-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Association of eGFR-Related Loci Identified by GWAS with Incident CKD and ESRD
Family studies suggest a genetic component to the etiology of chronic kidney disease (CKD) and end stage renal disease (ESRD). Previously, we identified 16 loci for eGFR in genome-wide association studies, but the associations of these single nucleotide polymorphisms (SNPs) for incident CKD or ESRD are unknown. We thus investigated the association of these loci with incident CKD in 26,308 individuals of European ancestry free of CKD at baseline drawn from eight population-based cohorts followed for a median of 7.2 years (including 2,122 incident CKD cases defined as eGFR <60ml/min/1.73m2 at follow-up) and with ESRD in four case-control studies in subjects of European ancestry (3,775 cases, 4,577 controls). SNPs at 11 of the 16 loci (UMOD, PRKAG2, ANXA9, DAB2, SHROOM3, DACH1, STC1, SLC34A1, ALMS1/NAT8, UBE2Q2, and GCKR) were associated with incident CKD; p-values ranged from p = 4.1e-9 in UMOD to p = 0.03 in GCKR. After adjusting for baseline eGFR, six of these loci remained significantly associated with incident CKD (UMOD, PRKAG2, ANXA9, DAB2, DACH1, and STC1). SNPs in UMOD (OR = 0.92, p = 0.04) and GCKR (OR = 0.93, p = 0.03) were nominally associated with ESRD. In summary, the majority of eGFR-related loci are either associated or show a strong trend towards association with incident CKD, but have modest associations with ESRD in individuals of European descent. Additional work is required to characterize the association of genetic determinants of CKD and ESRD at different stages of disease progression.
Published in the journal: . PLoS Genet 7(9): e32767. doi:10.1371/journal.pgen.1002292
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002292Summary
Family studies suggest a genetic component to the etiology of chronic kidney disease (CKD) and end stage renal disease (ESRD). Previously, we identified 16 loci for eGFR in genome-wide association studies, but the associations of these single nucleotide polymorphisms (SNPs) for incident CKD or ESRD are unknown. We thus investigated the association of these loci with incident CKD in 26,308 individuals of European ancestry free of CKD at baseline drawn from eight population-based cohorts followed for a median of 7.2 years (including 2,122 incident CKD cases defined as eGFR <60ml/min/1.73m2 at follow-up) and with ESRD in four case-control studies in subjects of European ancestry (3,775 cases, 4,577 controls). SNPs at 11 of the 16 loci (UMOD, PRKAG2, ANXA9, DAB2, SHROOM3, DACH1, STC1, SLC34A1, ALMS1/NAT8, UBE2Q2, and GCKR) were associated with incident CKD; p-values ranged from p = 4.1e-9 in UMOD to p = 0.03 in GCKR. After adjusting for baseline eGFR, six of these loci remained significantly associated with incident CKD (UMOD, PRKAG2, ANXA9, DAB2, DACH1, and STC1). SNPs in UMOD (OR = 0.92, p = 0.04) and GCKR (OR = 0.93, p = 0.03) were nominally associated with ESRD. In summary, the majority of eGFR-related loci are either associated or show a strong trend towards association with incident CKD, but have modest associations with ESRD in individuals of European descent. Additional work is required to characterize the association of genetic determinants of CKD and ESRD at different stages of disease progression.
Introduction
Chronic kidney disease (CKD) and end stage renal disease (ESRD) are associated with significant cardiovascular morbidity and mortality, with substantial economic burden [1]–[4]. Diabetes and hypertension are the primary risk factors for CKD and ESRD [5]–[8] but do not fully account for CKD and ESRD risk [9]–[11]. Studies indicate familial aggregation of ESRD [12]. In African Americans, high risk common variants in the MYH9/APOL1 locus account for much of the excess genetic risk for non-diabetic ESRD compared to their counterparts of European descent. In contrast, comparable genetic risk loci of severe renal phenotypes have not been identified in individuals of European ancestry [13]–[15].
Recently, 16 genetic risk loci associated with estimated glomerular filtration rate (eGFR) and prevalent CKD were identified and replicated by genome wide association studies (GWAS) in about 70,000 individuals of European ancestry in the CKDGen consortium [16], [17]. Two of these loci were also identified by an independent consortium [18]. However, these studies focused on eGFR and prevalent CKD (defined as eGFR <60 ml/min/1.73m2) at one time point, which encompasses the entire spectrum of CKD, and does not does not address the question of whether these genetic factors are involved in the initiation of CKD or in the progression to ESRD, the most advanced stage of CKD. We thus sought to analyze the association of the previously identified 16 eGFR-associated loci with the development of CKD and with ESRD in a total of over 34,000 individuals of European descent.
Results
Association of SNPs with Incident CKD
Overall, 26,308 individuals of European descent, from eight population-based prospective studies, who were free of CKD at baseline were included in the incident CKD analysis (Table 1). At baseline, mean age ranged from 40.5 to 71.7 years. After a median follow-up of 7.2 years, 2122 participants developed incident CKD.
Tab. 1. Cohort characteristics of the incident CKD analysis (n = 26,308). 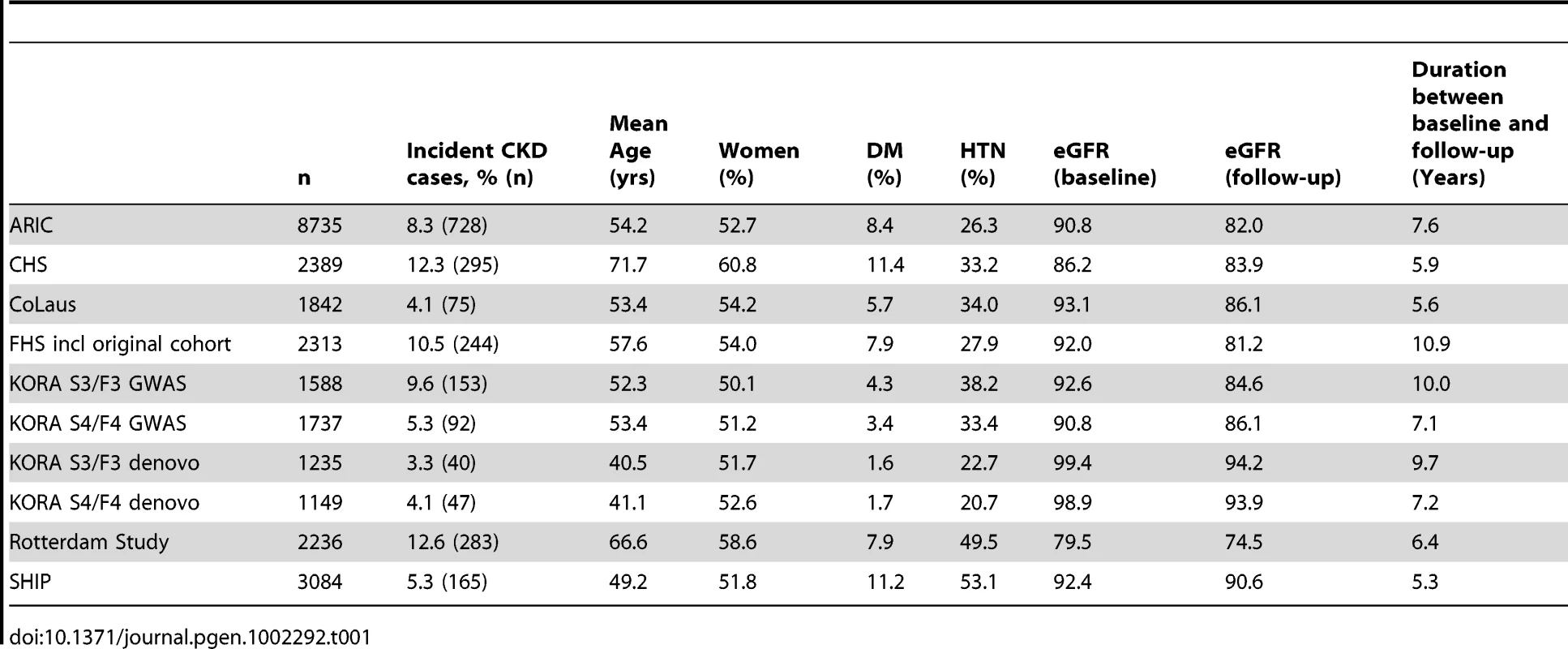
Of the 16 SNPs analyzed, 11 were associated with incident CKD (Table 2): SNPs in UMOD, PRKAG2, ANXA9, DAB2, SHROOM3, DACH1, STC1, SLC34A1, ALMS1/NAT8, UBE2Q2 and GCKR showed p-values ranging from p = 4.1×10−9 in UMOD to p = 0.03 in GCKR. The odds ratios (OR) for incident CKD of the minor alleles at each of the 11 loci ranged from 0.76 per copy of the T allele (allele frequency 18%) at the UMOD locus to 1.19 per copy of the A allele (allele frequency 22%) at PRKAG2. After additional adjustment for baseline eGFR, 6 SNPs (at the UMOD, PRKAG2, ANXA9, DAB2, DACH1 and STC1 loci) remained significantly associated with incident CKD, with minimal attenuation of effect size (Table 2).
Tab. 2. Results for incident CKD and ESRD, CKDGen consortium. 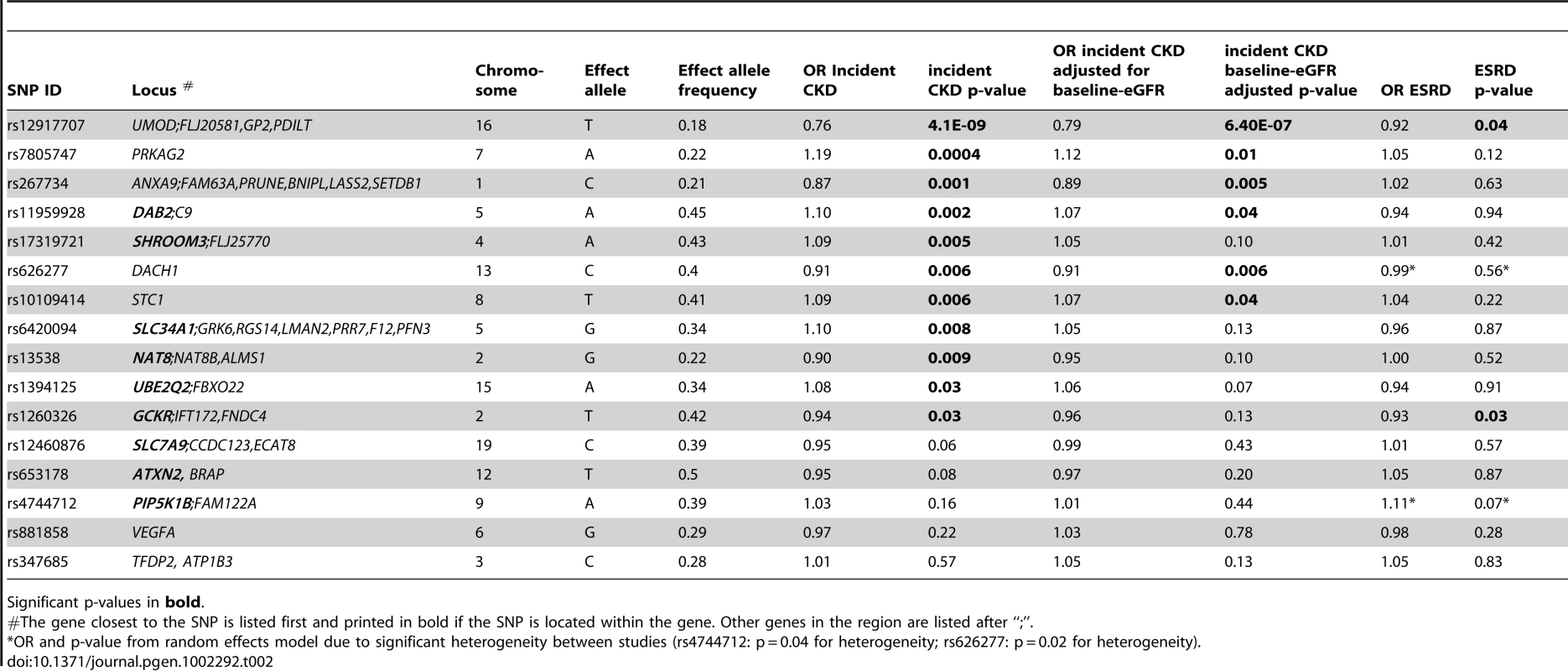
Significant p-values in bold. At each of the significant loci, the direction and the magnitude of the association was similar to those from the discovery analyses of eGFR and prevalent CKD [17]. For example, at the UMOD locus, each copy of the minor T allele at rs12917707 was associated with a 24% reduced risk for incident CKD, while in the CKDGen consortium the same allele was associated with higher eGFR [17]. Though the associations between incident CKD and SNPs in SLC7A9, ATXN2, PIP5K1B and VEGFA were not significant, the direction and magnitude of associations were consistent with our previous findings for the phenotypes eGFR and prevalent CKD [16], [17]. TFDP2 was the only locus where we did not observe association with incident CKD. Of the 16 SNPs tested, 15 had the same direction of association with incident CKD as their original associations with prevalent CKD. The probability of observing this many SNPs with consistency in direction of associations is 0.0002. We did not observe evidence for heterogeneity between studies at any of the 16 loci (test for heterogeneity p>0.05 for all SNPs).
Association of SNPs with ESRD
For the ESRD analysis, we included four case-control studies with a total of 3775 ESRD patients and 4577 controls of European descent without CKD (Table 3). Mean age ranged from 50.7 to 66.2 years in cases and from 47.7 to 62.1 years in controls. Although the direction and magnitude of association for 8 SNPs (at the UMOD, GCKR, PIP5K1B, PRKAG2, STC1, VEGFA, SHROOM3, and ALMS1/NAT8 loci) were consistent with our previous findings for eGFR and prevalent CKD [16], [17], only two SNPs showed nominally significant associations with ESRD (Table 2): rs1260326 in GCKR (OR = 0.93; p-value = 0.03) and rs12917707 in UMOD (OR = 0.92; p-value = 0.04). The lack of association was not likely due to heterogeneity of ESRD cases as only two SNPs showed moderate heterogeneity in their associations with ESRD (Table 2): rs4744712 at the PIP5K1B locus (p = 0.04 for heterogeneity) and rs626277 at the DACH1 locus (p = 0.02 for heterogeneity).
Tab. 3. Characteristics of the ESRD case-control studies (n = 3,775 cases, n = 4,577 controls).§ 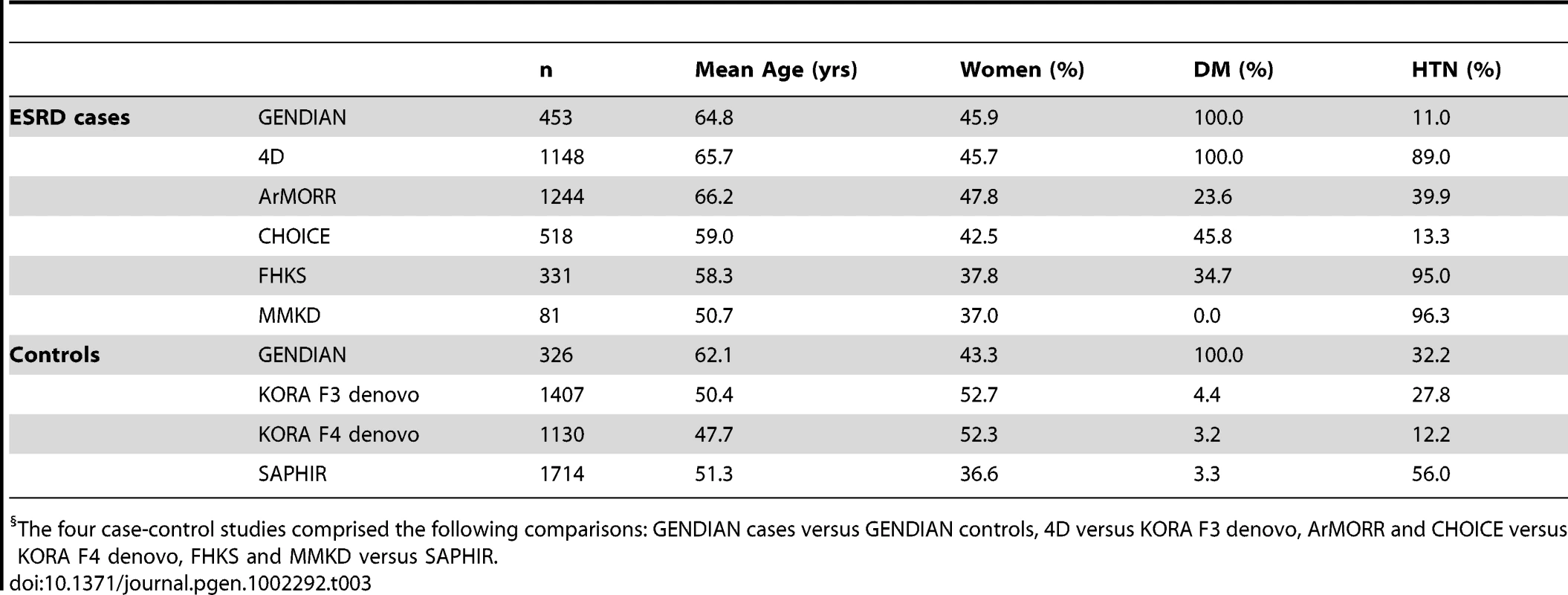
The four case-control studies comprised the following comparisons: GENDIAN cases versus GENDIAN controls, 4D versus KORA F3 denovo, ArMORR and CHOICE versus KORA F4 denovo, FHKS and MMKD versus SAPHIR. Discussion
Among individuals of European Ancestry, most genetic loci associated with the quantitative trait eGFR are also associated with risk for initiation of CKD, with more than half of these associations independent of eGFR at the baseline examination. In contrast, only two SNPs were nominally associated with ESRD.
To date, the genetic loci showing significant and replicated associations with ESRD are limited [13]–[15], [19]–[26], and genetic studies for incident CKD or for renal function decline in established kidney disease are only recently emerging [27]–[29]. The loci we analyzed were identified in association with renal function cross-sectionally and with prevalent CKD by GWAS in the general population. Typical of many SNPs uncovered in GWAS, the majority of these SNPs reside in intronic regions with unknown functional consequences, although several are associated with cis expression levels in liver tissue or leukocytes (Table S3) [16], [17]. These newly identified loci are non-overlapping with those previously identified in individuals of European or Asian descent with advanced diabetic nephropathy [19]–[26], or in African Americans with non-diabetic ESRD [13]–[15].
For the ESRD analysis, we had adequate power to detect effects that were similar to those for prevalent CKD in the discovery GWAS, where odds ratios ranged from 0.8 to 1.19 [16], [17]. In the present study, where associations were observed, the odds ratios for ESRD tended to be smaller and ranged from 0.92 to 1.11. There are several potential explanations for this effect dilution. First, the mechanisms involved in the initiation of CKD, the progression of CKD, and the incidence of ESRD may differ [30]–[33]. Experimental animal data and gene expression profiling in human kidney biopsies suggest differential biological pathways contributing to kidney disease initiation and progression [34]–[36]. Second, the majority of patients with CKD die of cardiovascular disease before developing ESRD [37]–[39]. Thus, the genetic findings for kidney function in the general population may not apply to the highly selected group of dialysis populations. Finally, the process of progression from CKD to ESRD often involves repeated insults including episodes of acute kidney injury by diagnostic and operative procedures and therapies [40]–[43], cardiac function deterioration [44], variation in access to adequate health care [45], [46] and other non-genetic factors [47]. Jointly, these factors may further decrease the relative impact of the small effects of SNPs derived from GWAS of eGFR in the general population at the earliest stage of disease initiation.
The observed small effect sizes for ESRD in our study are in contrast to the large effect sizes observed in relatively small cohorts of individuals of African descent for variants in the MYH9/APOL1 locus, where odds ratios for ESRD ranged from 7.3 for the G1–G2 haplotype at the APOL1 locus to 2.38 for the E1 haplotype in the MYH9 locus [13]–[15]. However, the strong effect at this locus is an exceptional case and may be a consequence of a pronounced positive selection against vulnerability for Trypanosoma brucei rhodesiense infection at the price of a higher susceptibility for non-diabetic ESRD in African Americans not observed in other ethnicities. The establishment of large cohorts is thus needed for performing GWAS of CKD initiation and progression as well as ESRD to overcome the challenge of identifying novel loci significantly associated with these phenotypes with small effect sizes.
The strength of our work lies in the large number of individuals studied. Further, we exclusively analyzed candidate SNPs identified by the unbiased method of GWAS [16], [17]. However, some limitations warrant mention. First, seven of the eight cohorts used for the incident CKD analysis were also part of the CKDGen discovery effort; thus the two samples are not entirely “independent”. However, the phenotype studied differs substantially: in Köttgen et al [17], we used prevalent eGFR data including those with CKD, while follow-up data in those without CKD at the baseline examination was used for the present incident CKD analysis. In the present work, we demonstrate robustness of our findings independent of baseline GFR. Second, we relied on only two serum creatinine measurements to define incident CKD, which may have introduced misclassification and biased our findings towards the null. Third, we did not account for pharmacological treatment with inhibitors of the renin-angiotensin-aldosterone system. Since these drugs may affect kidney function independently of kidney damage, their use may have diluted observable genetic effects [48]. Fourth, our study was not designed to detect fluctuations in eGFR. Furthermore, the etiology of ESRD in the cases we examined may vary between studies, though we observed a low degree of heterogeneity. Finally, our sample consisted of individuals of European ancestry; findings may not be generalizable to other ethnicities.
SNPs associated with eGFR in population-based studies are associated with incident CKD, whereas modest associations were observed with ESRD. Additional work is necessary to characterize the genetic underpinnings across the full range of kidney disease phenotypes, which could ultimately lead to novel diagnostic and therapeutic strategies.
Materials and Methods
Ethics statement
In all studies, all participants gave informed consent. All studies were approved by their appropriate Research Ethics Committees.
Study design and phenotype definition
In population based cohorts, serum creatinine measurements were calibrated to the National Health and Nutrition Examination Study (NHANES) standards in all studies to account for between-laboratory variation across studies, as described previously [10], [16], [17]. Using calibrated serum creatinine, we calculated the estimated glomerular filtration rate (eGFR) with the 4-variable MDRD equation [49].
For incident CKD, we analyzed studies of incident CKD in eight population-based cohorts in the CKDGen consortium with follow-up available: ARIC, CHS, CoLaus, FHS, KORA S3/F3, KORA S4/F4, the Rotterdam Study and SHIP. Each study's design is shown in Text S1. Incident CKD cases were defined as those free of CKD at baseline (defined as eGFR≥60 ml/min/1.73m2) but with a follow-up eGFR<60 ml/min/1.73m2. Controls were those free of CKD at baseline and at follow-up.
For the ESRD analysis, we performed four case control studies of ESRD. Cases were ESRD patients from six cohorts of ESRD patients: CHOICE, ArMORR, GENDIAN, 4D, MMKD and FHKS. Controls were those free of CKD (defined as eGFR≥60 ml/min/1.73m2) in three population-based cohorts (KORA F3, KORA F4, SAPHIR) and one type 2 diabetes cohort (GENDIAN). Each study's design is shown in Text S1.
Statistical methods
In each study, we performed age - and sex adjusted logistic regression of incident CKD, with and without additional adjusting for baseline eGFR, or ESRD status with each SNP. In multicenter studies further adjustment for study-center was performed to account for possible differences between recruiting centers. For family-based studies, we applied logistic regression via generalized estimating equations (GEE) to account for the familial relatedness. Study-specific results were then combined by meta-analysis using a fixed effects model, using METAL (http://www.sph.umich.edu/csg/abecasis/Metal/index.html) [50]. When significant heterogeneity between studies was observed (p for heterogeneity between studies <0.05) we used the random effects model [51]. Statistical significance was defined as a one-sided p-value <0.05 for each SNP without adjustment for multiple testing since all SNPs examined had strong prior probabilities of being associated with the outcomes and the same alleles were hypothesized to be associated with lower eGFR, incident CKD, and ESRD.
Power estimation
We used the QUANTO software for power estimation, assuming an additive genetic model (http://hydra.usc.edu/GxE) [52]. For the ESRD analysis and for SNPs with minor allele frequency ranging from 0.2 to 0.4 we had 80–100% power to detect an OR ≥ 1.10, whereas power was borderline for an OR of 1.05 to 1.09. For example, for the SNP rs12917707 at UMOD, we had 100% power to detect an association with ESRD in the 3775 ESRD cases and 4577 controls assuming that the effect in ESRD would be the same or larger than the effect observed for prevalent CKD previously [16], [17].
Genotyping methods and quality control
For the incident CKD analysis, we used the allele dosage information of each of the 16 SNPs from each study's genome wide data set imputed to HAPMAP CEU samples described previously [17], [18]. Imputation provides a common SNP panel across all studies to facilitate a meta-analysis across all contributing SNPs. Information on each study's genotyping and imputation platform and quality control procedures are shown in Table S1. Table S2 summarizes each SNPs imputation quality.
De novo genotyping of the 16 SNPs was performed in each of the ESRD case-control studies as described previously [17]. Briefly, genotyping was performed either on a MassARRAY system using Assay Design v.3.1.2 and the iPLEX™ chemistry (Sequenom, San Diego, USA) at the Helmholtz Zentrum in Munich, Germany (ArMORR, GENDIAN, 4D, MMKD, FHKS, KORA S3/F3-subset without GWAS data, KORA S4/F4-subset without GWAS data, SAPHIR); by using 5′ nuclease allelic discrimination assays on 7900HT Fast Real-Time Taqman PCR genotyping systems (Applied Biosystems, Foster City, CA, USA) at the Innsbruck Medical University (ArMORR, GENDIAN, 4D, MMKD, FHKS, KORA F3-subset without GWAS data, KORA F4-subset without GWAS data, SAPHIR); or as part of a larger panel of 768 SNPs genotyped on the Illumina Bead Station (CHOICE). The SNPs rs347685, rs11959928, rs4744712 and rs12460876 were not available for de novo genotyping on the Sequenom platform, thus the proxy SNPs rs6773343, rs11951093, rs1556751 and rs8101881, with pairwise r2 of 1.0, 0.87, 0.87 and 1.0 respectively [53], were included in the MassARRAY multiplex PCR.
For the obtained duplicate genotypes (9–22% of the subjects in GENDIAN, 4D, MMKD, FHKS, KORA F3-subset without GWAS data, KORA F4-subset without GWAS data, and SAPHIR; no duplicate genotyping possible due to limited DNA-availability in CHOICE and ArMORR) concordance was 96–100% (median: 100%). SNPs with a per-study call rate <90% or with a per-study HWE p value <0.0001 were excluded from further analysis (rs6773343 and rs653178 in GENDIAN cases; rs13538, rs267734, rs10109414, rs1394125 in ArMORR, rs6773343, rs10109414, rs1556751, rs653178, rs8101881 in CHOICE). In addition, individual samples with <80% successfully genotyped SNPs were excluded from further analysis. After these exclusions, call rates ranged from 91–100% (mean: 98%) across all studies and all SNPs.
Supporting Information
Zdroje
1. LeveyASAtkinsRCoreshJCohenEPCollinsAJ 2007 Chronic kidney disease as a global public health problem: approaches and initiatives - a position statement from Kidney Disease Improving Global Outcomes. Kidney Int 72 247 59
2. MatsushitaKvan der VeldeMAstorBCWoodwardMLeveyAS 2010 Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 375 2073 81
3. BaumeisterSEBögerCAKrämerBKDoringAEhebergD 2010 Effect of chronic kidney disease and comorbid conditions on health care costs: A 10-year observational study in a general population. Am J Nephrol 31 222 9
4. Meguid El NahasABelloAK 2005 Chronic kidney disease: the global challenge. Lancet 365 331 40
5. AdlerAIStevensRJManleySEBilousRWCullCA 2003 Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int 63 225 32
6. HsuCYMcCullochCEDarbinianJGoASIribarrenC 2005 Elevated blood pressure and risk of end-stage renal disease in subjects without baseline kidney disease. Arch Intern Med 165 923 8
7. KastarinenMJuutilainenAKastarinenHSalomaaVKarhapaaP Risk factors for end-stage renal disease in a community-based population: 26-year follow-up of 25,821 men and women in eastern Finland. J Intern Med 267 612 20
8. RitzEStefanskiA 1996 Diabetic nephropathy in type II diabetes. Am J Kidney Dis 27 167 94
9. CoreshJSelvinEStevensLAManziJKusekJW 2007 Prevalence of chronic kidney disease in the United States. JAMA 298 2038 47
10. FoxCSLarsonMGLeipEPCulletonBWilsonPW 2004 Predictors of new-onset kidney disease in a community-based population. JAMA 291 844 50
11. FoxCSMuntnerP 2008 Trends in diabetes, high cholesterol, and hypertension in chronic kidney disease among U.S. adults: 1988-1994 to 1999-2004. Diabetes Care 31 1337 42
12. SatkoSGSedorJRIyengarSKFreedmanBI 2007 Familial clustering of chronic kidney disease. Semin Dial 20 229 36
13. GenoveseGFriedmanDJRossMDLecordierLUzureauP 2010 Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329 841 5
14. KaoWHKlagMJMeoniLAReichDBerthier-SchaadY 2008 MYH9 is associated with nondiabetic end-stage renal disease in African Americans. Nat Genet 40 1185 92
15. KoppJBSmithMWNelsonGWJohnsonRCFreedmanBI 2008 MYH9 is a major-effect risk gene for focal segmental glomerulosclerosis. Nat Genet 40 1175 84
16. KöttgenAGlazerNLDehghanAHwangSJKatzR 2009 Multiple loci associated with indices of renal function and chronic kidney disease. Nat Genet 41 712 7
17. KöttgenAPattaroCBögerCAFuchsbergerCOldenM 2010 New loci associated with kidney function and chronic kidney disease. Nat Genet 42 376 84
18. ChambersJCZhangWLordGMvan der HarstPLawlorDA 2010 Genetic loci influencing kidney function and chronic kidney disease. Nat Genet 42 373 5
19. MaRCTamCHWangYLukAOHuC 2010 Genetic variants of the protein kinase C-beta 1 gene and development of end-stage renal disease in patients with type 2 diabetes. JAMA 304 881 9
20. ShimazakiAKawamuraYKanazawaASekineASaitoS 2005 Genetic variations in the gene encoding ELMO1 are associated with susceptibility to diabetic nephropathy. Diabetes 54 1171 8
21. PezzolesiMGKatavetinPKureMPoznikGDSkupienJ 2009 Confirmation of genetic associations at ELMO1 in the GoKinD collection supports its role as a susceptibility gene in diabetic nephropathy. Diabetes 58 2698 702
22. PezzolesiMGPoznikGDMychaleckyjJCPatersonADBaratiMT 2009 Genome-wide association scan for diabetic nephropathy susceptibility genes in type 1 diabetes. Diabetes 58 1403 10
23. AlkhalafABakkerSJBiloHJGansRONavisGJ A polymorphism in the gene encoding carnosinase (CNDP1) as a predictor of mortality and progression from nephropathy to end-stage renal disease in type 1 diabetes mellitus. Diabetologia 53 2562 8
24. FreedmanBIBostromMDaeihaghPBowdenDW 2007 Genetic factors in diabetic nephropathy. Clin J Am Soc Nephrol 2 1306 16
25. HeBOsterholmAMHoverfaltAForsblomCHjorleifsdottirEE 2009 Association of genetic variants at 3q22 with nephropathy in patients with type 1 diabetes mellitus. Am J Hum Genet 84 5 13
26. ZhangDEfendicSBrismarK Gu HF Effects of MCF2L2, ADIPOQ and SOX2 genetic polymorphisms on the development of nephropathy in type 1 Diabetes Mellitus. BMC Med Genet 11 116
27. KöttgenAHwangSJRampersaudECoreshJNorthKE 2008 TCF7L2 variants associate with CKD progression and renal function in population-based cohorts. J Am Soc Nephrol 19 1989 99
28. LiuMShiSSenthilnathanSYuJWuE 2010 Genetic variation of DKK3 may modify renal disease severity in ADPKD. J Am Soc Nephrol 21 1510 20
29. WheelerHEMetterEJTanakaTAbsherDHigginsJ 2009 Sequential use of transcriptional profiling, expression quantitative trait mapping, and gene association implicates MMP20 in human kidney aging. PLoS Genet 5 e1000685 doi:10.1371/journal.pgen.1000685
30. BrancatiFLWheltonPKRandallBLNeatonJDStamlerJ 1997 Risk of end-stage renal disease in diabetes mellitus: a prospective cohort study of men screened for MRFIT. Multiple Risk Factor Intervention Trial. JAMA 278 2069 74
31. 2002 K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis 39 S1 266
32. BashLDAstorBCCoreshJ 2010 Risk of incident ESRD: a comprehensive look at cardiovascular risk factors and 17 years of follow-up in the Atherosclerosis Risk in Communities (ARIC) Study. Am J Kidney Dis 55 31 41
33. KronenbergF 2009 Emerging risk factors and markers of chronic kidney disease progression. Nat Rev Nephrol 5 677 89
34. PilleboutEBurtinMYuanHTBriandPWoolfAS 2001 Proliferation and remodeling of the peritubular microcirculation after nephron reduction: association with the progression of renal lesions. Am J Pathol 159 547 60
35. ViauAEl KarouiKLaouariDBurtinMNguyenC 2010 Lipocalin 2 is essential for chronic kidney disease progression in mice and humans. J Clin Invest 120 4065 76
36. SchmidHBoucherotAYasudaYHengerABrunnerB 2006 Modular activation of nuclear factor-kappaB transcriptional programs in human diabetic nephropathy. Diabetes 55 2993 3003
37. Al-AlyZZeringueAFuJRauchmanMIMcDonaldJR 2010 Rate of Kidney Function Decline Associates with Mortality. J Am Soc Nephrol 21 1961 9
38. DalrympleLSKatzRKestenbaumBShlipakMGSarnakMJ 2011 Chronic Kidney Disease and the Risk of End-Stage Renal Disease versus Death. J Gen Intern Med 26 379 85
39. AgarwalRBunayeZBekeleDMLightRP 2008 Competing risk factor analysis of end-stage renal disease and mortality in chronic kidney disease. Am J Nephrol 28 569 75
40. BorthwickEFergusonA Perioperative acute kidney injury: risk factors, recognition, management, and outcomes. BMJ 341 c3365
41. KellyKJDominguezJH Rapid Progression of Diabetic Nephropathy Is Linked to Inflammation and Episodes of Acute Renal Failure. Am J Nephrol 32 469 75
42. van KuijkJPFluWJChoncholMHoeksSEWinkelTA 2010 Temporary perioperative decline of renal function is an independent predictor for chronic kidney disease. Clin J Am Soc Nephrol 5 1198 204
43. JamesMTGhaliWATonelliMFarisPKnudtsonML Acute kidney injury following coronary angiography is associated with a long-term decline in kidney function. Kidney Int 78 803 9
44. RoncoCMcCulloughPAAnkerSDAnandIAspromonteN Cardiorenal syndromes: an executive summary from the consensus conference of the Acute Dialysis Quality Initiative (ADQI). Contrib Nephrol 165 54 67
45. WinkelmayerWCOwenWFJrLevinRAvornJ 2003 A propensity analysis of late versus early nephrologist referral and mortality on dialysis. J Am Soc Nephrol 14 486 92
46. WardMM 2009 Access to care and the incidence of end-stage renal disease due to diabetes. Diabetes Care 32 1032 6
47. SoderlandPLovekarSWeinerDEBrooksDRKaufmanJS Chronic kidney disease associated with environmental toxins and exposures. Adv Chronic Kidney Dis 17 254 64
48. MannJFSchmiederREMcQueenMDyalLSchumacherH 2008 Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet 372 547 53
49. LeveyASCoreshJGreeneTStevensLAZhangYL 2006 Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med 145 247 54
50. WillerCJLiYAbecasisGR METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26 2190 1
51. DerSimonianRLairdN 1986 Meta-analysis in clinical trials. Control Clin Trials 7 177 88
52. GaudermanWJ 2002 Sample size requirements for matched case-control studies of gene-environment interaction. Stat Med 21 35 50
53. JohnsonADHandsakerREPulitSLNizzariMMO'DonnellCJ 2008 SNAP: a web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 24 2938 9
Štítky
Genetika Reprodukční medicína
Článek Produces a Long Noncoding RNA and Has General Functions in the Germline, Stem Cells, and BrainČlánek Inference of Relationships in Population Data Using Identity-by-Descent and Identity-by-StateČlánek A Genome-Wide Meta-Analysis of Six Type 1 Diabetes Cohorts Identifies Multiple Associated LociČlánek Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional PolymorphismsČlánek Essential Roles of BCCIP in Mouse Embryonic Development and Structural Stability of ChromosomesČlánek The Receptor Tyrosine Kinase Alk Controls Neurofibromin Functions in Drosophila Growth and LearningČlánek PCNA Ubiquitination Is Important, But Not Essential for Translesion DNA Synthesis in Mammalian CellsČlánek Genome-Wide Association Study Identifies Four Loci Associated with Eruption of Permanent Teeth
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 9- Akutní intermitentní porfyrie
- Růst a vývoj dětí narozených pomocí IVF
- Farmakogenetické testování pomáhá předcházet nežádoucím efektům léčiv
- Pilotní studie: stres a úzkost v průběhu IVF cyklu
- Vliv melatoninu a cirkadiálního rytmu na ženskou reprodukci
-
Všechny články tohoto čísla
- Temporal Trends in Results Availability from Genome-Wide Association Studies
- Association of eGFR-Related Loci Identified by GWAS with Incident CKD and ESRD
- Genetic Variants at Chromosomes 2q35, 5p12, 6q25.1, 10q26.13, and 16q12.1 Influence the Risk of Breast Cancer in Men
- Large-Scale Gene-Centric Analysis Identifies Novel Variants for Coronary Artery Disease
- Genetic Association for Renal Traits among Participants of African Ancestry Reveals New Loci for Renal Function
- Transcriptome Kinetics Is Governed by a Genome-Wide Coupling of mRNA Production and Degradation: A Role for RNA Pol II
- Conserved Regulation of p53 Network Dosage by MicroRNA–125b Occurs through Evolving miRNA–Target Gene Pairs
- Heterozygous Mutations of Are Associated with an Increased Risk of Isolated Metopic Craniosynostosis in Humans and Mice
- Study of FoxA Pioneer Factor at Silent Genes Reveals Rfx-Repressed Enhancer at and a Potential Indicator of Esophageal Adenocarcinoma Development
- Cholesterol Metabolism Is Required for Intracellular Hedgehog Signal Transduction
- Produces a Long Noncoding RNA and Has General Functions in the Germline, Stem Cells, and Brain
- Age-Dependent Recombination Rates in Human Pedigrees
- Sequence Conservation and Functional Constraint on Intergenic Spacers in Reduced Genomes of the Obligate Symbiont
- Sex Chromosome Mosaicism and Hybrid Speciation among Tiger Swallowtail Butterflies
- A Negative Feedback Loop That Limits the Ectopic Activation of a Cell Type–Specific Sporulation Sigma Factor of
- Phased Whole-Genome Genetic Risk in a Family Quartet Using a Major Allele Reference Sequence
- Mutations in or near the Transmembrane Domain Alter PMEL Amyloid Formation from Functional to Pathogenic
- Inactivation of Alters Melanosome Shape But Has Only a Subtle Effect on Visible Pigmentation
- Novel Interactions between Actin and the Proteasome Revealed by Complex Haploinsufficiency
- Germline Genetic Variants Disturbing the /LIN28 Double-Negative Feedback Loop Alter Breast Cancer Susceptibility
- Separation of Recombination and SOS Response in RecA Suggests LexA Interaction Sites
- Inference of Relationships in Population Data Using Identity-by-Descent and Identity-by-State
- Misregulation of Scm3p/HJURP Causes Chromosome Instability in and Human Cells
- A Noncoding Point Mutation of Causes Multiple Developmental Malformations and Obesity in Twirler Mice
- The Evolutionarily Conserved Longevity Determinants HCF-1 and SIR-2.1/SIRT1 Collaborate to Regulate DAF-16/FOXO
- A Genome-Wide Metabolic QTL Analysis in Europeans Implicates Two Loci Shaped by Recent Positive Selection
- Bacterial Communities of Diverse Species: Ecological Context of a Host–Microbe Model System
- A Genome-Wide Meta-Analysis of Six Type 1 Diabetes Cohorts Identifies Multiple Associated Loci
- Elongator Complex Influences Telomeric Gene Silencing and DNA Damage Response by Its Role in Wobble Uridine tRNA Modification
- Elevated Proteasome Capacity Extends Replicative Lifespan in
- Genome-Wide Analysis of Heteroduplex DNA in Mismatch Repair–Deficient Yeast Cells Reveals Novel Properties of Meiotic Recombination Pathways
- MicroRNA Predictors of Longevity in
- An Iterative Genetic and Dynamical Modelling Approach Identifies Novel Features of the Gene Regulatory Network Underlying Melanocyte Development
- Atypical AT Skew in Firmicute Genomes Results from Selection and Not from Mutation
- Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional Polymorphisms
- Genomic Analysis of QTLs and Genes Altering Natural Variation in Stochastic Noise
- The Abnormal Phenotypes of Cartilage and Bone in Calcium-Sensing Receptor Deficient Mice Are Dependent on the Actions of Calcium, Phosphorus, and PTH
- Cell Type–Specific Transcriptome Analysis Reveals a Major Role for and miR-200b in Mouse Inner Ear Morphogenesis
- Essential Roles of BCCIP in Mouse Embryonic Development and Structural Stability of Chromosomes
- IAP1-Mediated Ubiquitylation Controls Activation of the Initiator Caspase DRONC Independent of Protein Degradation
- VANG-1 and PRKL-1 Cooperate to Negatively Regulate Neurite Formation in
- The Receptor Tyrosine Kinase Alk Controls Neurofibromin Functions in Drosophila Growth and Learning
- Comparative and Functional Genomics of PD630 for Biofuels Development
- Identification of Type 1 Diabetes–Associated DNA Methylation Variable Positions That Precede Disease Diagnosis
- PCNA Ubiquitination Is Important, But Not Essential for Translesion DNA Synthesis in Mammalian Cells
- Genetic Effects at Pleiotropic Loci Are Context-Dependent with Consequences for the Maintenance of Genetic Variation in Populations
- Genome-Wide Association Study Identifies Four Loci Associated with Eruption of Permanent Teeth
- Bmp and Nodal Independently Regulate Expression to Maintain Unilateral Nodal Activity during Left-Right Axis Specification in Zebrafish
- Inter-Allelic Prion Propagation Reveals Conformational Relationships among a Multitude of [] Strains
- Emergence and Modular Evolution of a Novel Motility Machinery in Bacteria
- Histone Methyltransferase MET-2 Shields the Male X Chromosome from Checkpoint Machinery and Mediates Meiotic Sex Chromosome Inactivation
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional Polymorphisms
- The Evolutionarily Conserved Longevity Determinants HCF-1 and SIR-2.1/SIRT1 Collaborate to Regulate DAF-16/FOXO
- Genome-Wide Analysis of Heteroduplex DNA in Mismatch Repair–Deficient Yeast Cells Reveals Novel Properties of Meiotic Recombination Pathways
- Association of eGFR-Related Loci Identified by GWAS with Incident CKD and ESRD
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



