Mitotic Exit Network (MEN) by PP2A-Cdc55 Phosphatase
- Expanding the Marine Virosphere Using Metagenomics
- Detection of Slipped-DNAs at the Trinucleotide Repeats of the Myotonic Dystrophy Type I Disease Locus in Patient Tissues
- Interaction between and during Mammalian Jaw Patterning and in the Pathogenesis of Syngnathia
- Mutations in the UQCC1-Interacting Protein, UQCC2, Cause Human Complex III Deficiency Associated with Perturbed Cytochrome Protein Expression
- Reactivation of Chromosomally Integrated Human Herpesvirus-6 by Telomeric Circle Formation
- Anoxia-Reoxygenation Regulates Mitochondrial Dynamics through the Hypoxia Response Pathway, SKN-1/Nrf, and Stomatin-Like Protein STL-1/SLP-2
- The Midline Protein Regulates Axon Guidance by Blocking the Reiteration of Neuroblast Rows within the Drosophila Ventral Nerve Cord
- Tomato Yield Heterosis Is Triggered by a Dosage Sensitivity of the Florigen Pathway That Fine-Tunes Shoot Architecture
- Selection on Plant Male Function Genes Identifies Candidates for Reproductive Isolation of Yellow Monkeyflowers
- Role of Tomato Lipoxygenase D in Wound-Induced Jasmonate Biosynthesis and Plant Immunity to Insect Herbivores
- Meiotic Cohesin SMC1β Provides Prophase I Centromeric Cohesion and Is Required for Multiple Synapsis-Associated Functions
- Identification of Sphingolipid Metabolites That Induce Obesity via Misregulation of Appetite, Caloric Intake and Fat Storage in
- Genome-Wide Screen Reveals Replication Pathway for Quasi-Palindrome Fragility Dependent on Homologous Recombination
- Histone Methylation Restrains the Expression of Subtype-Specific Genes during Terminal Neuronal Differentiation in
- A Novel Intergenic ETnII-β Insertion Mutation Causes Multiple Malformations in Mice
- The NuRD Chromatin-Remodeling Enzyme CHD4 Promotes Embryonic Vascular Integrity by Transcriptionally Regulating Extracellular Matrix Proteolysis
- A Domesticated Transposase Interacts with Heterochromatin and Catalyzes Reproducible DNA Elimination in
- Acute Versus Chronic Loss of Mammalian Results in Distinct Ciliary Phenotypes
- MBD3 Localizes at Promoters, Gene Bodies and Enhancers of Active Genes
- Positive and Negative Regulation of Gli Activity by Kif7 in the Zebrafish Embryo
- A Hereditary Spastic Paraplegia Mouse Model Supports a Role of ZFYVE26/SPASTIZIN for the Endolysosomal System
- The CCR4-NOT Complex Mediates Deadenylation and Degradation of Stem Cell mRNAs and Promotes Planarian Stem Cell Differentiation
- Reconstructing Native American Migrations from Whole-Genome and Whole-Exome Data
- Contributions of Protein-Coding and Regulatory Change to Adaptive Molecular Evolution in Murid Rodents
- Comprehensive Analysis of Transcriptome Variation Uncovers Known and Novel Driver Events in T-Cell Acute Lymphoblastic Leukemia
- A -Acting Protein Effect Causes Severe Eye Malformation in the Mouse
- Clustering of Tissue-Specific Sub-TADs Accompanies the Regulation of Genes in Developing Limbs
- Germline Progenitors Escape the Widespread Phenomenon of Homolog Pairing during Development
- Transcription Factor Occupancy Can Mediate Active Turnover of DNA Methylation at Regulatory Regions
- Somatic mtDNA Mutation Spectra in the Aging Human Putamen
- ESCRT-I Mediates FLS2 Endosomal Sorting and Plant Immunity
- Ethylene Promotes Hypocotyl Growth and HY5 Degradation by Enhancing the Movement of COP1 to the Nucleus in the Light
- The PAF Complex and Prf1/Rtf1 Delineate Distinct Cdk9-Dependent Pathways Regulating Transcription Elongation in Fission Yeast
- Dual Regulation of Gene Expression Mediated by Extended MAPK Activation and Salicylic Acid Contributes to Robust Innate Immunity in
- Quantifying Missing Heritability at Known GWAS Loci
- Smc5/6-Mms21 Prevents and Eliminates Inappropriate Recombination Intermediates in Meiosis
- Smc5/6 Coordinates Formation and Resolution of Joint Molecules with Chromosome Morphology to Ensure Meiotic Divisions
- Tay Bridge Is a Negative Regulator of EGFR Signalling and Interacts with Erk and Mkp3 in the Wing
- Meiotic Crossover Control by Concerted Action of Rad51-Dmc1 in Homolog Template Bias and Robust Homeostatic Regulation
- Active Transport and Diffusion Barriers Restrict Joubert Syndrome-Associated ARL13B/ARL-13 to an Inv-like Ciliary Membrane Subdomain
- An Regulatory Circuit Modulates /Wnt Signaling and Determines the Size of the Midbrain Dopaminergic Progenitor Pool
- Variants Induce Differential Protection to Viruses in : A Phenotypic and Phylogenomic Analysis
- Base Pairing Interaction between 5′- and 3′-UTRs Controls mRNA Translation in
- Evidence That Masking of Synapsis Imperfections Counterbalances Quality Control to Promote Efficient Meiosis
- Insulin/IGF-Regulated Size Scaling of Neuroendocrine Cells Expressing the bHLH Transcription Factor in
- Sumoylated NHR-25/NR5A Regulates Cell Fate during Vulval Development
- TATN-1 Mutations Reveal a Novel Role for Tyrosine as a Metabolic Signal That Influences Developmental Decisions and Longevity in
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- The NuRD Chromatin-Remodeling Enzyme CHD4 Promotes Embryonic Vascular Integrity by Transcriptionally Regulating Extracellular Matrix Proteolysis
- Mutations in the UQCC1-Interacting Protein, UQCC2, Cause Human Complex III Deficiency Associated with Perturbed Cytochrome Protein Expression
- The Midline Protein Regulates Axon Guidance by Blocking the Reiteration of Neuroblast Rows within the Drosophila Ventral Nerve Cord
- Tomato Yield Heterosis Is Triggered by a Dosage Sensitivity of the Florigen Pathway That Fine-Tunes Shoot Architecture
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



