-
Medical journals
- Career
Abnormal Sleep Microstructure and Autonomic Response in Narcolepsy
Authors: J. Vaňková; P. Bušek; J. Volná; K. Šonka; S. Nevšímalová
Authors‘ workplace: Neurologická klinika 1. LF MU a VFN Praha
Published in: Cesk Slov Neurol N 2007; 70/103(2): 158-162
Category: Original Paper
Overview
The present study was aimed at analysing the non-rapid eye movement (NREM) sleep microstructure by the cyclic alternating pattern (CAP) and at assessing the heart rate variability (HRV) changes in patiens with narcolepsy, hypothesizing a correlation of an abnormal sleep microstructure and abnormal autonomic response with a selective loss of hypocretin-containing neurons in narcolepsy. Fifteen patients with narcolepsy-cataplexy (mean age 35±8.5; age range 22-44), and 15 age and sex matched controls (mean age 31±11.4; age range 19-48) were included in the study. All subjects underwent polysomnography recordings for two consecutive nights in a standard laboratory setting. The sleep scoring was focused on the CAP and HRV analysis. A significant decrease in CAP rate as well as significant reduction of the LF spectral band and the LF/HF ratio, and the elevation of the HF spectral component during NREM 4 stage were revealed in narcoleptics compared to controls. Our results suggest that physiological fluctuation of arousal during sleep described as CAP is impaired in narcolepsy and accompained by reduced sympathetic tone during SWS. We have hypothesized that the whole sleep regulation process is altered in narcolepsy and not only REM sleep mechanisms.
Key words:
narcolepsy · hypocretin · cyclic alternating pattern · heart rate variability
Narcolepsy with cataplexy is a homogeneous disabling neurological disease linked to the DQB1*0602 haplotype [1] affecting approximately one in 2000 individuals. It is characterized by excessive daytime sleepiness, cataplexy and disturbed nocturnal sleep, often also by hypnagogic hallucinations [2] and sleep paralysis, is related to a severe hypocretin deficiency [3,4,5]. Hypocretin deficiency accounts for REM sleep abnormalities [6,7] but also for non-rapid eye movement (NREM) sleep abnormalities [8].
We have focused on the cyclic alternating pattern (CAP) analysis, which is the natural arousal rhythm of NREM sleep [9,10]. CAP consists of arousal-related phasic events (phase A) that interrupt, at the intervals of 20-40s, the tonic theta/delta activities of NREM sleep (phase B). Functionally, CAP translates a condition of sustained arousal instability, while the complementary EEG pattern, i.e. non-CAP (NCAP), characterized by a rhytmic background activity reflects a condition of stable arousal. The CAP sequences constitute the microstructural component of sleep that accompanies dynamic shifts between the NREM stages and seems to play its role in the transition from NREM to REM sleep. The physiological balance between CAP and NCAP has been found to be altered in a number of sleep-disturbed conditions [11,12]. CAP and NCAP also have repercussions on motor and vegetative activites. Some relations between these microstructural parameters and autonomic function have been preliminary reported in young adults using the spectral analysis of the heart rate variability during sleep [13] and in children and adolescents [14]. The heart rate is under the control of efferent sympathetic and vagal activities directed to the sinus node, which are modulated by the central brainstem and peripheral oscillators [15], this method allows to analyse the modullatory effects of neural mechanisms on the sinus node, and two main components are currently considered, high-frequency (HF) and low-frequency (LF).The vagal activity is the major contributor to the HF component, while the LF is considered by both vagal and sympathetic influences.
The aim of the study was to assess not only conventional sleep parameters, but also microstructure by analysing CAP and HRV in adult narcoleptic patients.
Patients and Methods
Fifteen patients with narcolepsy – cataplexy (mean age 35±8.5; age range 22-44), and 15 age - and sex-matched controls (mean age 31±11.4; age range 19-48) were included in the study. All subjects had classical history of imperative sleepiness and cataplexy, while the presence of further symptoms was not unconditional. All the patients had findings typical of narcolepsy on overnight polysomnography and the Multiple Sleep Latency Test (MSLT), all of them were HLA DQB1*0602 positive. The hypocretin level in the cerebrospinal fluid was not measured. None of the patients suffered from other sleep disorders. All medication was discontinued at least 2 weeks before recording (metylphenidate, modafinil).
The control group consisted of healthy volunteers reporting subjectively normal sleep habits, with no complaint of either insomnia or excessive daytime sleepiness, receiving no sedatives or stimulants. All subjects gave informed consent before the investigation.
Polygraphic recording and scoring
Each subject underwent polysomnography recordings for two consecutive nights in a standard laboratory setting, only the data from the second night were analysed further. The polygraphic recording started between 10 : 00 and 11 : 00 p.m., and ended by spontaneous awakening between 6 : 00 and 8 : 00 a.m., the recordings were made using the standard placement of EEG electrodes (F4-C4, C4-P4, F3-C3, C3-P3, C4-A1, and C3-A2, sampling rate 128 Hz), submental and bilateral anterior tibialis EMG, horizontal electro-oculogram, termistor for recording the respiration, abdominal and thoracic sensors for recording the breathing effort, pulse oxymeter, and infrared video camera. The sleep macrostructure was visually scored according to international criteria [16].
CAPs were visually scored by one scorer [17]. The cyclic alternating pattern is a periodic EEG activity of NREM sleep characterized by repeated spontaneous sequences of transient events (phase A). The return to background activity identifies the interval, that separates the repetitive elements (phase B). CAP A phases are subdivided into a 3-stage hierarchy of arousal strength: A1 with synchronized EEG patterns and mild polygraphic variations; A2 with desynchronized EEG patterns preceded by or mixed with slow high-voltage waves and a moderate increase of muscle tone and/or cardiorespiratory rate; A3 with desynchronized EEG patterns alone and coupled with a remarkable enhancement of muscle tone and/or cardiorespiratory rate.
In the course of polygraphic recording, the heart rate signal was obtained from a surface pre-cordial ECG lead (sampling rate 500Hz) , and the R-R interval sequence was obtained by a computerized algorithm of the ECG registration. The time series trend was calculated, and the non-equidistant time sequence was interpolated using a third grade function – cubic spline – and further sampled at a sampling frequency of 4 Hz. Then a fast-Fourier transformation of the adjusted time series was carried out to determine the power spectral analysis, which was decomposed into the following spectral components: VLF 0.02-0.05 Hz, LF 0.05-0.15Hz, and HF 0.15-0.4Hz. The analysis was performed from repeated ectopic-free intervals of approximately 5 min (256 beats) during (i) 15-20min of the wake state before sleep onset (when the lights were on, and wakefulness was controlled by a technician and by EEG), during (ii) stage 2 NREM sleep, (iii) stage 4 NREM sleep, and (iv) REM sleep. In this way, the whole course of the mentioned sleep stages was analysed, eliminating only the segments containing ectopic beats, movement artefacts or arousal.
Statistical analysis included the analysis of variance (ANOVA), Friedman test, Tukey test (for post-hoc multiple comparison), Student´s t-test and Wilcoxon matched paired test. The normal distribution was evaluated using D´Agostino Omnibus test and Shapiro-Wilks test.
Results
Macrostructural polygraphic parameters of narcoleptics and of normal subjects are shown in Table 1. The sleep onset latency and REM sleep latency were both significantly shorter in patients with narcolepsy than in normal subjects, narcoleptic patients also showed significantly lower values of sleep efficiency. The number of awakenings per hour, percentage of WASO and that of NREM 1 and REM were significantly higher, while the percentages of NREM 2 and slow-wave sleep (SWS) were significantly reduced in narcoleptic patients.
1. Polygraphic macrostructural parameters in narcoleptics and normal subjects. 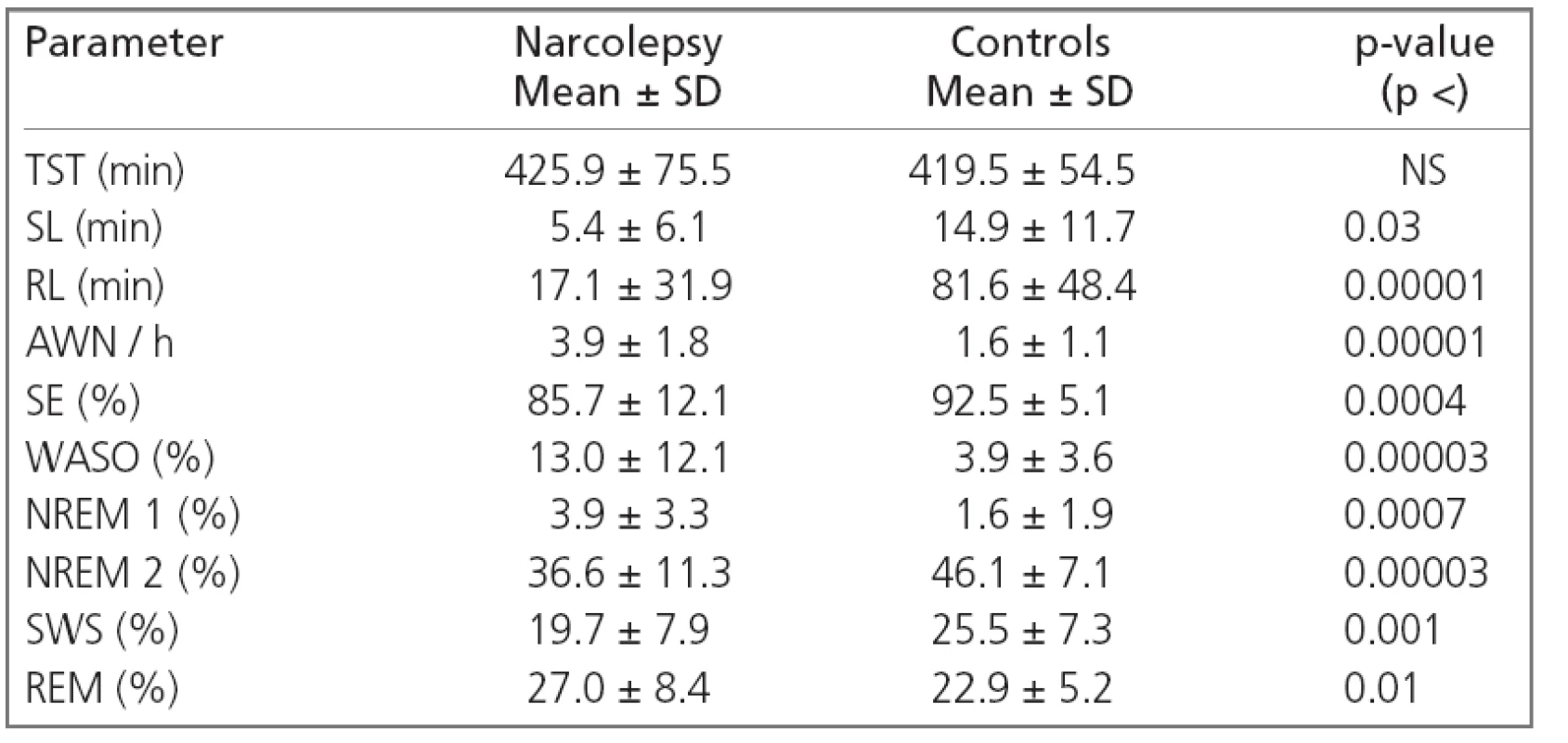
TST: total sleep time, SL: sleep latency, RL: first REM latency, AWN/h: number of awakenings per hour, SE: sleep efficiency, WASO: wakefulness after sleep onset, SWS: slow-wave sleep The comparison between CAP parameters from narcoleptic patients and controls disclosed significantly reduced total CAP rate (i.e. the percentage of NREM sleep occupied by CAP sequences) in our patients with narcolepsy because of its reduction during NREM 2 and SWS. The relative proportion of the different CAP subtypes also differed in our patients because of its reduction in the percentage of A1 subtypes and the increase in A2 and A3 subtypes. This disproportion was due to a selective significant reduction in the number of A1 subtypes/h (A1 index) and, to a lesser extent, a reduction in A3 index. Finally, a significant difference was also found for the average number of CAP sequences, which was smaller in narcoleptic patients than in controls (Table 2).
2. CAP parameters in narcoleptics and normal subjects. 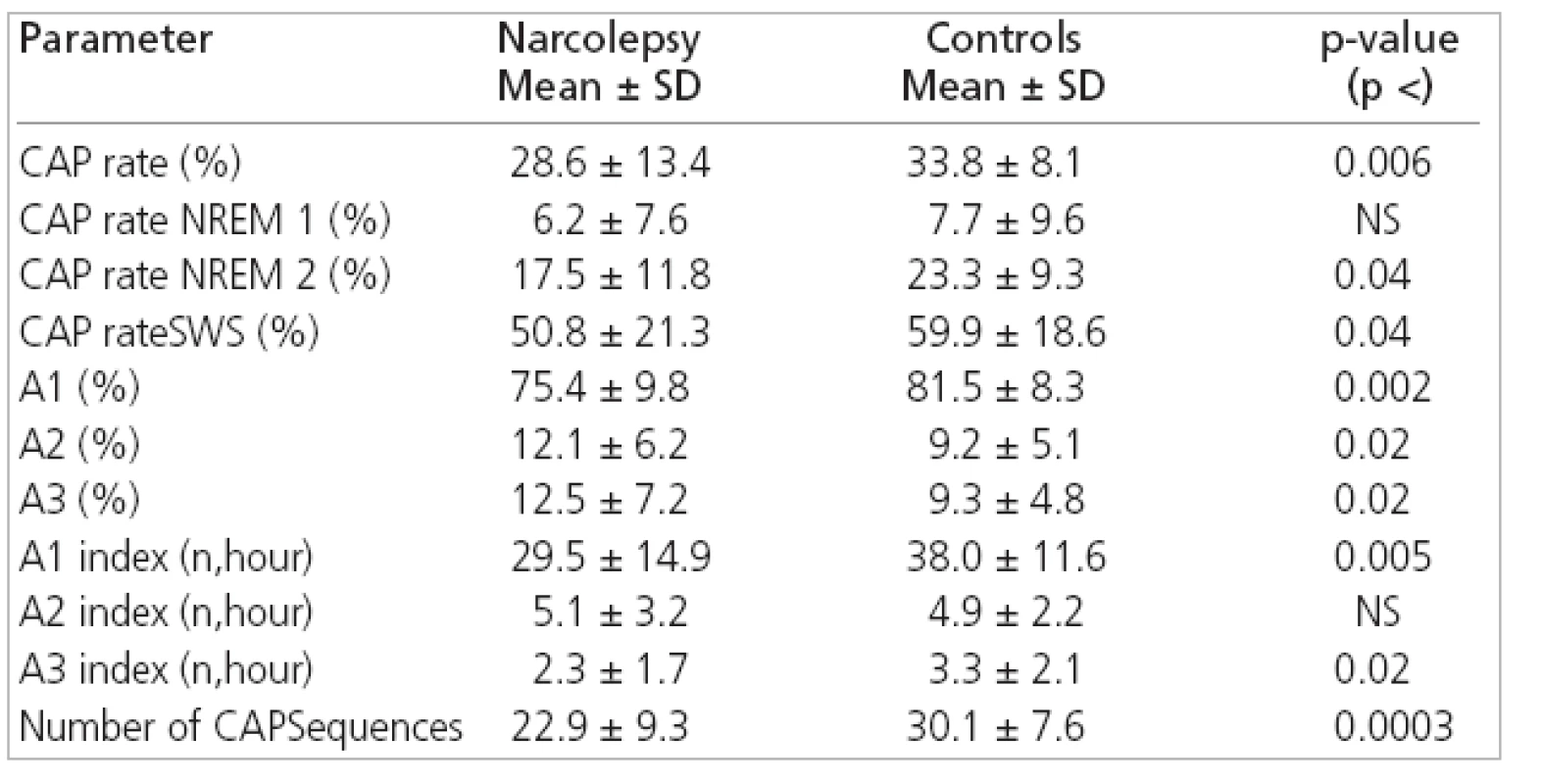
CAP rate: percentage of NREM sleep occupied by CAP sequences, A index: number of A subtype per hour 3. Parameters of spectral analysis of HRV during sleep in narcoleptics and normal subjects. 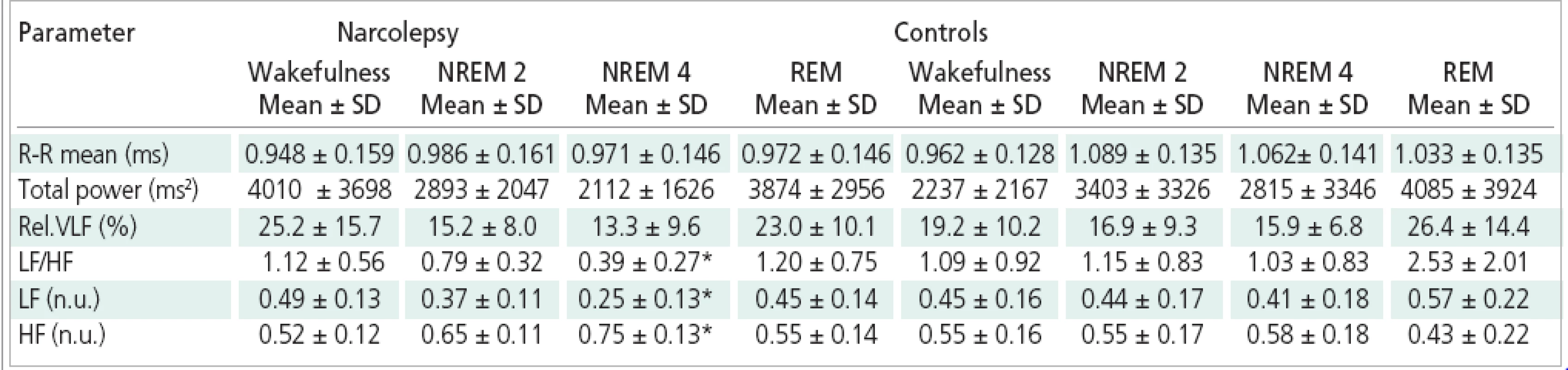
Rel.VLF = relative value of the VLF component; LF (n.u.), HF (n.u.) = values of the LF and HF components in normalized units *p < 0.05 vs controls Table 3 shows the comparison of autonomic response between narcoleptic patiens and healthy controls. There was no significant difference in the assessed part of the VLF spectral band between the groups in any sleep stage. The statistical analysis indicates a significant difference between narcoleptics and control group in LF component, this component being lower in the patient group during NREM 4. In both groups, the individual sleep stages differed in the normalized values of the LF band with significantly lower values in NREM 4 sleep and in NREM 2 sleep compared to REM sleep in the control group and lower values in NREM 4 sleep compared to wakefulness and REM sleep in the narcoleptics. The difference in HF spectral component between narcoleptics and controls was statistically borderline (p=0.05) - the HF component in normalized units was higher in narcoleptics than in controls in NREM 4 sleep. The other sleep stages and wakefulness did not differ between narcoleptics and controls in this spectral band. In both groups the HF component in normalized units differed significantly between the sleep stages; in controls the HF component was significantly higher in NREM 2 and NREM 4 sleep than in REM sleep; in narcoleptics the HF component was significantly higher in NREM 4 sleep compared to wakefulness and REM sleep. The LF/HF ratio was significantly lower in narcoleptics than in control group during NREM 4, and changed significantly between the individual sleep stages. In controls the LF/HF ratio was significantly higher in REM sleep in comparison with NREM 2, NREM 4 and wakefulness.In narcoleptics, the LF/HF ratio was significantly lower in NREM 4 compared to the wakefulness and REM sleep.
Neither group differed in the total spectrum power in any sleep stage. Statistically significant changes of the total spectrum power between the individual sleep stages were found in both groups of subjects with significantly higher value of the total spectrum power in REM sleep in comparison with NREM 4 sleep.
The group of narcoleptics did not differ in the length of the R-R interval from the control group in any sleep stage or during the wakefulness. The R-R interval length has changed significantly between the individual sleep stages in both groups. In controls, the R-R interval was significantly shorter in wakefulness than in NREM 2, in NREM 4 and in REM sleep. The R-R interval in narcoleptics was also shorter during wakefulness than in the individual sleep stages, but the differences were not statistically significant.
Discussion
While our patients´ sleep structure changes are in a good agreement with previous data, NREM sleep microstructure, as reflected by the CAP changes and autonomic response, was significantly impaired. The sleep macrostructural changes found in our narcoleptic patients were expected. In fact, DQB1*0602 positive narcoleptic patients show a reduced REM latency and sleep efficiency, and increased percentage of WASO and stage 1, as well as a reduced amount of stage 2 and SWS [18].
The new finding in our study was a reduced amount of CAP in DQB1*0602 positive narcoleptic patients. CAPs were composed of different transient events indicated as subtypes A1, A2 and A3 [17] which functionally are thought to translate a condition of sustained arousal instability oscillating between greater (phase A) and lesser (phase B) arousal level. Among the different subtypes of the CAP A phases, the most common is A1 characterized by sequences of K-complexes or delta bursts in NREM sleep stages, associated with mild polygraphic variations and activation of somatic and autonomic systems [17,19].
Our results have indicated that the occurrence of this CAP component is impaired during NREM sleep in narcoleptic patients. Thus, narcolepsy is accompained not only by alterations of REM but also NREM sleep which is impaired subtly, but significantly. Our results are in a good agreement with those presented previously [20].
The alteration between sleep and wakefulness seems to be regulated by multiple arousal systems and each of them seems to play a specific role in waking and sleep. Hypocretin appears only to affect nonspecific thalamocortical projection neurons, with no effects on the specific sensory relay thalamic neurons. Hypocretin may thus act in the thalamus to promote wakefulness by exciting neurons of the nonspecific thalamocortical projection system which stimulate and maintain cortical activation via widespread projections to the cerebral cortex. Also in the cortex, hypocretin has been shown to excite a specific subset of cortical neurons, which may be also involved in promoting widespread cortical activation via corticocortical projections [21]. In narcolepsy hypocretin defficiency may reflect in the reduced activity of cortical arousal regions resulting in the reduced CAP rate encountered in NREM sleep.
During sleep, the patient group differed from controls in a higher normalized value of HF component, lower normalized LF component, and lower LH/HF ratio during NREM 4 sleep. In the course of the other sleep stages, the differences in these parameters of HRV between narcoleptics and controls were similar, but did not reach statistical significance. This observation indicates that, compared to normal subjects, in narcoleptics the sympathovagal balance is more shifted towards prevailing parasympathetic influence in stage 4 NREM sleep. From the CAP standpoint, transient slow-wave activity bursts (A1 subtypes) are considered cortical activation events also accompained with autonomic subcortical changes [13,14,19].
Our findings of both microstructural and autonomic alterations may originate in a deficient hypocretin system in narcolepsy [3,22]. The hypocretin neurons in the perifornical region of the hypothalamus not only participate in the regulation of the sleep-wake cycle, but they are also involved in the regulation of autonomic nervous functions [23,24]. The hypocretinergic fibres are widespread in the hypothalamus, brainstem and spinal cord. Among the sites receiving the hypocretinergic innervation and playing an important role with respect to the cardiovascular regulation there are the paraventricular nucleus in the hypothalamus, nucleus of the solitary tract, rostral ventrolateral area in the medulla oblongata, and intermediolateral column in the spinal cord [25]. The effect of hypocretin on the cardiovascular function probably arises predominantly in the rostral ventrolateral area in the medulla oblongata – hypocretin injected into this region produces hypertension and tachycardia [26,27,28,29].
Autonomic disturbances, such as abnormalities of pupil reactions, sexual dysfunction, or cardiovascular dysregulation in narcolepsy have been described repeatedly [30,31,32]. Increased blood pressure and decreased heart rate during the onset of cataplexy were documented in literature [33], and the reduced blood pressure reactivity to the handgrip and subnormal sinus respiratory arrhythmia in narcoleptics were observed [31].
In conclusion, our findings have shown that not only REM but also NREM sleep are significantly altered in narcolepsy. Neurophysiological alterations of NREM sleep microstructure and autonomic alterations in narcolepsy are significant and provide new information on the sleep neurophysiology.
Práce vznikla za podpory grantu GACR 309/03/P026
MUDr. Jitka Vaňková-Boušková, Ph.D.
Neurologická klinika
1. LF UK a VFN
Kateřinská 30, 128 08 Praha 2
Doručeno: 27. 1. 2006
Přijato do tisku: 13. 3. 2006
Sources
1. Mignot E, Lin X, Arrigoni J, Macaubas C, Olive F, Hallmayer J et al. DQB1*0602 and DQA1*0102 (DQ1) are better markers than DR2 for narcolepsy in Caucasian and black Americans. Sleep 1994 17 : 60–7.
2. American Academy of Sleep Medicine. International classification of sleep disorders (ICSD): Diagnostic and coding manual. 2nd ed. Westchester (Ill): American Academy of Sleep Medicine, 2005.
3. Nishino N, Ripley B, Overeem S, Lammers GJ, Mignot E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 2000 355 : 39–40.
4. Mignot E, Lammers GJ, Ripley B, Okun M, Nevsimalova S, Overeem S et al. The role
of cerebrospinal fluid hypocretin measurement in the diagnosis of narcolepsy and other hypersomnias. Arch Neurol 2002 59 : 1553–62.
5. Dauvilliers Y, Baumann CR, Maly FE, Billiard M, Bassetti C. CSF hypocretin-1 levels in narcolepsy, Kleine–Levin syndrome, other hypersomnias and neurological conditions. J Neurol Neurosurg Psychiatry 2003 74 : 1667–73.
6. Hungs M, Mignot E. Hypocretin/orexin, sleep, and narcolepsy. BioEssays 2001 23 : 397–408.
7. Burlet S, Tyler CJ, Leonard CS: Direct and indirect excitation of laterodorsal tegmental neurons by hypocretin/orexin peptides: implications for wakefulness and narcolepsy. J Neurosci 2002 22 : 2862–72.
8. Mochizuki T, Crocker A, McCormack S, Yanagisawa M, Sakurai T, Scamell TE. Behavioral state instability in orexin knock-out mice. J Neurosci 2004 24 : 6291 – 300.
9. Terzano MG, Mancia D, Salati MR, Costani G, Decebrino A, Parrino L.
The cyclic alternating pattern as a physiologic component of normal NREM sleep. Sleep 1985 8 : 137–45.
10. Terzano MG, Parrino L, Spaggiari MC. The cyclic alternating pattern sequences in the dynamic organization of sleep. Electroencephalogr Clin Neurophysiol 1988 69 : 437–47.
11. Terzano MG, Parrino L. Clinical applications of cyclic alternating pattern. Physiol Behav 1993 54 : 807-13.
12. Zucconi M, Oldani A, Ferini-Strambi L, Smirne S. Arousal fluctuations in non-rapid eye movement parasomnias: the role of cyclic alternating pattern as a measure of sleep instability. J Clin Neurophysiol 1995 12 : 147-54.
13. Ferini-Strambi L, Bianchi A, Zucconi M, Oldani A, Castronovo V, Smirne S. The impact of cyclic alternating pattern on heart rate variability during sleep in healthy young adults. Clin Neurophysiol 2000 111 : 99–101.
14. Ferri R, Parrino L, Smerieri A, Terzano MG, Elia M, Musumeci SA et al. Cyclic alternating pattern and spectral analysis of heart rate variability during normal sleep. J Sleep Res 2000 9 : 13–18.
15. Malliani A, Pagani M, Lombardi F, Cerutti S. Cardiovascular neural regulation explored in the frequency domain. Circulation 1991 84 : 1482-92.
16. Rechtschaffen A, Kales A. A manual of standardized terminology, techniques and scoring system of sleep stages of human subjects. Washington DC: US Government Printing Office, US Public Health Service1968.
17. Terzano MG, Parrino L, Smerieri A, Chervin R, Chocroverty S, Guilleminault C et al. Consensus report. Atlas, rules, and recording techniques for the scoring of cyclic alternating pattern (CAP) in human sleep. Sleep Med 2001 2 : 537–53.
18. Mukai J, Uchida S, Miyazaki S, Nishihara K, Honda Y. Spectral analysis of all-night human sleep EEG in narcoleptic patients and normal subjects. J Sleep Res 2003 12 : 63–71.
19. Sforza E, Jouny C, Ibanez V. Cardiac activation during arousal in humans: further evidence for hierarchy in the arousal response. Clin Neurophysiol 2000 111 : 1611–9.
20. Ferri R, Miano S, Bruni O, Vankova J. NREM sleep alterations in narcolepsy/cataplexy. Clin Neurophysiol 2005 116 : 2675-84.
21. Bayer L, Serafin M, Eggerman E, Saint-Mleuux B, Machard D, Jones BE et al. Exclusive postsynaptic action of hypocretin–orexin on sublayer 6b cortical neurons. J Neurosci 2004 24 : 6760–4.
22. Kilduff TS, Peyron C. The hypocretin/orexin ligand-receptor system: implications for sleep and sleep disorders. Trends Neurosci 2000 23 : 359-65.
23. Date Y, Ueta Y, Yamashita H, Yamaguchi H, Matsukura S, Kangawa K, et al. Orexins, orexinergic hypothalamic peptides, interact with autonomic, neuroendocrine and neuroregulatory systems. Proc Natl Acad Sci USA 1999 96 : 748-53.
24. Peyron C, Tighe DK, van den Pol AN, Lecea L, Heller HC, Sutcliffe JG et al. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J Neurosci 1998 18(23): 9996-10015.
25. Ehmke H, Just A. The orexins: linking circulatory control with behavior. Am J Physiol Regul Integr Comp Physiol 2003 285: R519-21.
26. Ciriello J, Li Z, De Oliviera CV. Cardioacceleratory responses to hypocretin-1 injections into rostral ventromedial medulla. Brain Res 2003 991(1-2):84-95.
27. Dun NJ, Le Dun S, Chen CT, Hwang LL, Kwok EH, Chang JK. Orexins: a role in medullary sympathetic outflow. Regul Pept 2000 96 : 65-70.
28. Machado BH, Bonagamba LG, Dun SL, Kwok EH, Dun, NJ. Pressor response to microinjection of orexin/hypocretin into rostral ventrolateral medulla of awake rats. Regul Pept 2002 104(1-3): 75-81.
29. Llewellyn-Smith IJ, Martin CL, Marcus JN, Yanagisawa M, Minson JB, Scammell TE. Orexin-immunoreactive inputs to rat sympathetic preganglionic neurons. Neurosci Lett 2003 351 : 115-119.
30. Yoss R, Moyer A, Ogle K. The pupillogram and narcolepsy: a method to measure decreased levels of wakefulness. Neurology 1969 19 : 921-928.
31. Sachs C, Kaijser L. Autonomic control of cardiovascular reflexes in narcolepsy. J Neurol Neurosurg Psychiatry 1980 43(6): 535-539.
32. Caracan I: Erectile dysfunction in narcoleptic patients. Sleep 1996 9 : 227 - 231.
33. Guilleminault C, Heinzer R, Mignot E, Black J. Investigations into the neurologic basis of narcolepsy. Neurology 1998 50 (2 Suppl 1): S8-S15.
Labels
Paediatric neurology Neurosurgery Neurology
Article was published inCzech and Slovak Neurology and Neurosurgery

2007 Issue 2-
All articles in this issue
- The Current View of the Diagnostics and Therapy of Aphasias
- Effect of Bolus Dose of Intrathecal Baclofen Followed by Pump Implantation in Severe Spasticity in Multiple Sclerosis Patients
- Surgical Treatment of Ependymomas in the Cervical and Upper Thoracic Spinal Cord
- Complications of the Anterior Cervical Spine Surgery for a Degenerative Disease
- Solitary Fibrous Tumor of Meninges – a Case Report
- Epilepsy and the Sleep-Waking Cycle
- Worsening of Epileptic Seizures and Epilepsies due to Antiepileptic Drugs – is it Possible?
- The Importance of MRI for the Indication of Systemic Thrombolysis – Analysis of the First 30 Patients
- Specific anti-beta tubulin antibodies in differential diagnosis of dementias
- The Risk of Ischaemic Stroke during the Fluvastatine and Fenofibrate Treatment
- Correlation between ptiO2 and Apoptosis in Focal Brain ischaemia and the Influence of Systemic Hypertension
- Leksell Gamma Knife Radiosurgery for Trigeminal Schwannoma
- Abnormal Sleep Microstructure and Autonomic Response in Narcolepsy
- A Heart Rate Turbulence Study to Assess Cardiac Autonomic Function in Migraineurs
- Pontocerebellar Angle Extension of a Basal Cell Carcinoma of the Scalp Followed by Ipsilateral Acusticus Neurinoma – A Case Report
- Czech and Slovak Neurology and Neurosurgery
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Epilepsy and the Sleep-Waking Cycle
- The Current View of the Diagnostics and Therapy of Aphasias
- Complications of the Anterior Cervical Spine Surgery for a Degenerative Disease
- Worsening of Epileptic Seizures and Epilepsies due to Antiepileptic Drugs – is it Possible?
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

