-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Meropenem serum concentrations in intensive care patients: a retrospective analysis
Sérové koncentrace meropenemu u pacientů vyžadujících intenzivní péči: retrospektivní analýza
Úvod: Cílem retrospektivní analýzy bylo posoudit, zda stávající dávkovací režimy meropenemu vedou k adekvátní expozici meropenemu.
Metody: Do práce byly zahrnuty pouze údolní kon-centrace při prvním měření. Ke zhodnocení dosažení farmakokineticko/farmakodynamického (PK/PD) cíle byly použity MIC patogenů definovaných v European Committee on Antimicrobial Susceptibility testing.
Výsledky: Do studie bylo zahrnuto 83 pacientů. Byla pozorována velká variabilita sérových hladin mero-penemu (medián 34,3 mg/l, rozptyl < 1,0–146,1 mg/l). Nejnižší PK/PD cíl pro citlivé patogeny (100 % T > MIC) byl dosažen u 100 % pacientů na dialýze a kontinuální eliminační metodě (CRRT) a u 91 % pacientů bez eliminační metody. Pro patogeny s intermediární citlivostí 100 % T > MIC bylo dosaženo u všech pacientů na CRRT a 96 % pacientů na dialýze, jen 74 % pacientů bez eliminační metody dosáhlo tento PK/PD cíl. Pacienti na RRT měli vyšší pravděpodobnost dosažení nejvyššího PK/PD cíle 100 % T > 5 × MIC, P < 0,05. Vyšší podíl pacientů na RRT by vyžadoval snížení dávky meropenemu, pokud by byl zvolen horní limit 100 % T > 10 × MIC, P < 0,05.
Závěr: Aplikace standardní dávky meropenemu kriticky nemocným pacientům vede k velké variabilitě hladin. Nastavení dávky pro konkrétního pacienta je důležité pro dosažení adekvátní expozice meropenemu.
Klíčová slova:
meropenem – údolní koncentrace – MIC – farmakokineticko/farmakodynamický cíl
Authors: Jana Uricová; Ivana Kacířová; Hana Brozmanová
Authors place of work: Department of Clinical Pharmacology, Faculty of Medicine, University of Ostrava, Czech Republic
Published in the journal: Čes. slov. Farm., 2020; 69, 230-236
Category: Původní článek
Summary
Objectives: To determine whether currently used meropenem dosages in our hospital provide adequate serum concentrations.
Methods: Trough blood samples taken during the first meropenem concentration monitoring were included. For the evaluation of achievement of the pharmacokinetic/pharmacodynamic (PK/PD) target, MIC of the pathogens defined by the European Committee on Antimicrobial Susceptibility testing was selected.
Results: Eighty three patients were enrolled. A large variability in meropenem trough serum concentrations was observed (median 34.3 mg/L, range < 1.0–146.1 mg/L). The lowest PK/PD target for susceptible pathogens (100% T > MIC) was achieved in 100% of patients on dialysis and continuous renal replacement therapy (CRRT) and in 91% non-RRT patients. For pathogens with intermediate susceptibility, 100% T > MIC was attained in all patients on CRRT and 96% on dialysis, only 74% non-RRT patients achieved this PK/PD target. Patients on RRT were more likely to achieve the highest PK/PD target 100% T > 5 × MIC, P < 0.05. Higher proportion of patients on RRT would also require meropenem dose reduction if upper limit 100% T > 10 × MIC was chosen, P < 0.05.
Conclusions: Administration of a standard meropenem dose to critically ill patients leads to a large concentration variability. Thus, a personalised dosing regimen is crucial for the achievement of adequate meropenem exposure.
Keywords:
meropenem – trough concentrations – MIC – pharmacokinetic/pharmacodynamic target
Introduction
Infection and sepsis are major causes of morbidity and mortality in intensive care patients1, 2). Treatment of critically ill patients with sepsis and septic shock is complex. Early and adequate antibiotic (ATB) therapy is crucial and associated with favourable clinical outcome. The mainstay of successful antibiotic therapy is selection of a right antibiotic and its application in a sufficient dose so that adequate concentration at the site of infection can be reached. Improper dosing of ATB has a negative impact on the patient’s clinical outcome and increases the risk of developing resistance3, 4). A critically ill patient differs in his characteristics from other patients. A rapidly dynamic physiology can cause unpredictable pharmacokinetic alterations that effect antibiotic exposure. Thus antibiotic dosing in critically ill patients might be a challenging and complicated process5, 6). Summary of Product Characteristics (SPC), the basis of dosage regimen information for health professionals, is unfortunately based on studies in non-critically ill patients. The pharmacokinetic parameters reported in these patients may differ from those in critically ill patients, so dosing regimens may not be equally applicable and may result in inadequate dosing5).
Intravenously administered broad-spectrum β-lactam antibiotics belong among the most frequently prescribed antimicrobials in critically ill patients7). When using β-lactam antibiotics in critically ill patients, clinical evaluations suggest maintaining at least 100% fT > MIC may be necessary (100% of time of free antibiotic concentration above minimal inhibitory concentration). This pharmacokinetic/pharmacodynamic (PK/PD) target is associated with better bacterial eradication and therapeutic outcome8). Some authors suggest that maximum killing of bacteria occurs when serum concentrations are maintained above the MIC of the causative pathogen for four to five times the MIC (fT > 4–5 × MIC)9–12). Susceptibility data for confirmed or suspected pathogens are used to establish the MIC target. Where local data are not available or β-lactam antibiotic was introduced on empirical ground, the highest MIC in the susceptible range is selected from breakpoints published by the European Committee on Antimicrobial Susceptibility Testing (EUCAST)13). Effective antibiotic concentrations need to be achieved in the interstitial fluid of tissues as this is the site of most infections5). For infections in which tissue penetration is crucial, it is necessary to ensure an adequate β-lactam antibiotic concentration at the site of infection14, 15). Severe infections can cause vascular dysfunction including microvascular failure, which can impair ATB delivery into body tissues. Furthermore, infections in the ICU are often caused by pathogens with higher MICs compared with other clinical settings16). Thus, higher serum ATB concentrations might be required in intensive care patients to increase the likelihood of effective antibiotic concentrations at the site of infection.
In this respect, therapeutic drug monitoring (TDM) of β-lactam antibiotics has a crucial role in optimising adequate antibiotic concentrations in critically ill patients. TDM of some β-lactam antibiotics has already been implemented in certain laboratories in the Czech Republic. In our hospital, TDM of five β-lactam antibiotics (meropenem, ceftazidime, cefotaxime, cefepime and piperacillin/tazobactam) has been available routinely from January 2019, with meropenem being one of the most commonly measured β-lactam ATB. Meropenem is a carbapenem ATB characterized by a broad antibacterial spectrum used for the treatment of serious infections including hospital-acquired infections (e.g. sepsis, severe pneumonia, complicated intra-abdominal infections). Meropenem is a hydrophilic ATB with very low plasma protein binding (approximately 2%) and it achieves good penetration into a wide range of tissues. Meropenem undergoes primarily renal elimination, extra-renal elimination accounts for 20% up to 50% in conditions of reduced renal function17, 18). A large inter-patient variability in meropenem concentrations after standard dosing has been described in critically ill patients19, 20). Meropenem has been shown to be readily dialyzable and effectively removed by haemodialysis18). A strong relationship between renal function and meropenem exposure and consequently PK/PD target attainment has been observed20). The main objective of this retrospective study was to determine whether the currently used meropenem dosages in our hospital provide adequate serum concentrations and utility of β-lactam TDM in routine clinical practice.
Experimental part
Patient selection
This is a retrospective study performed at the University Hospital Ostrava. TDM of meropenem has been available routinely from January 2019. All patients admitted to any of our intensive care unit between January 2019 and July 2020 were considered for inclusion. Exclusion criteria were: age less than 18 years and admission to another than intensive care unit. Meropenem dosing was at the discretion of the treating physician. Demographic data, including age, sex, height, renal function and presence of renal replacement therapy were collected.
Blood sampling
Blood samples taken during the first meropenem concentration monitoring were included. Only trough serum meropenem concentrations were taken. Meropenem dosing data including the dose, infusion duration, frequency of administration, the time of dosing and sampling, and the day of meropenem therapy were collected. The TDM service is provided in our hospital on daily basis during week days, results are typically available within the next antibiotic dose. Antibiotic dosing is optimised using an iterative process of TDM combined with Bayesian forecasting within dosing software (MWPharm 3.30).
Meropenem serum concentration analysis
Blood samples were processed and stored according to validation protocol to maintain integrity. Total serum meropenem concentrations were determined by liquid chromatography-tandem mass spectrometry method. The sample preparation procedure included precipitation of protein with a mixture composed of methanol : acetonitrile (3 : 2, v/v). The method was validated by the US Food and Drug Administration (FDA) rules as follows: Linearity was found between 1–200 mg/L. The within-day and between-day precision and accuracy were measured at three concentration levels. Coefficients of variation of all tested concentrations were between 1.6–14.9% and recovery 93.1–109.5%, respectively. The method was tested by external quality assurance based on proficiency testing schemes (external quality control EQC Instand Germany) twice a year.
PK/PD data
In most of the patients meropenem was initiated empirically. Therefore, for the evaluation of achievement of PK/PD targets, MIC of the pathogens defined by the EUCAST was selected. The highest MIC for susceptible bacteria to meropenem was chosen: clinical breakpoint for susceptibility MIC 2.0 mg/L, clinical breakpoint for intermediate pathogens MIC 8.0 mg/L. Attainment of PK/PD target 100% T > MIC and 100% T > 5 × MIC was evaluated. The consensus about the upper limit of β-lactam antibiotics serum concentrations has not been reached yet. Some authors arbitrarily selected the PK/PD 100% T > 10 × MIC as an upper limit for β-lactam antibiotics dose reduction, exposure above this PK/PD target is likely of no additional therapeutic value, but there may be an increased risk of toxicity11, 21).
Data and statistical analysis
Continuous data are presented as median with interquartile range, categorical data are reported as numbers and percentages. Achievement of various PK/PD targets between groups of patients with and without renal replacement therapy was compared by the Fisher’s exact test. A value of P < 0.05 was considered statistically significant. Statistical analysis was performed using the GraphPad Prism for Windows version 5.0 (GraphPad Prism Software, Inc).
Results
Patient characteristics
Eighty three intensive care patients were enrolled in this study. For demographic and clinical characteristics see Table 1. Patients were divided into three groups according to their renal function and presence of renal replacement therapy:
- non-RRT patients (patients not on any renal replacement therapy),
- patients on intermittent dialysis either chronic or acute (iHD),
- patients on continuous renal replacement therapy (CRRT).
Tab. 1. Demographics and clinical characteristics of the studied patients 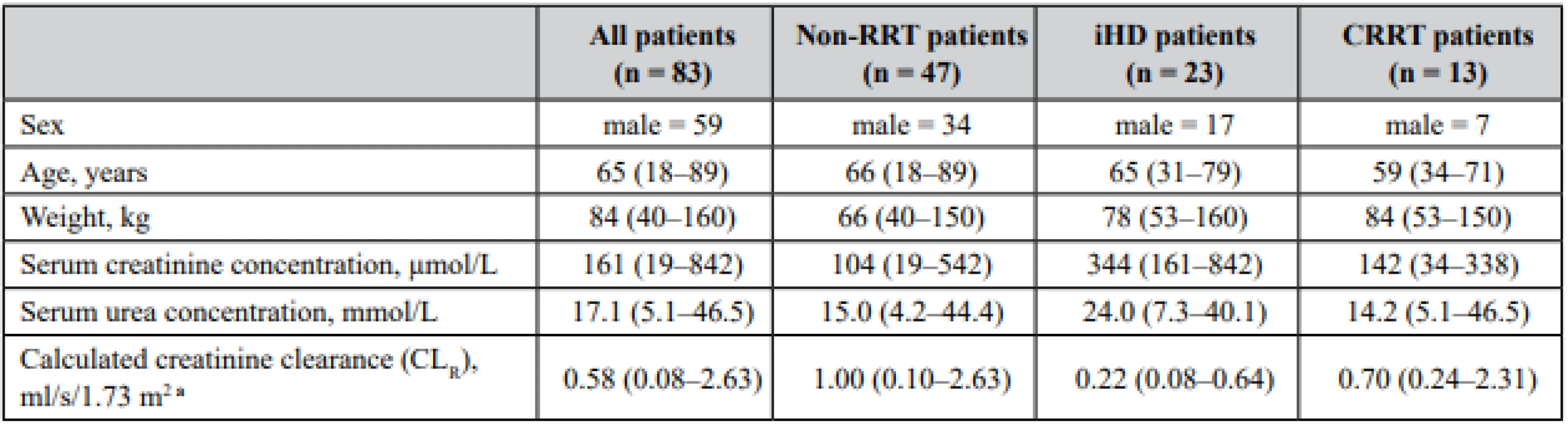
Data are presented as median (interquantile range) unless otherwise specified: non-RRT patients – patients without renal replacement therapy, iHD – intermittent dialysis, CRRT – continuous renal replacement therapy aCLR was estimated using formula CKD-EPI 2009 Meropenem was mostly used for the treatment of sepsis, the most common source of infection were the lungs (30.7%), followed by the urinary tract (14.9%), gastrointestinal tract (10%), skin and soft tissue (5.8%) and in 10% the source of infection was not specified.
PK/PD data
The data describing the meropenem dose, meropenem serum trough concentrations and the achievement of PK/PD targets are depicted in Table 2. A large variability in meropenem trough serum concentrations was observed in each patient group. Even when only meropenem serum concentrations at the same daily dose (6 g daily, which was the most common administered dose) were chosen, the variability was still considerable.
Tab. 2. Meropenem (MER) dosage and trough serum concentrations and achievement of pharmacokinetic/pharmacodynamic targetsa 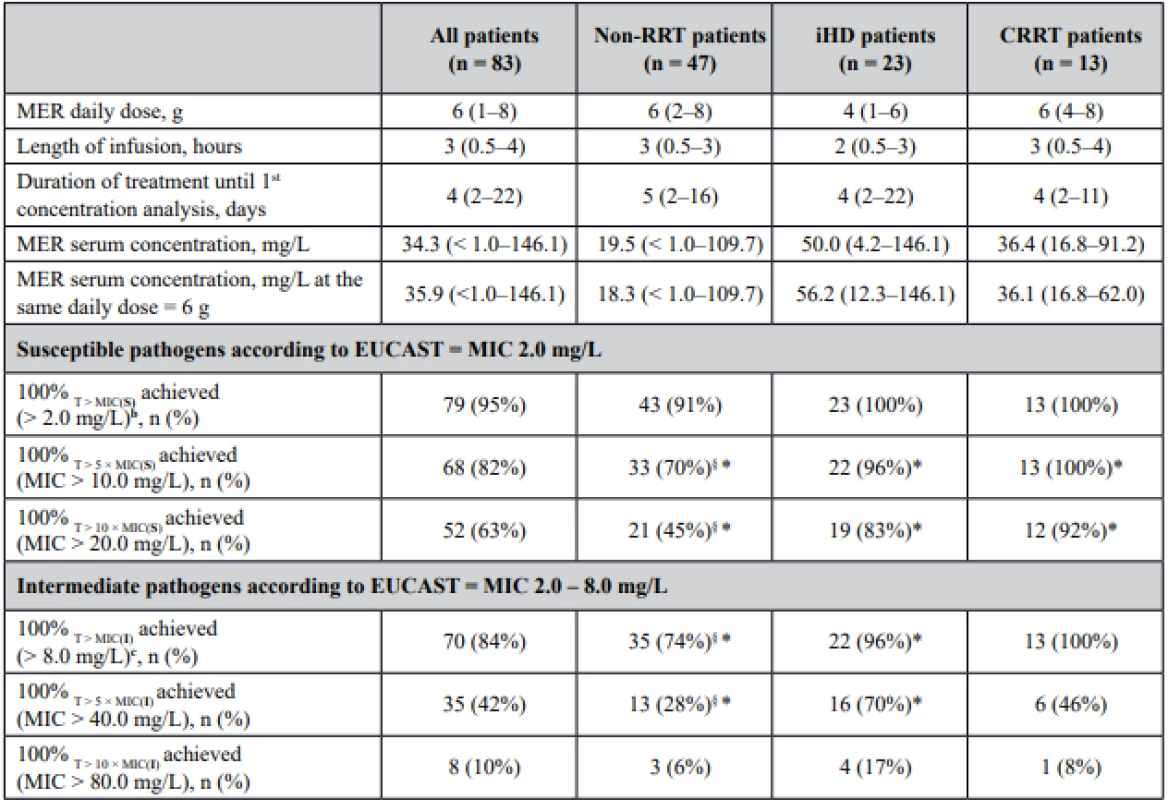
amedian (interquartile range)
b MIC (S) ≤ 2.0 mg/L – denotes the highest MIC in the susceptible (S) range for applicable pathogens, such as Pseudomonas spp., Enterobacterales, anaerobes
c MIC (I) ≤ 8.0 mg/L – denotes the highest MIC in the intermediate (I) range for applicable pathogens, such as Pseudomonas spp., Enterobacterales, anaerobes
§ statistical analysis between non-RRT group and RRT group (iHD + CRRT patients), P < 0.05
*statistical analysis between non-RRT group and iHD, non-RRT group and CRRT, P < 0.05
RRT – renal replacement therapy, iHD – intermittent dialysis, CRRT – continuous renal replacement therapy, MIC – minimal inhibitory concentrationWhen aiming at the lowest PK/PD target for susceptible pathogens (100% T > MIC, MIC = 2.0 mg/L), 100% of patients on iHD and CRRT achieved this target, in patients without any renal replacement therapy the achievement of the target was a little bit lower. For pathogens with intermediate susceptibility, 100% T > MIC for the highest MIC in this range (MIC 8.0 mg/L) was attained in all patients on CRRT and slightly less in patients on iHD. In the group of patients without any renal replacement therapy, 74% of patients achieved this PK/PD target. Patients on renal replacement therapy were more likely to achieve the highest PK/PD target 100% T > 5 × MIC for both MIC values (MIC 2.0 mg/L and 8.0 mg/L), P < 0.05. Higher proportion of patients on RRT would also require meropenem dose reduction if upper limit 100% T > 10 × MIC (MIC 2.0 mg/L) was chosen, P < 0.05.
Discussion
Critically ill patients exhibit extreme inter-individual variability in pharmacokinetic parameters, making decision about the right antibiotic dose difficult. Traditionally, TDM of antibiotics has been employed to minimise toxicity of glycopeptides and aminoglycosides. However, with an increasing number of less sensitive pathogens, large pharmacokinetic variability and increasing intensive care possibilities, TDM concern of these antibiotics has also spread to ensure adequate therapeutic concentrations. With improving understanding of altered pharmacokinetics in critically ill patients, the TDM of antibiotics has also expanded to β-lactams11, 21). Insufficient β-lactam concentrations have been observed in the early phase of severe sepsis and septic shock under standard antibiotic doses22). TDM of β-lactam antibiotics has been available in our hospital from early 2019. The results of our study retrospectively describe meropenem serum concentrations taken at first TDM sampling in our intensive care patients. Several studies have addressed meropenem pharmacokinetics in ICU septic patients, a large heterogenity of PK parameters was found19, 23, 24). In accordance with those findings we observed a large variability in meropenem trough serum concentrations in our intensive care patients, even for the same daily dose. Higher proportion of patients on renal replacement therapy (intermittent or continuous) achieved the PK/PD target compared to patients without renal replacement therapy. Roberts JA et al. evaluated practicality and utility of β-lactam TDM in their prospective study in critical care unit. According to TDM meropenem dose adjustment was required in 84% of patients with 57% of patients requiring dose increases and 27% of patients requiring dose decreased after the first TDM. A PK/PD target of 100% fT > 4–5 × MIC and 100% fT < 10 × MIC was arbitrarily chosen to maximise the likelihood of clinical cure and minimize the toxicity11). When choosing the same PK/PD targets, dose adjustment would be required in 81.9% of our patients for MIC 2.0 mg/L, a finding similar to a previous study. However, only 18.1% of patients would require dose increases and on the contrary up to 63.9% of patients would require dose decreases. In a fear of under-dosing and to maximize meropenem exposure, most of our patients received the maximal recommended meropenem dose 6 g daily, without any dose adjustment according to renal functions compared to 3 g daily in normal renal function in the previous study. When adjusting meropenem PK/PD target for MIC 8.0 mg/L, dose adjustment would be required in 67.4% of patients (of them 57.8% of patients requiring dose increases and 9.6% dose decreases). However, there is still unclarity regarding the best PK/PD target for β-lactam antibiotics TDM11, 25, 26). Even, when the PK/PD target was set at fT > 50% MIC, 16% of intensive care patients did not achieve this most conservative target and these patients were 32% less likely to have a positive clinical outcome25). Critically ill patients have larger volume of distribution, varying renal functions (either decreased or augmented), have low albumin concentrations and may require some artificial organ support27). However, dosing regimens for antibiotics are generally based on studies in healthy volunteers or patients not critically ill. Application of these regimens to critically ill patient may lead to sub-therapeutic concentrations and clinical failure28–30). A great deal of work has been published on the pharmacokinetics of β-lactam antibiotics in intensive care patients23, 24, 31, 32). However, due to great inter-individual variability as a result of multiple variables, extrapolation of these data cannot be applicable to every patient. Thus implementation of TDM of β-lactam antibiotics in routine clinical practice seems crucial for individualization of the optimal dose in a critical care patient.
β-lactam antibiotics exert its efficacy mostly in tissues. Although it has been commonly believed that most antibiotics nearly achieve equilibrium in tissues and plasma, recent studies have indicated that the antibiotic distribution process is characterized by high intertissue and intersubject variabilities and that target site drug levels may substantially differ from corresponding plasma drug levels. Suboptimal target site concentrations may have important clinical implications, as they may explain therapeutic failure and may also trigger bacterial resistance14, 23, 33). However, measurement of β-lactam concentrations at the site of infection is not performed routinely and is reserved mainly for clinical studies. Choosing the “aggressive” PK/PD target (T > 4–5 × MIC) one might reduce the risk of tissue underexposure. Prolonged or continuous infusions of β-lactams have consistently been shown to improve PK/PD target attainment, particularly in patients with severe infections given the increasing resistance rate34, 35). Some studies have suggested a clinical benefit of prolonged/continuous infusions versus intermittent bolus administrations35–38). On the other hand, high β-lactam concentrations may lead to a higher risk of toxicity, especially neurotoxicity. The most vulnerable group of patients are those with renal dysfunction. However, the toxic concentration threshold is yet unknown. The PK/PD 100% T > 8–10 × MIC as an upper limit for β-lactam antibiotics dose reduction has been chosen arbitrarily by some authors11, 21, 26). Some studies have focused on the concentration–neurotoxicity relationship of β-lactams in ICU patients with the aim to define thresholds above which toxicity is more likely39, 40).
There are limitations of our study. First, this is a retrospective observation of meropenem serum concentrations at the first TDM occasion, this study was not designed to evaluate clinical outcome of the patients or concentration-related toxicity. Second, blood samples were taken at the discretion of treating physician, meropenem serum concentrations in some patients might not be in the steady-state. Third, PK/PD targets were based on the highest MIC in the susceptibility range from EUCAST, which might be higher than those of isolated pathogens. However, in most of the patients, meropenem was initiated empirically, with further de-escalation according to cultivations. Using these highest MIC values we elected the PK/PD target that would maximise meropenem exposure in intensive care patients given the non-standard pharmacokinetics and occurrence of less sensitive pathogens in these patients. Fourth, only total serum meropenem concentrations were analysed. By measuring the unbound concentrations it is more likely to have similar concentrations to those present at the site of infection. However, meropenem binding to serum albumin is negligible (about 2%), so we do not assume a significant difference between total and unbound meropenem serum concentration17).
Conclusion
In conclusion, we have observed that meropenem trough serum concentrations vary widely in critically ill patients using the same dose. Administration of a standard β-lactam antibiotic dose to a critically ill patient may not lead to adequate exposure. Thus, a personalised dosing regimen is crucial for achievement of beneficial clinical outcome. Implementation of TDM of β-lactams in routine clinical practice is a useful tool to optimise β-lactam dosing in critically ill patients. Ideally, TDM of β-lactams should be performed within the first 2–3 days after antibiotic initiation, so that early dose adjustment could be performed to ensure rapid achievement of adequate antibiotic exposure. As intraindividual β-lactam concentration may vary over time in critically ill patients, repeated therapeutic drug monitoring may be required until the patient is stable.
Conflict of interest: none.
PharmDr. Jana Ďuricová, PhD. (∗) • I. Kacířová • H. Brozmanová
Department of Clinical Pharmacology, Department of Laboratory
Diagnostics, University Hospital Ostrava, Czech Republic
17. listopadu 1790/5, 708 52 Ostrava-Poruba
e-mail: jankaduricova@seznam.cz
Department of Clinical Pharmacology, Faculty of Medicine, University of Ostrava, Czech Republic
Zdroje
1. Vincent J. L., Rello J., Marshall J., Silva E., Anzueto A., Martin C. D., Moreno R., Lipman J., Gomersall C., Sakr Y., Reinhart K. International study of the prevalence and outcomes of infection in intensitve care units. JAMA 2009; 302, 2323–2329.
2. Kula R., Sklienka P., Chýlek V., Sturz P. Současné trendy antibiotické terapie v léčbě těžké sepse. Klin. Farmakol. Farm. 2007; 21, 74–78.
3. Burgess D. S. Pharmacodynamic principles of antimicrobial therapy in the prevention of resistance. Chest 1999; 115, 19S–23S.
4. Roberts J. A., Kruger P., Paterson D. L., Lipman J. Antibiotic resistance – what’s dosing got to do with it? Crit. Care Med. 2008; 36, 2433–2440.
5. Roberts J. A., Abdul-Aziz M. H., Lipman J., Mouton J. W., Vinks A. A., Felton T. W., Hope W. W., Farkas A., Neely M. N., Schentag J. J., Drusano G., Frey O. R., Theuretzbacher U., Kuti J. L. Individualised antibiotic dosing for patients who are critically ill: challenges and potential solutions. Lancet Infect. Dis. 2014; 14, 498–509.
6. Blot S. I., Pea F., Lipman J. The effect of pathophysiology on pharmacokinetics in the critically ill patient — Concepts appraised by the example of antimicrobial agents. Adv. Drug Deliv. Rev. 2014; 77, 3–11.
7. Evans L. E., Alhazzani W., Levy M. M., Antonelli M., Ferrer R., Kumar A., Sevransky J. E., Sprung C. L., Nunnally M. E., Rochwerg B., Rubenfeld G. D., Angus D. C., Annane D., Beale R. J., Bellinghan G. J., Bernard G. R., Chiche J. D., Coopersmith C., de Backer D. P., French C. J., Fujishima S., Gerlach H., Hidalgo J. L., Hollenberg S. M., Jones A. E., Karnad D. R., Kleinpell R. M., Koh Y., Lisboa T. C., Machado F. R., Marini J. J., Marshall J. C., Mazuski J. E., McIntyre L. A., McLean A. S., Mehta S., Moreno R. P., Myburgh J., Navalesi P., Nishida O., Osborn T. M., Perner A., Plunkett C. M., Ranieri M., Schorr C. A., Seckel M. A., Seymour C. W., Shieh L., Shukri K. A., Simpson S. Q., Singer M., Thompson B. T., Townsend S. R., van der Poll T., Vincent J. L., Wiersinga W. J., Zimmerman J. L., Dellinger R. P. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017; 43, 304–377.
8. McKinnon P. S., Paladino J. A., Schentag J. J. Evaluation of area under the inhibitory curve (AUIC) and time above the minimum inhibitory concentration (T>MIC) as predictors of outcome for cefepime and ceftazidime in serious bacterial infections. Int. J. Antimicrob. Agents 2008; 31, 345–351.
9. Mouton J. W., Vinks A. A. Is continuous infusion of beta-lactam antibiotics worth while? Efficacy and pharmacokinetic considerations. J. Antimicrob. Chemother. 1996; 38, 5–15.
10. Sinnollareddy M. G., Roberts M. S., Lipman J., Roberts J. A. β-lactam pharmacokinetics and pharmacodynamics in critically ill patiens and strategies for dose optimization: A structured review. Clin. Exp. Pharmacol. Physiol. 2012; 39, 489–496.
11. Roberts J. A., Ulldemolins M., Roberts M. S., McWhinney B., Ungerer J., Paterson D. L., Lipman J. Therapeutic drug monitoring of β-lactams in critically ill patients: proof of concept. Int. J. Antimicrob. Agents 2010; 36, 332–339.
12. Li Ch., Du X., Kuti J. L., Nicolau D. P. Clinical pharmacodynamics of meropenem in patients with lower respiratory tract infections. Antimicrob. Agents Chemother. 2007; 51, 1725–1730.
13. The European Committee on Antimicrobial Susceptibility Testing https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_10.0_Breakpoint_Tables.pdf
14. Heffernan A. J., Sime F. B., Lipman J., Dhanani J., Andrews K., Ellwood D., Grimwood K., Roberts J. A. Intrapulmonary pharmacokinetics of antibiotics used to treat nosocomial pneumonia caused by Gram-negative bacilli: A systematic review. Int. J. Antimicrob. Agents 2019; 53, 234–245.
15. Lonsdale D. O., Udy A. A., Roberts J. A., Lipman J. Antibacterial therapeutic drug monitoring in cerebrospinal fluid: difficulty in achieving adequate drug concentrations. J. Neurosurg. 2013; 118, 297–301.
16. Sumi C. D., Heffernan A. J., Lipman J., Roberts J. A., Sime F. B. What Antibiotic Exposures Are Required to Suppress the Emergence of Resistance for Gram-Negative Bacteria? A Systematic Review. Clin. Pharmacokinet. 2019; 58 : 1407–1443.
17. Baldwin C. M., Lyseng-Williamson K. A., Keam S. J. Meropenem: a review of its use in the treatment of serious bacterial infections. Drugs 2008; 68, 803–838.
18. Christensson B. A., Nilsson-Ehle I., Hutchison M., Haworth S. J., Oqvist B., Norrby S. R. Pharmacokinetics of meropenem in subjects with various degrees of renal impairment. Antimicrob. Agents. Chemother. 1992; 36, 1532–1537.
19. Mattioli F., Fucile C., del Bono V., Marini V., Parisini A., Molin A., Zuccoli M. L., Milano G., Danesi R., Marchese A., Polillo M., Viscoli C., Pelosi P., Martelli A., di Paolo A. Population pharmacokinetics and probability of target attainment of meropenem in critically ill patients. Eur. J. Clin. Pharmacol. 2016; 72, 839–848.
20. Ehmann L., Zoller M., Minichmayr I. K., Scharf C., Maier B., Schmitt M. V., Hartung N., Huisinga W., Vogeser M., Frey L., Zander J., Kloft C. Role of renal function in risk assessment of target non-attainment after standard dosing of meropenem in critically ill patients: a prospective observational study. Crit. Care 2017; 21, 263.
21. Wong G., Briscoe S., McWhinney B., Ally M., Ungerer J., Lipman J., Roberts J. A. Therapeutic drug monitoring of β-lactam antibiotics in the critically ill: direct measurement of unbound drug concentrations to achieve appropriate drug exposures. J. Antimicrob. Chemother. 2018; 73, 3087–3094.
22. Taccone F. S., Laterre P. F., Dugernier T., Spapen H., Delattre I., Wittebole X., de Backer D., Layeux B., Wallemacq P., Vincent J. L., Jacobs F. Insufficient β-lactam concentrations in the early phase of severe sepsis and septic shock. Crit. Care 2010; 14, R126.
23. Gonçalves-Pereira J., Póvoa P. Antibiotics in critically ill patients: a systematic review of the pharmacokinetics of β-lactams. Crit. Care 2011; 15, R206.
24. Sime F. B., Roberts M. S., Peake S. L., Lipman J., Roberts J. A. Does Beta-lactam Pharmacokinetic Variability in Critically Ill Patients Justify Therapeutic Drug Monitoring? A Systematic Review. Ann. Intensive Care 2012; 2, 35.
25. Roberts J. A., Paul S. K., Akova M., Bassetti M., de Waele J. J., Dimopoulos G., Kaukonen K. M., Koulenti D., Martin C., Montravers P., Rello J., Rhodes A., Starr T., Wallis S. C., Lipman J. DALI: defining antibiotic levels in intensive care unit patients: are current β-lactam antibiotic doses sufficient for critically ill patients? Clin. Infect. Dis. 2014; 58, 1072–1083.
26. Guilhaumou R., Benaboud S., Bennis Y., Dahyot-Fizelier C., Dailly E., Gandia P., Goutelle S., Lefeuvre S., Mongardon N., Roger C., Scala-Bertola J., Lemaitre F., Garnier M. Optimization of the treatment with beta-lactam antibiotics in critically ill patients-guidelines from the French Society of Pharmacology and Therapeutics (Société Française de Pharmacologie et Thérapeutique-SFPT) and the French Society of Anaesthesia and Intensive Care Medicine (Société Française d’Anesthésie et Réanimation-SFAR). Crit. Care 2019; 23, 104.
27. Roberts J. A., Joynt G. M., Choi G. Y., Gomersall C. D., Lipman J. How to optimise antimicrobial prescriptions in the Intensive Care Unit: principles of individualised dosing using pharmacokinetics and pharmacodynamics. Int. J. Antimicrob. Agents 2012; 39, 187–192.
28. Udy A. A., Varghese J. M., Altukroni M., Briscoe S., McWhinney B. C., Ungerer J. P., Lipman J., Roberts J. A. Subtherapeutic initial β-lactam concentrations in select critically ill patients: association between augmented renal clearance and low trough drug concentrations. Chest 2012; 142, 30–39.
29. Hayashi Y., Lipman J., Udy A. A., Ng M., McWhinney B., Ungerer J., Lust K., Roberts J. A. β-Lactam therapeutic drug monitoring in the critically ill: optimising drug exposure in patients with fluctuating renal function and hypoalbuminaemia. Int. J. Antimicrob. Agents 2013; 41, 162–166.
30. Sadilová Z., Halačová M., Černý D. Pharmacokinetic aspects of beta-lactam antibiotic therapy in intensive care unit patients: A one-center experience with TDM. Čes. slov. Farm. 2020; 69, 17–23.
31. Ulldemolins M., Vaquer S., Llauradó-Serra M., Pontes C., Calvo G., Soy D., Martín-Loeches I. Beta-lactam dosing in critically ill patients with septic shock and continuous renal replacement therapy. Crit. Care 2014; 18, 227.
32. Hahn J., Choi J. H., Chang M. J. Pharmacokinetic changes of antibiotic, antiviral, antituberculosis and antifungal agents during extracorporeal membrane oxygenation in critically ill adult patiens. J. Clin. Pharm. Ther. 2017; 42, 661–671.
33. Müller M., de la Peña A., Derendorf H. Issues in pharmacokinetics and pharmacodynamics of anti-infective agents: distribution in tissue. Antimicrob. Agents Chemother. 2004; 48, 1441–1453.
34. Roberts J. A., Kirkpatrick C. M., Roberts M. S., Robertson T. A., Dalley A. J., Lipman J. Meropenem dosing in critically ill patients with sepsis and without renal dysfunction: intermittent bolus versus continuous administration? Monte Carlo dosing simulations and subcutaneous tissue distribution. J. Antimicrob. Chemother. 2009; 64, 142–150.
35. Abdul-Aziz M. H., Lipman J., Akova M., Bassetti M., de Waele J. J., Dimopoulos G., Dulhunty J., Kaukonen K. M., Koulenti D., Martin C., Montravers P., Rello J., Rhodes A., Starr T., Wallis S. C., Roberts J. A. Is prolonged infusion of piperacillin/tazobactam and meropenem in critically ill patients associated with improved pharmacokinetic/ pharmacodynamic and patient outcomes? An observation from the Defining Antibiotic Levels in Intensive care unit patients (DALI) cohort. J. Antimicrob. Chemother. 2016; 71, 196–207.
36. Lorente L., Lorenzo L., Martín M. M., Jiménez A., Mora M. L. Meropenem by continuous versus intermittent infusion in ventilator-associated pneumonia due to gram-negative bacilli. Ann. Pharmacother. 2006; 40, 219–223.
37. Roberts J. A., Abdul-Aziz M. H., Davis J. S., Dulhunty J. M., Cotta M. O., Myburgh J., Bellomo R., Lipman J. Continuous versus Intermittent b-Lactam Infusion in Severe Sepsis. A Meta-analysis of Individual Patient Data from Randomized Trials. Am. J. Respir. Crit. Care Med. 2016; 194, 681–691.
38. Vardakas K. Z., Voulgaris G. L., Maliaros A., Samonis G., Falagas M. E. Prolonged versus short-term intravenous infusion of antipseudomonal β-lactams for patients with sepsis: a systematic review and meta-analysis of randomised trials. Lancet Infect. Dis. 2018; 18, 108–120.
39. Imani S., Buscher H., Marriott D., Gentili S., Sandaradura I. Too much of a good thing: a retrospective study of β-lactam concentration-toxicity relationships. J. Antimicrob. Chemother. 2017; 72, 2891–2897.
40. Quinton M. C., Bodeau S., Kontar L., Zerbib Y., Maizel J., Slama M., Masmoudi K., Lemaire-Hurtel A. S., Bennis Y. Neurotoxic Concentration of Piperacillin during Continuous Infusion in Critically Ill Patients. Antimicrob. Agents Chemother. 2017; 61 e00654–17.
Štítky
Farmacie Farmakologie
Článek vyšel v časopiseČeská a slovenská farmacie
Nejčtenější tento týden
2020 Číslo 5-6- Psilocybin je v Česku od 1. ledna 2026 schválený. Co to znamená v praxi?
- Ukažte mi, jak kašlete, a já vám řeknu, co vám je
- Přerušovaný půst může mít významná zdravotní rizika
-
Všechny články tohoto čísla
- Pohľad na perchlozón, antituberkulotikum z Ruskej federácie
- Vývoj a validace metody HPLC pro kvantifikaci nečistot degradace salbutamol-sulfátu s následujícími dlouhodobými stabilitními testy ve vícesložkovém sirupu proti kašli
- Automatizovaná příprava radiofarmak jako nástroj optimalizace radiační ochrany personálu
- Hepatoprotektivní účinek kombinace glucosaminových derivativů s quercetinem proti methotrexátem indukovanému toxickému poškození jater
- Sérové koncentrace meropenemu u pacientů vyžadujících intenzivní péči: retrospektivní analýza
- Stanovenie tiamínu a pyridoxínu vo výživových doplnkoch a nápojoch jednoduchou metódou kapilárnej zónovej elektroforézy v spojení s UV detekciou
- K životnímu jubileu prof. RNDr. Jozefa Csölleiho, CSc.
- Česká a slovenská farmacie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Sérové koncentrace meropenemu u pacientů vyžadujících intenzivní péči: retrospektivní analýza
- K životnímu jubileu prof. RNDr. Jozefa Csölleiho, CSc.
- Automatizovaná příprava radiofarmak jako nástroj optimalizace radiační ochrany personálu
- Stanovenie tiamínu a pyridoxínu vo výživových doplnkoch a nápojoch jednoduchou metódou kapilárnej zónovej elektroforézy v spojení s UV detekciou
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



