-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Effective Tissue Layer Separation of Donor Cornea for DMEK by Fluid Injection Between Descemet
Authors: P. Stodůlka 1,2; M. Horáčková 1; M. Šramka 1,3; M. Slovák 1
Authors place of work: Gemini oční klinika, Zlín, Česká republika 1; Oftalmologická klinika, 3. lékařské fakulty, Univerzita Karlova v Praze 2; Katedra teorie obvodů, Fakulta elektrotechnická, České vysoké učení technické v Praze 3
Published in the journal: Čes. a slov. Oftal., 75, 2019, No. 1, p. 32-37
Category: Původní práce
doi: https://doi.org/10.31348/2019/1/4Summary
Aim: To evaluate clinical outcomes of Descemet Membrane Endothelial Keratoplasty (DMEK) using a novel hydro-separation of donor Descemet membrane (DM) endothelial graft with 6 months follow up.
Material and methods: In this retrospective single-center; single surgeon cohort study, 16 consecutive eyes underwent DMEK or combined DMEK and cataract surgery with DMEK donor graft hydro-separation. The hydroseparation method used Ringer’s solution injected by a syringe with a flat end cannula between DM and corneal stroma to separate DMEK graft from donors’ cornea. Endothelial cell count, corrected and uncorrected distance visual acuities (CDVA, UDVA), corneal astigmatism and keratometric values were evaluated before and 6 months after the surgery.
Results: DMEK grafts were successfully hydro separated, without a membrane tear in all cases and successful adhesion to recipient corneas was achieved in all cases. Overall CDVA improved significantly (p < 0.0001) from preop. 0.69 logMAR (0.45–0.92, 95%CI) to 0.1 logMAR (0.04–0.15) 6 months postop. However UDVA was slightly worse in cataract combined cases 0.38 logMAR (0.18–0.43) vs 0.3 logMAR (0.14–0.61) in DMEK cases at 6 months. Corneal astigmatism K2-K1 improved significantly (p=0.0137) from preop. mean 2.89 D (0.19–0.43) to 0.98 D (0.48–1.48) 6 months postop.
Conclusion: Hydro-separation is fast and effective donor graft preparation method for DMEK. The DMEK using donor graft hydro-separation results in improved vision and induces a low amount of astigmatism at 6 months follow up. Further clinical data are needed to confirm the success rate and to explore optimization of the selection of IOLs in combined cataract cases.
Keywords:
DMEK – transplantation cornea – Endothelium – Descemet membrane – hydro-separation
INTRODUCTION
Descemet Membrane Endothelial Keratoplasty (DMEK), in which only a thin Descemet Membrane (DM) with an endothelial layer is transplanted, is becoming increasingly popular as a surgical solution for Fuchs’ dystrophy and bullous keratopathy [8], and is considered the method of choice in these indications. Separation of the DMEK graft is usually performed by careful scraping and peeling of the DM from the donor cornea [1, 6]. This process is time consuming, requires a high degree of dexterity and entails a considerable risk of tearing of the membrane [2, 8, 15]. Recently new methods have appeared, in which the separation of the DM is achieved with the aid of air/fluid injected between the DM and corneal stroma [9, 13, 14, 16]. Direct hydro-separation with the aid of a needle inserted between the DM and the corneal stroma is a fast and simple method, which minimises the risk of tearing of the membrane [11, 16]. In addition, during the course of separation with the aid of fluid, it is simultaneously possible to stain only the DM on the side of the corneal stroma, thereby minimising the chemical effect of trypan blue on the sensitive endothelial cells [19]. The separated DM is transplanted and its adhesion to the recipient’s cornea is supported by an air bubble in the anterior chamber.
This study deals with the separation of a DMEK graft by means of the injection of fluid (hydro-separation) between the DM and corneal stroma with a Dua’s layer [3]. The aim is to provide the first clinical results of this new technique performed on two groups of patients – DMEK and combined procedure of DMEK with cataract surgery.
MATERIALS AND METHODS
Consent and ethical commission: Consent to the performance of the procedure was obtained from all the patients before the procedure, and the ethical commission granted its approval for a retrospective analysis of the anonymised data.
Donor tissue: Donor corneas were obtained from the tissue bank of the National Tissue and Cell Centre (Brno, Czech Republic) and the Královské Vinohrady University Hospital (Prague, Czech Republic).
Design of study: Retrospective analysis of DMEK and combined cataract surgery with DMEK in a series of consecutive operations on patients operated on in a single centre in the period from November 2014 to February 2016. The separations and transplantations of the membrane were performed by a single surgeon (PS) at the Gemini Eye Clinic (Zlín, Czech Republic) during the course of a single visit. The sex representation of the patients was six men and nine women, with an age of 67 ± 7.7 years (average ± standard deviation). In eight cases the procedure concerned DMEK only, and in eight cases a combined procedure of DMEK together with cataract surgery.
Preparation of membrane for DMEK: The donor cornea is extracted, rinsed with Ringer’s solution and affixed to a suction pad. An artificial anterior chamber need not be used. A tunnel is created using a semi-blunt spatula inserted beneath the DM from the outside from the trabecula (Fig. 1) in such a manner as to ensure that the spatula is being moved forwards and at the same time slightly raises the DM. A flat end cannula is then inserted into this tunnel (e.g. cat. number 585155, BVI Beaver-Visitec International, Waltham, MA, USA), affixed to a syringe filled with Ringer’s solution (Fig. 2). A pointed needle need not be used, because the cannula is inserted directly between the DM and the corneal stroma. The mouth of the cannula may remain on the side, or may be inserted closer to the centre of the cornea. Its precise position will not have an influence on the size of the bubble, because this is generated between the corneal stroma and the DM layer, and not within the DM layer as such. It is important for the tunnel to be sufficiently long and tight so that the majority of the injected fluid flows into the space between the DM and the corneal stroma. Upon injection the cannula is maintained in the same position or is carefully shifted beneath the DM in the direction towards the centre of the cornea. Ringer’s solution is used, which creates a space beneath the DM in front of the needle. When the fluid reaches a certain pressure, a separation of the DM from the corneal stroma with the Dua’s layer takes place (Fig. 3), and a large convex bubble is formed [19]. The following links demonstrate the technique of the method and performance of the operation: https://youtu.be/Mr5DynQT1Ow and https://youtu.be/2GRNiIiCrWs. The fluid beneath the membrane is drained using the same cannula by which it was injected, and the DMEK graft is prepared for trepanning to the required diameter. The cornea is placed into a concave holder with the endothelial side upwards, and is then trimmed by a trephine of an average of 8.5 to 9.0 mm. The hydro-separated DM with endothelium is lightly detached using mantis point tweezers and placed in a bowl with Ringer’s solution. The membrane has a tendency to roll outwards from the endothelial side. After staining with trypan blue it is prepared for transplantation.
Fig. 1. Opening created beneath DM before separation with the aid of a liquid bubble 
Fig. 2. Needle is inserted into opening beneath DM in such a manner as to separate DM from corneal stroma and Dua’s layer 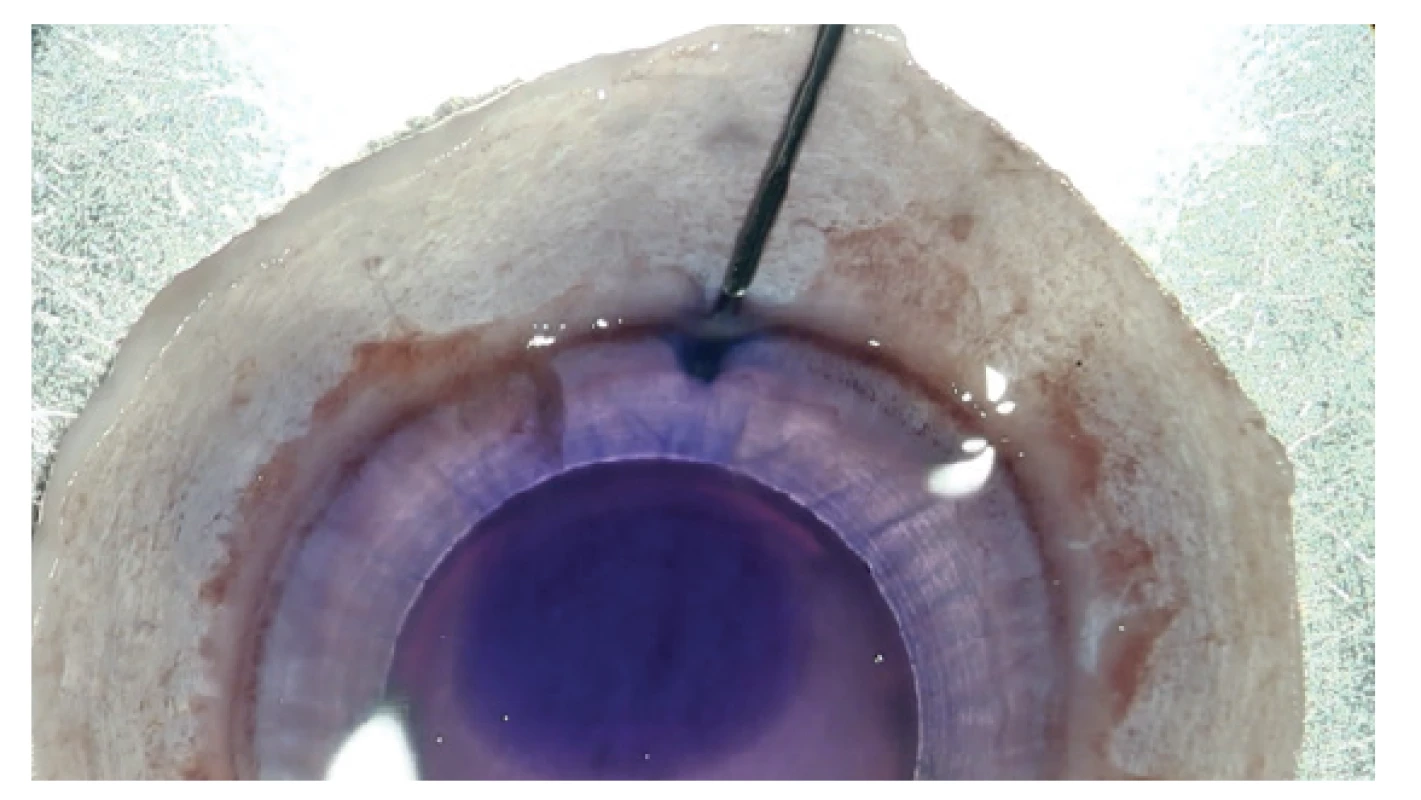
Fig. 3. Convex DM separation by liquid bubble. Air bubble and trypan blue for staining of DM 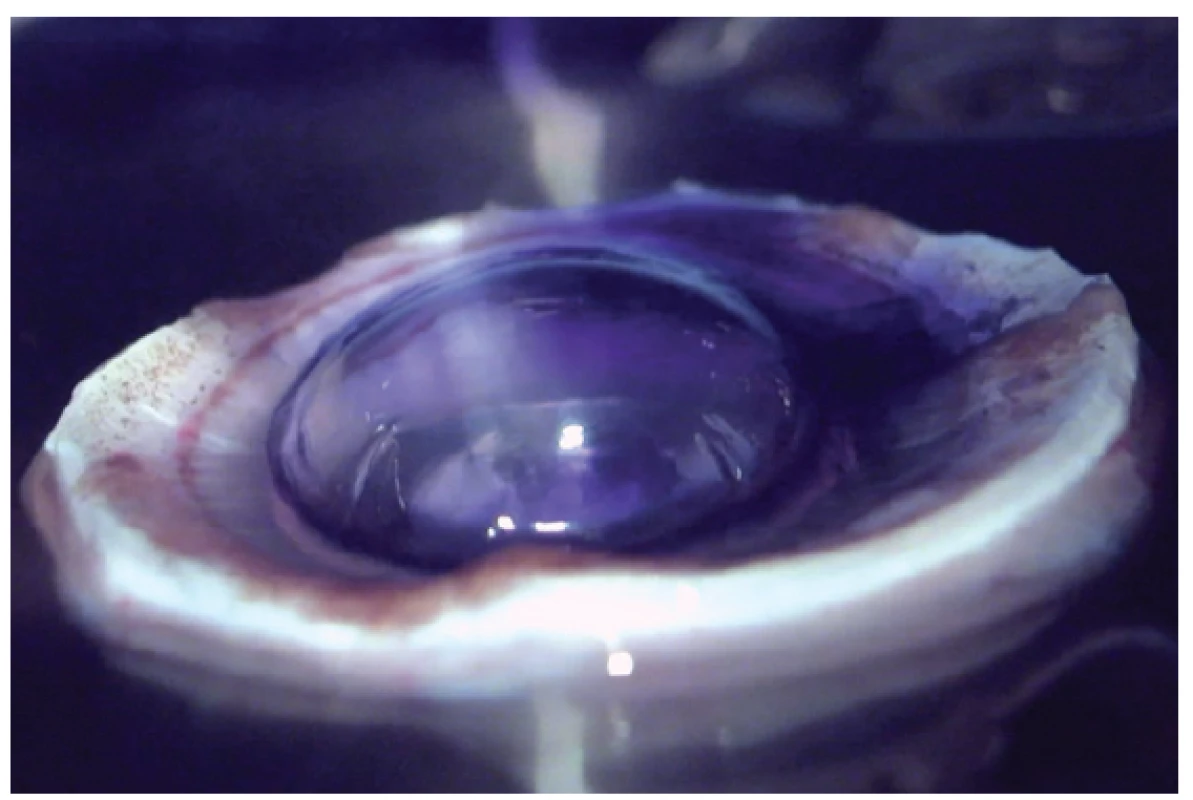
Cataract surgery with implantation of intraocular lens: In some cases DMEK was combined with cataract surgery. Cataract surgery was performed by means of a 2.2 mm wide incision using the Stellaris PC instrument (Bausch and Lomb, Bridgewater, NJ, USA). No viscous material (OVD) was used intraocularly during the operation. Both continual circular capsulorhexis (CCC) and implantation of the artificial intraocular lens were performed upon stabilisation of the eye by an irrigation cannula inserted by means of an auxiliary corneal incision. A monofocal hydrophilic intraocular lens was implanted in the lens capsule.
Transplantation of DM: The DM of the recipient is removed by scraping beneath the infusion of air into the anterior chamber under pressure of 40 mmHg. The DMEK graft is placed in the anterior chamber through the main incision of 2.2 mm by means of a straight glass pipette (DORC International BV, Netherlands) and unrolled with the endothelium downwards, if possible by noncontact method. The anterior chamber is partially filled with an air bubble so that in the position with the face upwards the bubble presses the graft onto the corneal stroma.
Postoperative care: After surgery, combined anti-inflammatory and antibiotic drops were prescribed (Tobradex, Alcon), and the patients received instructions to keep the head in a position with the face upwards for a period of three days until the absorption of the air bubble, and were asked to attend a follow-up examination one week after the procedure. Anterior chamber optical coherence tomography (AC-OCT) on the Casia SS-1000 instrument (Tomey, Japan) was performed in order to display attachment of the DM. If complete adhesion of the DM had not taken place, the generation of an air bubble in the anterior chamber of the eye was repeated.
Evaluation: The average number of cell density of the central endothelium was evaluated using a specular microscope EM-3000 (Tomey, Japan) on the eye of the recipient in the number of cells per mm2. Uncorrected and corrected distance visual acuity (UDVA, CDVA) in logMAR, subjective refraction (SR) converted to spherical equivalent, subjective astigmatism (SA) and corneal astigmatism expressed as the difference of K2-K1, were measured on Tonoref II (Nidek Ltd., Japan) before transplantation, in the first week, first, third and sixth month after surgery. For the sake of simplification this data is presented only before transplantation and six months after transplantation.
Statistical method: Unless stated otherwise, all the values are stated as the average and 95% interval of reliability. Since the analysed data is not from a normal-Gaussian distribution, a non-parametric Wilcoxon text was used for evaluation of the data before surgery and 6 months after surgery. The number of cells of the endothelium and UDVA in the sixth month were compared with the aid of a Mann-Whitney test.
RESULTS
Shortly before transplantation, hydro-separation of the DMEK graft to the periphery was performed in the operating theatre on all 16 eyes. The results after 6 months are as follows: Overall there was a significant improvement of UDVA (p=0.0012) from average preoperative visual acuity of 0.85 logMAR (95% CI, 0.64-1.05) to an average of 0.31 logMAR (0.19-0.43). CDVA also improved significantly (p<0.0001) from 0.69 logMAR (0.45-0.92) to 0.1 logMAR (0.04-0.15), an improvement was achieved in all patients, 15/16 of whom attained minimal visual acuity of 0.3 logMAR (Graphs 1 and 2). UDVA was worse in combined cataract cases in comparison with DMEK alone, on average 0.38 logMAR (0.18–0.43) in comparison with 0.3 logMAR (0.14 – 0.61), nevertheless this difference was not significant (p=0.7786, Mann-Whitney). Average subjective distance refraction and subjective astigmatism were not changed significantly in either group. Keratometry K1 and K2 could not be measured in 3 out of 16 patients before transplantation. Overall there was a significant reduction of corneal astigmatism K2-K1 (p=0.0137) from 2.89 D (0.19-0.43) to 0.98 D (0.48–1.48) in all measured eyes, and average corneal curvature was statistically significantly reduced (p=0.0234) from 44.9 D (43.63–46.22) preoperatively to 44.35 D (42.86–45.83). Table 1.
Graphs 1 and 2. Corrected and uncorrected visual acuity (CDVA and UDVA) in both groups six months after transplantation 
Tab. 1. Subjective refraction, subjective astigmatism and corneal topography 6 months after transplantation in DMEK and DMEK combined with cataract 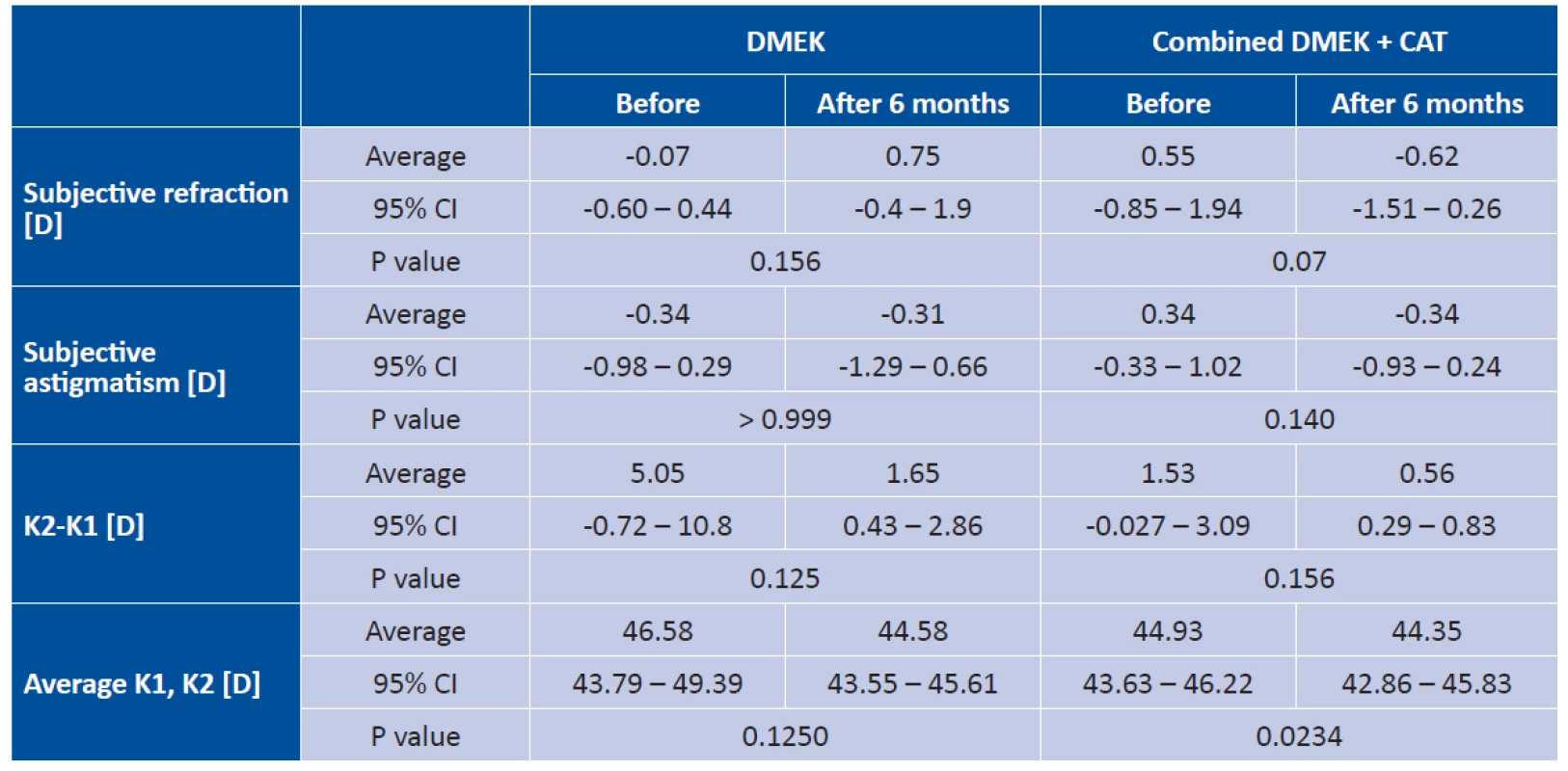
Average density of the endothelial cells provided by the tissue bank was 2826 cells/mm2 (95% CI, 2670-2983) in the donor cornea. Six months after transplantation, the average density of endothelial cells in all eyes was 1336 (95% CI, 1111-1561) cells/mm2. In the DMEK operations the density was 1230 (95% CI, 997 1463) cells/mm2 and in combined operations 1483 (925-2042) cells/mm2. The difference between these two groups was not statistically significant (p=0.4318, Mann-Whitney).
At a follow-up examination after one week the transplanted membrane was not entirely attached in 8/16 eyes. Attachment of the membrane was ensured by repeated application of an air bubble into the anterior chamber. The lower part of the membrane adhered better in 5/8 cases than the upper part. No further complications appeared.
DISCUSSION
The method of separating the membrane of the donor cornea for DMEK by an injection of fluid between the DM and corneal stroma shortens the preparation time and reduces the risk of tearing of the membrane [11, 16]. This method theoretically has the potential for an overall higher number of endothelial cells after surgery due to the lesser mechanical manipulation on the side of the endothelium in comparison with the classic method of scraping. Here we present the first six months of clinical results using this new method of DMEK operation with the aid of separation by fluid which is injected directly between the DM and corneal stroma, separating these two layers by pressure.
Because the corneal stroma remains untouched, the membrane can be unpeeled together with the surrounding tissue, affixed to a support and trimmed by a trephine. The remainder of the donor tissue thus remains intact and can be used e.g. for deep anterior lamellar keratoplasty (DALK), similarly as described by Groeneveld-van Beck [4], which is a further advantage of the described procedure.
Although the loss of endothelial cells we observed is substantial, the density value of endothelial cells six months after transplantation in this study does not differ fundamentally from the previous results of other multicentric studies [8, 10]. At the 26th annual congress of the Czech Ophthalmological Society it was discussed in the Cornea II section that the average density of the endothelial cells indicated by the tissue banks is rather higher than what ordinarily occurs in practice [16]. The absolute postoperative loss is thus dependent on the measurement indicated by the tissue banks. The stated density value of the endothelial cells may be lower due to a difference in the in-vivo and ex-vivo measuring method [20], an imprecise designation of dead cells [6, 22], and may also be dependent on the length of storage, especially in the case of young donors [15]. In future it shall be suitable to focus on a method of measuring endothelial cells before surgery so that it is possible to conduct a postoperative evaluation of the loss more precisely.
15 out of 16 eyes attained the minimum corrected distance visual acuity CDVA declared for driving (0.3 logMAR) after six months. Uncorrected visual acuity UDVA was worse in the cases with combined cataract surgery, because average SR was myopic. With regard to the fact that previous DMEK studies have shown hyperopic SR upon calculation to emmetropia [5, 17], the optical power of the intraocular lens was increased against the calculation, which caused a myopic resulting SR. Further optimisation of the calculation of intraocular lenses in combined cases shall be the subject of further studies.
The most common postoperative complication is incomplete adhesion of the implanted DM. The frequency at which an air bubble must have been reapplied is variable, and is stated within the range of 9-82 [13]. In our study the bubble had to be reapplied in 50 eyes, after which complete healing was achieved in all eyes. In cases where complete adhesion did not occur in the lower part of the cornea, we suspect that this could have been caused by failure to abide by the postoperative regimen of head position, in which there was a shifting of the bubble to the upper part of the anterior segment of the eye. For this reason we are developing an instrument which shall monitor the patient’s head position after the operation and help adhere to the postoperative regime.
This study presents the first clinical results for DMEK and combined DMEK with cataract surgery in which the donor membrane was prepared by injection of fluid between the DM and the corneal stroma. This method is effective, fast and involves minimal manipulation on the side of the endothelial cells. DMEK improves visual acuity and produces only a small value of astigmatism six months after surgery. Further clinical trials are necessary in order to confirm this success and for further optimisation of the calculation in the case of combination with cataract surgery.
The author of the study declares that no conflict of interest exists in the compilation, theme and subsequent publication of this professional communication, and that it is not supported by any pharmaceuticals company.
Received by the Editorial Department on: 20 July 2018
Accepted for printing on: 04 February 2019
Šramka M.
Address: Gemini Eye Clinic, U Gemini 360, 760 01 Zlín, Czech Republic
E-mail: sramka@gemini.cz
Zdroje
1. Altaan, S.L., Gupta, A., Sidney, L.E., et al.: Endothelial cell loss following tissue harvesting by pneumodissection for endothelial keratoplasty: An ex vivo study. Br. J. Ophthalmol., 99(5); 2015 : 710–3.
2. Droutsas, K., Giallouros, E., Melles, G.R.J., et al.: Descemet Membrane Endothelial Keratoplasty. Cornea, 32; 2013 : 1075–1079.
3. Dua, H.S., Faraj, L.A., Said, D.G., et al.: Human Corneal Anatomy Redefined. Ophthalmology, 120; 2013 : 1778–1785.
4. Groeneveld-van Beek, E.A., Lie, J.T., van der Wees, J., et al.: Standardized ‘no-touch’ donor tissue preparation for DALK and DMEK: harvesting undamaged anterior and posterior transplants from the same donor cornea. Acta Ophthalmol., 91; 2013 : 145–150.
5. Ham, L., Dapena, I., Moutsouris, K., et al.: Refractive change and stability after Descemet membrane endothelial keratoplasty. J. Cataract Refract. Surg., 37; 2011 : 1455–1464.
6. Jirsova, K.: Chapter 3 and Chapter 4. Light and Specular Microscopy of the Cornea. Springer International Publishing; Book. 2017:
7. Lie, J.T., Birbal, R., Ham, L., et al.: Donor tissue preparation for Descemet membrane endothelial keratoplasty. J. Cataract Refract. Surg., 34; 2008 : 1578–1583.
8. Melles, G.R.J., Ong, T.S., Ververs, B., et al.: Descemet Membrane Endothelial Keratoplasty (DMEK). Cornea, 25; 2006 : 987–990.
9. Monnereau, C., Quilendrino, R., Dapena, I., et al.: Multicenter Study of Descemet Membrane Endothelial Keratoplasty. JAMA Ophthalmol., 132; 2014 : 1192.
10. Muraine, M., Gueudry, J., He, Z., et al.: Novel Technique for the Preparation of Corneal Grafts for Descemet Membrane Endothelial Keratoplasty. Am. J. Ophthalmol., 156; 2013 : 851–859.
11. Oellerich, S., Baydoun, L., Peraza-Nieves, J., et al.: Multicenter Study of 6-Month Clinical Outcomes After Descemet Membrane Endothelial Keratoplasty. Cornea, 36; 2017 : 1467–1476.
12. Parekh, M., Borroni, D., Ruzza, A., et al.: A comparative study on different Descemet membrane endothelial keratoplasty graft preparation techniques. Acta Ophthalmol., 2018, DOI: 10.1111/aos.13746.
13. Parekh, M., Leon, P., Ruzza, A., et al.: Graft detachment and rebubbling rate in Descemet membrane endothelial keratoplasty. Surv. Ophthalmol., 63; 2018 : 245–250.
14. Parekh, M., Ruzza, A., Salvalaio, G., et al.: Descemet Membrane Endothelial Keratoplasty Tissue Preparation From Donor Corneas Using a Standardized Submerged Hydro-separation Method. Am. J. Ophthalmol., 158; 2014 : 277–285.
15. Rodríguez-Calvo de Mora, M., Groeneveld-van Beek, E.A., Frank, L.E., et al.: Association Between Graft Storage Time and Donor Age With Endothelial Cell Density and Graft Adherence After Descemet Membrane Endothelial Keratoplasty. JAMA Ophthalmol., 134; 2016 : 91.
16. Diskuze v sekci Rohovka II. na XXVI. Výroční Sjezd České Oftalmologické Společnosti ČLS JEP v Praze 13.-15.9.2018. BOS org s.r.o. ISBN 978-80-87562-80-2
17. Salvalaio, G., Parekh, M., Ruzza, A., et al.: DMEK lenticule preparation from donor corneas using a novel ‘SubHyS’ technique followed by anterior corneal dissection. Br. J. Ophthalmol., 98; 2014 : 1120–1125.
18. Satué, M., Rodríguez-Calvo-de-Mora, M., Naveiras, M., et al.: La estandarización en el trasplante endotelial de membrana de Descemet: resultados de las primeras 450 cirugías. Arch. Soc. Esp. Oftalmol., 90; 2015 : 356–364.
19. Szurman, P., Januschowski, K., Rickmann, A., et al.: Novel liquid bubble dissection technique for DMEK lenticule preparation. Graefe’s Arch. Clin. Exp. Ophthalmol., 254; 2016 : 1819–1823.
20. Thuret, G., Deb-Joardar, N., Manissolle, C., et al.: Assessment of the human corneal endothelium: In vivo Topcon SP2000P specular microscope versus ex vivo sambacornea eye bank analyser. Br. J. Ophthalmol. 91; 2007 : 256-266.
21. van Dijk, K., Ham, L., Tse, W.H.W., et al.: Near complete visual recovery and refractive stability in modern corneal transplantation: Descemet membrane endothelial keratoplasty (DMEK). Contact Lens Anterior Eye, 36; 2013 : 13–21.
22. Wilhelmus, K.R., Stulting, R.D., Sugar, J., et al.: Primary corneal graft failure. A national reporting system. Medical Advisory Board of the Eye Bank Association of America. Arch. Ophthalmol. (Chicago, Ill. 1960), 113; 1995 : 1497–502.
Štítky
Oftalmologie
Článek vyšel v časopiseČeská a slovenská oftalmologie
Nejčtenější tento týden
2019 Číslo 1- Stillova choroba: vzácné a závažné systémové onemocnění
- Familiární středomořská horečka
- Diagnostický algoritmus při podezření na syndrom periodické horečky
- Možnosti využití přípravku Desodrop v terapii a prevenci oftalmologických onemocnění
- Selektivní laserová trabekuloplastika nesnižuje nitroční tlak více než argonová laserová trabekuloplastika
-
Všechny články tohoto čísla
- Usage of shear wave elastography for diagnosis of changes of oculomotor muscles in endocrine orbitopathy
- Treatment of idiopathic choroidal neovascular membrane with ranibizumab - our experience
- RADIATION MACULOPATHY AFTER ONE-DAY SESSION STEREOTACTIC RADIOSURGERY IN PATIENTS WITH CILIARY BODY AND CHOROIDAL MELANOMA
- Effective Tissue Layer Separation of Donor Cornea for DMEK by Fluid Injection Between Descemet
- Primary orbital teratoma – case study
- Late postoperative capsular bag distension syndrome
- Česká a slovenská oftalmologie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Late postoperative capsular bag distension syndrome
- Primary orbital teratoma – case study
- Treatment of idiopathic choroidal neovascular membrane with ranibizumab - our experience
- RADIATION MACULOPATHY AFTER ONE-DAY SESSION STEREOTACTIC RADIOSURGERY IN PATIENTS WITH CILIARY BODY AND CHOROIDAL MELANOMA
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



