-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Obstructive sleep apnea in relation to orthodontic treatment in children
Authors: Z. Vranková 1; P. Turčáni 2; P. Horník 3; A. Bryšová 1; L. Izakovičová Hollá 1; P. Černochová 1; P. Bořilová Linhartová 1,4
Authors place of work: Stomatologická klinika, Lékařská fakulta Masarykovy univerzity a Fakultní nemocnice u sv. Anny, Brno 1; Klinika nemocí plicních a tuberkulózy, Lékařská fakulta Masarykovy univerzity a Fakultní nemocnice Bohunice, Brno 2; Dětské ORL, s. r. o., Brno 3; Ústav patologické fyziologie, Lékařská fakulta Masarykovy univerzity, Brno 4
Published in the journal: Česká stomatologie / Praktické zubní lékařství, ročník 120, 2020, 1, s. 13-25
Category: Přehledový článek
Summary
Introduction, aim: Obstructive Sleep Apnea (OSA) is defined as the collapse of the upper respiratory tract, complete or partial, leading to hypopnea to apnea. This collapse is repeated several times during the night. As this sleep-related breathing disorder (SRBD) manifests itself differently in adults and in children, there is also a separate diagnosis of OSA occuring in childhood. For most children, we can see loud night snoring, mouth breathing, disturbed sleep continuity and its architecture, and therefore OSA has an impact not only on the physical but also on the child's mental health and development. Correct diagnosis, in the most precise way with polysomnography and subsequent selection of therapy should be done as soon as possible. The risk factors for OSA in children include obesity, adenotonsillar hypertrophy, neuromuscular diseases or craniofacial malformations. The aim of the review article is to outline the relationship between OSA and orthodontic anomalies in children and to summarize current trends in their therapy.
Cephalometric studies in pediatric patients with OSA indicate that there are several craniofacial features that can interfere with upper respiratory tract patency and contribute to the onset or aggravation of this disorder. These include, for example, a vertical growth pattern and increased basal bones divergence, often visible on the frontal open bite and the lack of the lip seal. In patients, we often describe a second skeletal class with increased overjet, the jaws may be in retrognathic position, and the anterio-posterior dimension of the bony portion of the nasopharynx is therefore affected and reduced. The micrognathism or microgenia, narrowed nasomaxillary complex, high and narrow palate, often manifested by lateral crossbite, is also increasing the risk of OSA. The effect of agenesis or early tooth loss is being examined.
An imbalance in the development of orofacial structures in early childhood can lead to a disruption of the normal development of the bony support of the upper respiratory tract and therefore cause an increased risk of the development of SRBD.
Conclusion: Properly timed orthodontic treatment for children with dysmorphism leading to narrowed upper respiratory tract can stimulate the growth of the jaws, improving their proper development while reducing the risk of upper respiratory tract collapse. Orthodontic treatment should thus be considered as an alternative treatment for OSA children with craniofacial anomalies, however the most important aspect of therapy is an interdisciplinary approach.
Keywords:
polysomnography – pediatric obstructive sleep apnea – sleep related breathing disorders – craniofacial features – dysmorphism – orthodontic treatment
PEDIATRIC OBSTRUCTIVE SLEEP APNEA, SYMPTOMS AND PREVALENCE
Obstructive sleep apnea (OSA) is asleep-related disorder. The basic concepts of these disorders are defined in Fig. 1 [1–4].
Tab. 1. Basic definitions often used in the problematics of sleep-related breathing disorders 
Several studies point to a different OSA manifestation and management in children and in adult patients. In adults, risk factors include overweight to obesity with central fat distribution, a wide neck circumference, and abnormalities in the upper respiratory tract. Menopause, smoking or alcohol before sleep are considered to be suspected risk factors. Symptoms include also loud snoring or cough and are manifested mainly by excessive daytime sleepiness, drowsiness and psychosocial problems [5].
In 1976, pediatric OSA was first described as a diagnosis, based on its specific symptoms [6]. In children, the most common symptoms are daytime fatigue, sleepiness or hyperactivity, inability to concentrate and t related worse school results, antisocial, problematic behaviour and personality changes. The clinical symptoms were mainly hypertension, acute cardiac or cardiopulmonary failure and frequent upper respiratory tract infections. In most cases loud snoring, oral breathing, disturbed sleep continuity and its architecture, nightmares, waking, sweating, insomnia, wetting, sleepwalking, depression, and other problems were reported [6-12]. Given the above-mentioned manifestations, it is obvious that OSA affects not only the physical but also mental health and psychological development of the child. For this reason, OSA should be diagnosed and treated promptly.
In addition, a special category of pediatric OSA occurs in infants due to their different anatomical and physiological predisposition to abnormalities in respiration or obstructions (e.g. higher position of larynx, highly compliant chest wall, impairment in ventilation and perfusion or unstable gas exchange control) [13].
The prevalence of sleep-related breathing disorders in children is likely to be higher than previously thought due to a lack of standards for their diagnosis [14, 15].
MECHANISM OF OSA IN CHILDREN
The control of the pharyngeal muscle tone and its reflex responses differ during wakefulness and sleeping. If changes occur, the pharynx could get critically narrow, increasing the resistance in the upper airways and causing it to collapse more quickly. At night, also the horizontal position contributes to easier airway collapse. Although the pharynx is able to alter its lumen, its diameter is also affected by the surrounding bone structures, whose anatomy may also contribute to their easier collapse [16].
Apnea and hypopnea, which are repeated several times during OSA, are terminated by arousal, reaction based on a steadily increasing inspiratory muscle effort during apnea. Further, the induced hypoxia and hypercapnia is detected by chemoreceptors, which thus contribute to this reaction. Apnea is terminated by partial or complete awakening and airway resistance decreases again. However, this awakening causes sleep fragmentation, thus disturbing its quality and quantity, with all the consequences that transmit to the individual's life [17].
RISK FACTORS
Risk factors for the development of OSA in children are complex Various studies include obesity, adeno-tonsillar hypertrophy associated with local infection or inflammation, neuromuscular factors such as abnormally reduced muscle tone during sleep.
The family incidence of OSA and twin-studies also point to a genetic background; the estimated proportion of the genetic component is 35–40%. It is believed that genes related to the development of craniofacial structures, body fat distribution, and nerve control of the upper respiratory tract muscles, i.e. pharyngeal muscles, are involved in the etiopathogenesis of OSA [18]. The epigenetic effects may also be an important determinant of the inflammatory phenotype in OSA [19]. In severe OSA, increased numbers of Streptococcus, Prevotella and Veillonella were found, compared to nasal microbiome in healthy subjects. A specific composition of oral microbiome has also been associated with inflammatory biomarkers in OSA patients [20].
The OSA risk factors in children that can be diagnosed by dentists include short sublingual frenulum, mouth breathing, agenesis or premature tooth loss and craniofacial malformations [21–23]. Of course, the risk of developing OSA in children increases with a combination of several concurrent risk factors, as schematically shown in Fig. 1, and with their increasing severity.
Fig. 1. Summary of factors often occurring in children suffering from OSA 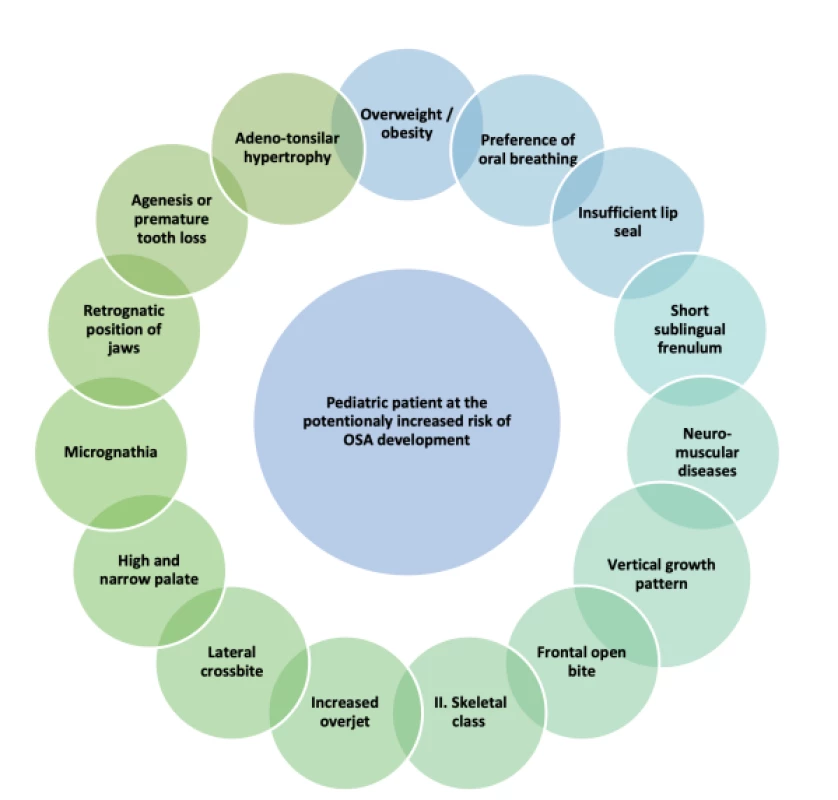
Short sublingual frenulum
The sublingual frenulum, fold of mucous membrane, that connects the tongue and the floor of the oral cavity, becomes physiologically thinner after birth. However, if the thinning is not sufficient, it can remain short and firm, preventing full language movements, especially elevation. The lightest form usually does not require surgery because the muscles of the tongue are well formed, and the tongue is movable. The second form is the middle-short sublingual frenulum, a firm fibrous strip between the caruncles and the tongue; the tongue cannot be completely dislodged. The left and right half of the tongue may contain scar tissue and surgical relaxation is fully indicated. The most serious form of the disorder is the tongue fully attached to the floor of the oral cavity, with a broad base of muscle attached to the lower incisors, a minimum of mucous membrane and without coordination between tongue movement and suction. Plastic surgery is required already in neonatal or infant age [24]. If in the indicated case the frenulum is not released in time, functions such as sucking, swallowing, chewing or speech, which are important for the proper development of craniofacial structures, are impaired. Further, an insufficient stimulation of the mid-palatal suture may lead to abnormal growth of the maxilla, lateral cross bite, disproportion in mandibular growth, or retrognation, thus creating OSA-associated traits in children [24, 25]. In another study, the authors point out that only the removal of short frenulum may not be sufficient and may not always result in automatic or complete respiratory adjustment. Therefore, it is sometimes advisable to supplement this procedure with myofunctional therapy, especially in older children [26].
In a study from 2016 published by Guilleminault et al., comprised 150 children with OSA, 63 of them had a short sublingual frenulum and without tonsil hypertrophy and 87 of them had a physiological sublingual frenulum but suffered from tonsil hypertrophy. In children suffering from OSA and short sublingual frenulum, abnormal anatomical findings in the form of a high and narrow palate were often apparent. 60 of them had a positive family history of short frenulum [28].
Oral breathing
Oral breathing results in a non-physiological shape of the airways during respiration and in its reduction by changing the pharyngeal lumen. Not only does it increase resistance in upper respiratory tract, it also contributes to the formation of microtrauma in the posterior part of throat, which may induce an inflammatory response and consequently more pronounced hypertrophy of adenoids [26, 29].
The combination of insufficient stimulation of bone growth and absence of nasal respiration with the preference of oral breathing results in impaired development of the orofacial skeletal structures, again increasing the risk of upper airways collapse during sleep. This has been confirmed by experimental data with artificially induced nasal obstruction in primates in which the neuromuscular changes that were necessary for the induction and maintenance of oral respiration resulted in a change in skeletal and soft tissue morphology. However, the data showed considerable individual variation in response to an identical stimulus [30–33].
Agenesis and premature tooth loss
Many authors focus on the influence of agenesis or early tooth loss on the development of the craniofacial structures and possible OSA development. It is agreed that the loss of teeth or their absence due to agenesis affect the anatomy of the alveolar process. Irrespective of why the teeth are missing, abnormal orofacial anatomy is described in children in a form that is at risk for collapse of upper respiratory tract in sleep [16, 34, 35].
Craniofacial features
In early childhood some basic functions such as nasal breathing, swallowing, mastication and sucking are very important for the development of the orofacial structures. The skeletal parts of these structures have a great influence on the upper respiratory tract dimension. Their anatomy could be classified as one of the key factors for respiration, especially in sleep, where without sufficient bone support and airway width, the respiratory tract could collapse and occlude more easily.
The development of the transversal part of the nasomaxillary complex is affected by the development of the nasal cavity, the eye sockets and the activity of the intermaxillary suture. Postnatally active intermaxillary suture and alveolar process formation are under the constant influence of the above mentioned activities, whose correct function is again due to the connected with the correct position of the teeth and the activity of the facial and tongue muscles involved in these activities, affecting thus the correct development of the orofacial region [30].
In connection with the development of pediatric OSA, a so-called concept of dysfunction, which may lead to dysmorphism affecting the airway size, is also mentioned. The authors of this concept suggested that an imbalance in the development of orofacial structures in early childhood leads to disruption of the normal development of bone support of upper respiratory tract thus increasing the risk of collapse and development of SRBD. This concept highlights the negative effect of treatment that is not directed at dysfunction leading to OSA, and the need to address the cause of this disorder [30].
Several cephalometric studies in pediatric patients with OSA have shown characteristic but diverse craniofacial features that may affect the patency of upper respiratory tract and thus contribute to OSA. The most common features include: mandibular posteriorotation, vertical growth pattern, often manifested by frontal open bite and insufficient lip seal, retrognathism, maxillo-mandibular micrognathia, leading to reduced anterio-posterior dimension of the bone nasopharynx. The nasomaxillary complex is narrow, constricted, hard palate is often V shaped or often it is so-called Gothic palate. In these patients, skeletal class II, sometimes combined with Angle class II and more pronounced overjet is often identified, laterally we can often see crossbite [13, 30, 36–50]. Fig. 2 shows the cephalometric analysis used by Min Gu et al, for the evaluation of the upper airways in their study conducted in 2014 [51]. Fig. 3 shows a lateral cephalogram made in a pediatric patient at an initial orthodontic examination, where arrows indicate the risk signs for OSA. In Fig. 4–6, intraoral images of patients with the aforementioned craniofacial features risky for SRBD development are presented.
Fig. 2. Cephalometric analysis and UR tract measurement used in the determination of cephalometric standards in 12-year-olds; taken from Min GU et al. [51] Po – porion, Or – orbitale, ANS – anterior nasal spine, PM – pterygo-maxillare, U – uvula, UPW – upper pharyngeal wall, MPW – middle pharyngeal wall, LPW – lower pharyngeal wall, V – vallecula, AH – anterior hyoid, C2 and C3 - 2nd and 3rd cervical vertebrae, FH – Frankort horizontal plane, NL – nasal line, CV – cervical vertebrae, the line joining the C2 and C3 ![Cephalometric analysis and UR tract measurement used in the determination of cephalometric standards in 12-year-olds; taken from Min GU et al. [51]
Po – porion, Or – orbitale, ANS – anterior nasal spine, PM – pterygo-maxillare, U – uvula, UPW – upper pharyngeal wall, MPW – middle pharyngeal wall, LPW – lower pharyngeal wall, V – vallecula, AH – anterior hyoid, C2 and C3 - 2nd and 3rd cervical vertebrae, FH – Frankort horizontal plane, NL – nasal line, CV – cervical vertebrae, the line joining the C2 and C3](https://www.prolekare.cz/media/cache/resolve/media_object_image_small/media/image_pdf/b7453c51f71b3b7e18c1ecae8380cc8a.jpeg)
Fig. 3. Lateral cephalogram – arrows pointing to risk factors: insufficient lip incompetence (1), enlarged overjet (2), vertical growth pattern (3), tonsila lingvalis (4) adenoid hypertrophy (5) 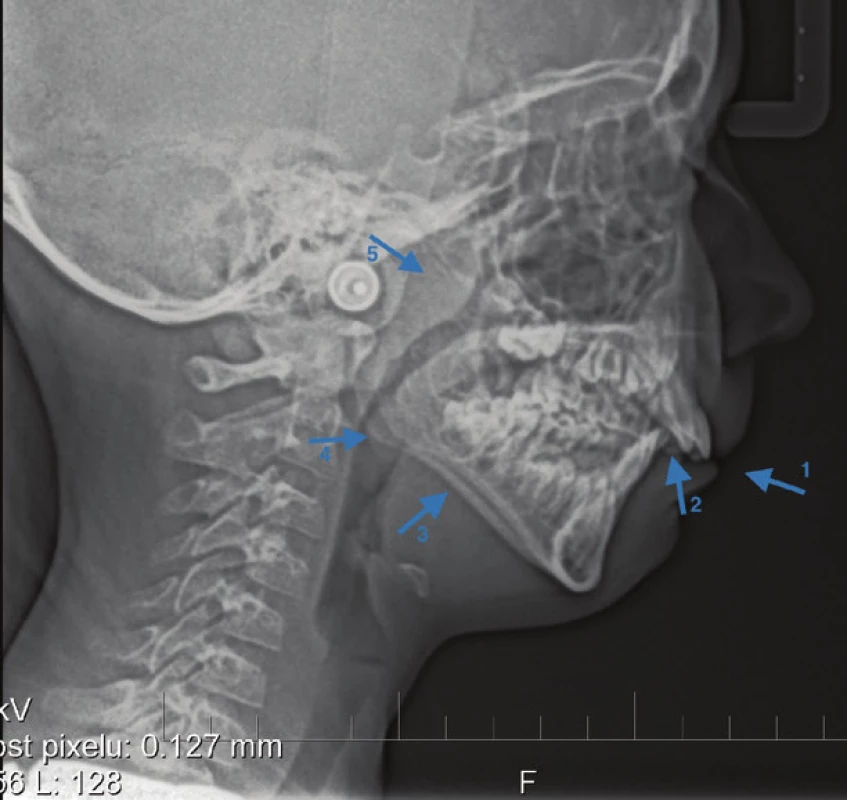
Fig. 4a Transverse maxillary deficiency – occlusal view 
Fig. 4b Posterior crossbite – right side view 
Fig 4c Posterior crossbite – left side view 
Fig 4d Bilateral cossbire – frontal view 
Fig. 5a Narrowing of the upper jaw – tendency to open bite 
Fig. 5b Narrowing of the upper jaw – tendency to open bite – side view 
Many studies reporting an increased frequency of OSA in children with congenital craniofacial malformation have been published. Driessen et al. devote their studies to children with syndromic craniosynostosis and MacLean et al. to pediatric patients with cleft of the lip and/or palate, prior to surgery. Both studies supported the association between craniofacial malformations and OSA in children [21, 22]. Craniofacial growth may be influenced by the presence of genetically conditioned syndromes associated with sleep disorders (Appert syndrome abd Crouzon syndrome, Treacher Collins syndrome or Pierre Robin syndrome) [52, 75].
SCREENING OF PEDIATRIC OSA IN DENTIST'S / ORTHODONTIST'S OFFICE AND POSSIBILITIES OF OSA DIAGNOSTICS
Appropriate tools for screening of the population and detecting an increased risk of OSA include a questionnaire as well as the use of imaging methods. These are within the capabilities of the dentist / orthodontist. Therefore, simple procedures and commonly used devices can be used also for detecting OSA and subsequently, in case of suspicion of this disorder, patients can be sent for detailed examination to be specialized workplaces. According to the recommendations of the Czech Society for Sleep Research and Sleep Medicine, the only reliable diagnostic method of OSA in children is overnight videopolysomnography in combination with examinations atd a somnologist and a ENT specialist, neurologist, pneumologist, pediatrician, and/or dental surgeon [53].
Questionnaires intended for children or parents of children
There are several methods for the detection of sleep disturbances, including questionnaires, for example : The Sleep-Related Breathing Scale of the Pediatric Sleep Questionnaire, an 18-item Obstructive Sleep Apnea QoL, which is rated by several studies as a questionnaire with good reliability, and Epworth Sleepiness Scale, which is probably the most commonly used in the Czech Republic, existing both in classic adults version but also in a modified version for children [54–57]. It consists of several questions that the patient or his / her legal representative evaluates by assigning a numerical value to a statement. This questionnaire is understandable, simple and quick and for these reasons it is used for OSA screening. At the same time, however, it is not very complex, it consists of only eight questions. In the Czech Republic no other questionnaire usable for screening OSA in the pediatric population exists.
Although questionnaires are an efficient and simple screening method, Constantin et al. in their study warned against their use in place of objective tests to detect OSA in children [58].
Imaging methods
Cephalogram, a picture showing the skull in a lateral projection, is an easily accessible and commonly used image for orthodontic examination. The orthodontist, who describes this picture comprehensively, is able to reveal important predictors of OSA, especially in pharyngeal dimensions [59, 45]. Since obstruction in OSA can occur anywhere within the airways, i.e. from the tip of the nose to the larynx, this image has some limits. Changes that occur in the transversal dimension cannot be shown [60]. It is also limited by its 2-dimensionality (2D). 3D imaging, Cone Beam computed tomography (CBCT) and magnetic resonance imaging (MRI), allows 3D segmentation and analysis of different parts. Thus, the functional relationships between skeletal, oral, and pharyngeal dimensions in patients can be better assessed, and possible airway obstruction as well as the exact place of constriction can be identified, thus allowing to solve the causes of the obstruction effectively [42, 58, 59]. For each imaging, it is important to achieve anatomical and functional imaging with the least necessary radiation burden on the body, which is currently possible in CBCT imaging [60, 63, 64].
Diagnostic tools to determine OSA in children – polysomnography, sleep monitoring
There are several ways to diagnose OSA in children. The more detailed and sensitive the examination is, the more difficult it is to apply it in a wider population.
The most accurate examination of OSA is polysomnography [60]. According to Tsar et al. this examination may be performed at any age of the patient. This six-channel device developed by the Department of Clinical Neurophysiology of Oulu University Hospital, records: respiratory movements, oronasal airflow, oxygen saturation of hemoglobin, terminal exhaled carbon dioxide, electrocardiogram, electroencephalogram, electrooculogram, and chin and tibia electromyogram. Combined analysis of these parameters can lead to very accurate diagnosis [3, 45]. However, due to the complexity of the equipment and the examination, it is often unavailable. While PSG is still considered the gold standard for OSA diagnostics, home sleep monitoring devices (such as the ApneaLink Air or SomnoCheck Micro Cardio device) are a suitable alternative. Although, compared to overnight PSG, these devices do not provide so complex records, more and more studies have identified them as cost-effective compared to PSG, more accessible and sufficiently effective in the early OSA capture [65–67].
OSA THERAPY IN PEDIATRIC PATIENTS
In history tracheostomy, later a domestic positive airway pressure devices for home testing were used to treat OSA, which however were not always tolerated well tonsilo-adenotomy was also common treatment. However, SRBD may not be fully cured after these procedures, despite a significant improvement immediately after surgery [68–70]. Of course, these therapies are still indicated in some cases, but due to the variability of the factors that cause OSA and contribute to this disorder, recently the opinion prevails that it is difficult to speak of universal therapy for all pediatric patients. Early OSA diagnose is important, therapy should then focus on the factor that causes it in a particular individual. For this reason, interdisciplinary cooperation, including the above-mentioned expertise, and complex examination of a patient are crucial [30, 71].
Influence of orthodontic therapy in OSA management in children
The above described craniofacial features frequently found in pediatric OSA patients often bring children to the orthodontist, and because orthodontic therapy affects orofacial structures, it also interferes with the bone support of upper respiratory tract. Change of its dimension can affect positively or even eliminate OSA. Several studies have confirmed that orthodontic therapy may lead to its elimination in some children with OSA without the need for tonsiloadenotomy [72–74].
Myofunctional therapy (MFT)
The correct positioning of the tongue, achieved by MFT, has been described since 1918 as a major factor leading to stimulation of mandibular growth and nasal breathing [75]. The amount of data collected over several years suggests an association between the orofacial muscle tension and the physiological development of the nasomaxillary complex and mandible. Disruption of the normal tone, hypotonia of the tongue, was associated with oral breathing or incorrect position of the tongue and led to defect in the development of the orofacial complex. Understanding the importance of MFT is related to understanding the relationship between linking the muscle activity of the tongue to the orofacial muscles and the development of anatomical structures that affect the airways [76].
MFT is a collection of isotonic and isometric exercises affecting the lips, tongue, oropharyngeal structures such as the soft palate or lateral pharynx. Its task is to ensure correct head posture, position of tongue on the palate against upper teeth, swallowing, mastication, speech and articulation. Parental activity is required for its proper implementation in children. Based on the results of several meta-analyses, Huang reported in his study that active and daily MFT in conjunction with other treatments may lead to complete remission of OSA in up to 60% of children [16]. Conversely, the absence of MFT may be associated with recurrence of SRBD [67].
According to the meta-analysis performed in 2015, which included nine studies on adult patients and two pediatric studies, MFT can reduce AHI in approximately 50% of adult and 62% of children patients. It also appears to be an appropriate complementary therapy to other OSA treatment methods [78].
Passive MFT
Although MFT belongs to the therapies recommended in OSA, frequently occurring problems with their observing in pediatric patients, lead to passive MFT in the form of night-time orthodontic appliances.
Chuang et al. investigated the influence of the Monoblock, a device used to advance the mandible, which is also modified in its lower part by an inserted bead. The appliance was worn for the night, and patients were asked to roll the bead with the tongue, this led the tongue to the forward position, thus opening the airways. Its construction was based on the assumption that the bead near the tip of the tongue would stimulate its activity at least during the lighter phase of sleep, and this activity generally would lead to the correct function and position of the tongue and proper development of the oral cavity. After six months of passive therapy a significant decrease in AHI and waking at night was observed [79].
Maxillary expansion
Within orthodontic therapy, maxillary expansion or rapid maxillary expansion (RME) is a relatively frequent procedure. It was first mentioned by E. C. Angell in 1860 and is nowadays one of the frequently used therapies applied in transversal narrowing of the maxilla [80].
Since reduced transverse maxilla width was often found in children at high risk of SRBD, RME could help to improve their quality of life, as confirmed by several studies. It is agreed that the RME expands the maxilla, enlarges the dental arch and increases intranasal capacity, widens the nasal cavities and reduces nasal resistance. Thus, in patients suffering from maxillary constriction and OSA symptoms, RME has been shown to improve nasal respiration and offer a useful therapy [81–92]. To clarify its impact on the tongue position and volume of the pharyngeal part of the airways, CBCT was used in some studies, also in combination with computational fluid dynamics (CFD) analyses before and after treatment. In children with nasal obstruction, RME has been shown not only to reduce this obstruction, but also to increase the position of the tongue and enlarge the pharyngeal part of the airways [93, 94]. CBCT results also confirmed a significant expansion of the anterior and posterior nasal floor and an increase in nasopharyngeal and nasal cavity volume after palate expansion [95].
Pirelli et al. observed after the RME the expansion of the maxilla and the decrease of the AHI index from 12 to <1 already after 4 months, anterior rhinometry was also normal [73, 74]. In another study of 42 children without enlarged tonsils or adenoids, who had a narrowed upper jaw and BMIs below 24, the authors demonstrated a significant improvement in OSA and increased airway patency after RME [94]. Follow-up studies reported a decrease in AHI after RME, which remained consistently normalized over time. The stability of the obtained expansion was demonstrated by measuring the width of the maxilla base and the distance of pterygoid processes by CT imaging after 24 months and 12 years follow-up [74, 96, 97]. Cistulli et al. also confirmed that with the maxilla width increase, nasal resistance reduced and AHI in OSA patients declined [92]. Similarly, the authors of a meta-analysis published in 2016 reported that RME was an effective therapy in children with OSA [98].
Lower jaw position and skeletal therapy II. Class
In children suffering from OSA and skeletal class II combined with Angle class II several different orthodontic appliances and therapies for changing the position of the lower jaw canbe used. In children aged 8–14 treated with the Twin Block appliance to advance the lower jaw, the sagittal dimension of the oro - and hypopharynx increased. The correction of the position of the mandible also improved the length, thickness, and slope of the soft palate [99]. Another study reported that after using this appliance for approximately 10.8 months, OSA symptoms as well as patient profile and PSG results improved. Cephalometric analyses have shown significant enlargement of the airway space, change in the angle between mandible plane and cranial base, and facial convexity, suggesting an increase in mandible growth and change in soft palate length [100]. An increased airway volume in the oropharynx and laryngeal compartments and a reduced rate of awakenings in children with skeletal class II and OSA have also been studied after the treatment with the Herbst appliance [101, 102]. According to a meta-analysis performed in 2005 that consisted of 178 articles, mandibular distraction osteogenesis, a surgical method used to extend the mandible, is an effective therapy for craniofacial deformities. A decrease in symptoms was observed in 97% of pediatric patients and 100% of adult patients with OSA [103]. A more recent meta-analysis from 2018, which included 98 studies, showed that pediatric patients suffering from mandibular undergrowth, experienced a dramatic improvement in OSA after treatment with mandibular advancement or distraction osteogenesis [104].
Combination therapy
In many patients, combination of several therapeutic principles in orthodontic therapy is needed. When the mandible is advanced, the width of both dental arches must be adjusted accordingly, the upper dental arch must be often enlarged. Positive results of such combination therapy are confirmed by several studies. Schütz et al. have reported improved breathing and reduced OSA symptoms in post-treatment patients using both maxillary expansion and mandibular advancement by the Herbst appliance [105]. The result is confirmed by a case report describing the treatment of a single child patient suffering from OSA, with an orthodontic device called Sleep Apnea Twin Expander, which simultaneously performs palate expansion and mandibular advancement. After the orthodontic treatment, the OSA-18 questionnaire showed an improvement in the main respiratory symptoms, and even a home sleep study revealed a reduction in obstructive apneic events. After treatment, clinical evaluation and cephalometric analysis, a reduction in maxillo-mandibular discrepancy was observed in the sagittal and vertical planes and an extension of upper respiratory tract was also confirmed. This case report suggests that orthodontic treatment may be a valuable treatment in OSA children with craniofacial anomalies [106].
INTERDISCIPLINARY COOPERATION IN OSA MANAGEMENT IN CHILDREN
Interdisciplinary cooperation is important in the diagnosis and the therapy of pediatric OSA. If a general practitioner/orthodontist suspects an increased risk of OSA, he/she should investigate the patient thoroughly within his/her expertise and subsequently propose examination by other specialists as recommended by the Czech Society for Sleep Research and Sleep Medicine [53]. Note. A dental surgeon as one of the specialists in OSA management in children [53] could be replaced by an invitation to cooperate with a dentist/orthodontist (especially if they are familiar with OSA) as dental surgery of craniofacial abnormalities in growing pediatric patients is not performed, with the exception of distraction osteogenesis, mentioned in the previous chapter. Therefore, the dental surgeon usually does not get into contact with pediatric patients. It is advisable to ask for cooperation of a speech therapist and a rehabilitation worker or a specialist in the myotherapy of the orofacial system. By analysing all the obtained information from the individual examinations, the diagnosis can be correctly established, and an appropriate/adequate therapy chosen to eliminate the most likely cause of OSA, as well as other factors that may aggravate or recur the disease.
Dentists, especially orthodontists, are often the doctors with a chance to detect the risk of the existence or potential development of OSA in a pediatric patient. At the same time their treatment influences craniofacial structures, which may play a role in the development of OSA in children. Last but not least, orthodontic therapy is gradually becoming one of the therapies used to treat this disease worldwide [16].
CONCLUSION
Based on the above-mentioned brief overview of the issue of pediatric OSA, the specificity and complexity of the disease as well as its possible impact on the general health of the child patient are evident. The most serious consequences are insufficient quantity and quality of sleep, nightmares, problematic awakening, significant daytime sleepiness as well as problematic behaviour, lack of concentration, hyperactivity and poor school results.
In pediatric OSA, efforts should be made to recognize it as early as possible and then select the appropriate causal therapy, which is only possible if the cause of OSA is accurately specified.
The already mentioned questionnaires specifically modified for children are relatively simple, fast and effective way of primary detection of OSA risk in children. Although several OSA screening questionnaires are used abroad, there are only its adult versions in the Czech Republic, and the Epworth Sleep Scale is mainly used. But even this questionnaire for the assessment of the risk of OSA in a pediatric patient is not commonly available at the dentist and orthodontist offices. However, dentists can certainly help raise awareness of SRBD in children. Dentists can also reveal an increased risk of OSA in pediatric patients with a simple anamnestic inquiry about the pediatric patient´s sleep, the presence of night snoring, and dental attention to the above mentioned craniofacial features, short sublingual frenulum or loss/agenesis of teeth and oral breathing.
For better screening and simplification of the anamnesis not only at the dentist/orthodontist, it would be definitely appropriate to use a comprehensive and validated questionnaire, based on the existing foreign questionnaires intended for the pediatric population. The questionnaire should focus not only on OSA's night-time manifestations alone, but also on the consequences that the disease causes during the day and which parents or children are often more likely to notice and define. We are working on a questionnaire specific for the detection of OSA in Czech children and we proposed a methodical procedure for its validation (validation study is planned for 2020).
If OSA is detected early in a pediatric patient, the range of therapeutic options increases, including the ability to influence the proper growth and development of the orofacial structures of a growing individual by orthodontic treatment, which is no longer applicable in late detection. The greatest benefit of early detection and successful treatment of OSA in children is an increase in the quality and quantity of sleep, which is extremely important for the overall development and growth of children.
The study was supported by grant NV17-30439A, specific research MUNI/A/1546/2018 and funds provided by the Faculty of Medicine MU to junior researcher Petra Bořilová Linhartová.
Declaration for photographic documentation
Images are published with the patient's written consent
Corresponding author
RNDr. Petra Bořilová Linhartová, Ph.D.
Department of Pathophysiology, Faculty of Medicine, Masaryk University, Brno
Kamenice 5
625 00 Brno
Email: peta.linhartova@gmail.com
Zdroje
1. Farlex Partner Medical Dictionary. S.v. „sleep-disordered breathing." [cit. Farlex 8. 3. 2019] Dostupné z: https://medical-dictionary.thefreedictionary.com/sleep-disordered+breathing 1981; 159(1): 275–287.
2. Staevska MT, Baraniuk JN. Rhinitis and sleep apnea. Clin Allergy Immunol. 2007; 19 : 449.
3. Tsara V, Amfilochiou A, Papagrigorakis JM, Georgopoulos D, Liolios E, Kadiths A, Koudoumnakis E, Aulonitou E, Emporiadou M, Tsakanikos M, Chatzis A. Guidelines for diagnosing and treating sleep related breathing disorders in adults and children (Part 3: obstructive sleep apnea in children, diagnosis and treatment). Hippokratia. 2010; 14(1): 57.
4. Crummy F, Piper AJ, Naughton MT. Obesity and the lung: 2·Obesity and sleep-disordered breathing. Thorax. 2008; 63(8): 738–746.
5. Young T, Skatrud J, Peppard PE. Risk factors for obstructive sleep apnea in adults. Jama. 2004; 291(16): 2013.
6. Guilleminault C, Eldridge FL, Simmons FB, Dement WC. Sleep apnea in eight children. Pediatrics. 1976; 58(1): 23–30.
7. Guilleminault C, Korobkin R, Winkle R. A review of 50 children with obstructive sleep apnea syndrome. Lung. 1981; 159(1): 275–287.
8. Galievsky M, Lambert A. Sleep respiratory problems in children: Diagnosis and contribution of the orthodontist. Int Orthod. 2017; 15(3): 405–423.
9. Owens J, Opipari L, Nobile C, Spirito A. Sleep and daytime behavior in children with obstructive sleep apnea and behavioral sleep disorders. Pediatrics. 1998; 102(5): 1178–1184.
10. Chervin RD, Dillon JE, Bassetti C, Ganoczy DA, Pituch KJ. Symptoms of sleep disorders, inattention, and hyperactivity in children. Sleep. 1997; 20(12): 1185–1192.
11. Chervin RD, Archbold KH. Hyperactivity and polysomnographic findings in children evaluated for sleep-disordered breathing. Sleep. 2001; 24(3): 313–320.
12. O’Brien LM, Holbrook CR, Mervis CB, Klaus CJ, Bruner JL, Raffield TJ, Rutherford J, Mehl RC, Wang M, Tuell A, Hume BC. Sleep and neurobehavioral characteristics of 5 - to 7-year-old children with parentally reported symptoms of attention-deficit/hyperactivity disorder. Pediatrics. 2003; 111(3): 554–563.
13. Katz ES, D'ambrosio CM. Pathophysiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008; 5(2): 253–262.
14. Carroll JL. Obstructive sleep-disordered breathing in children: new controversies, new directions. Clin Chest Med. 2003; 24(2): 261–282.
15. Lumeng JC, Chervin RD. Epidemiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008; 5(2): 242–252.
16. Huang YS, Guilleminault C. A review of treatment options in paediatric sleep-disordered breathing. Pediatr Respirol Crit Care Med. 2017; 1(3): 54.
17. Příhodová MI. Obstrukční spánková apnoe u dětí – opomíjená diagnóza. Pediatr pro Praxi. 2010; 11(1): 26–28.
18. Casale M, Pappacena M, Rinaldi V, Bressi F, Baptista P, Salvinelli F. Obstructive sleep apnea syndrome: from phenotype to genetic basis. Curr Genomics. 2009; 10(2): 119–126.
19. Kheirandish-Gozal L, Khalyfa A, Gozal D, Bhattacharjee R, Wang Y. Endothelial dysfunction in children with obstructive sleep apnea is associated with epigenetic changes in the eNOS gene. Chest. 2013; 143(4): 971–977.
20. Wu BG, Sulaiman I, Wang J, Shen N, Clemente JC, Li Y, Laumbach RJ, Lu SE, Udasin I, Le-Hoang O, Perez A. Severe obstructive sleep apnea is associated with alterations in the nasal microbiome and an increase in inflammation. Am J Respir Crit Care Med. 2019; 199(1): 99–109.
21. Driessen C, Joosten KF, Florisson JM, Lequin M, van Veelen ML, Dammers R, Bredero Boelhouwer H, Tasker RC, Mathijssen IM. Sleep apnoea in syndromic craniosynostosis occurs independent of hindbrain herniation. Childs Nerv Syst. 2013; 29(2): 289–296.
22. MacLean JE, Fitzsimons D, Fitzgerald DA, Waters KA. The spectrum of sleep-disordered breathing symptoms and respiratory events in infants with cleft lip and/or palate. Arch Dis Child. 2012; 97(12): 1058–1063.
23. Moraleda-Cibrián M, Edwards SP, Kasten SJ, Buchman SR, Berger M, O'Brien LM. Obstructive sleep apnea pretreatment and posttreatment in symptomatic children with congenital craniofacial malformations. J Clin Sleep Med. 2015; 11(01): 37–43.
24. Queiroz IM. Lingual frenulum: classification and speech interference. Int J Orofacial Myology. 2004; 30 : 31–38.
25. Wallace H, Clarke S. Tongue tie division in infants with breast feeding difficulties. Int J Pediatr Otorhinolaryngol. 2006; 70(7): 1257–1261.
26. Guilleminault C, Huseni S, Lo L. A frequent phenotype for paediatric sleep apnoea: short lingual frenulum. ERJ Open Res. 2016; 2(3): 00043–2016.
27. Pola M, Garcia MG, Martín JM, Gallas M, Lestón JS. A study of pathology associated with short lingual frenum ASDC J Dent Child. 2002; 69(1): 59–62.
28. Huang YS, Quo S, Berkowski JA, Guilleminault C. Short lingual frenulum and obstructive sleep apnea in children. Int J Pediatr Res. 2015; 1(003).
29. Fitzpatrick MF, McLean H, Urton AM, Tan A, O'donnell D, Driver HS. Effect of nasal or oral breathing route on upper airway resistance during sleep. Eur Respir J. 2003; 22(5): 827–832.
30. Huang YS, Guilleminault C. Pediatric obstructive sleep apnea: Where do we stand? In: Lin HC. Sleep-related breathing disorders. vol. 80. Basel: Karger; 2017, 136–144.
31. Pacheco MC, Fiorott BS, Finck NS, Araújo MT. Craniofacial changes and symptoms of sleep-disordered breathing in healthy children. Dental Press J Orthod. 2015; 20(3): 80–87.
32. Vargervik K, Miller AJ, Chierici G, Harvold E, Tomer BS. Morphologic response to changes in neuromuscular patterns experimentally induced by altered modes of respiration. Am J Orthod. 1984; 85(2): 115–124.
33. Harvold EP, Tomer BS, Vargervik K, Chierici G. Primate experiments on oral respiration. Am J Orthod. 1981; 79(4): 359–372.
34. Ben-Bassat Y, Brin I. Skeletal and dental patterns in patients with severe congenital absence of teeth. Am J Orthod Dentofacial Orthop. 2009; 135(3): 349–356.
35. Guilleminault C, Abad V, Chiu H, Quo S. Obstructive-sleep-apnea and alveolo-dental synchondrosis. Sleep Med. 2015; 16 : 80.
36. Shintani T, Asakura K, Kataura A. Adenotonsillar hypertrophy and skeletal morphology of children with obstructive sleep apnea syndrome. Acta Otolaryngol Suppl. 1996; 523 : 222–224.
37. Ågren K, Nordlander B, Linder-Aronsson S, Zettergren-Wijk L, Svanborg E. Children with nocturnal upper airway obstruction: postoperative orthodontic and respiratory improvement. Acta Otolaryngol. 1998; 118(4): 581–587.
38. Löfstrand-Tideström B, Thilander B, Ahlqvist-Rastad J, Jakobsson O, Hultcrantz E. Breathing obstruction in relation to craniofacial and dental arch morphology in 4-year-old children. Eur J Orthod. 1999; 21(4): 323–332.
39. Zucconi M, Caprioglio A, Calori G, Ferini-Strambi L, Oldani A, Castronovo C, Smirne S. Craniofacial modifications in children with habitual snoring and obstructive sleep apnoea: a case-control study. Eur Respir J. 1999; 13(2): 411–417.
40. Finkelstein Y, Wexler D, Berger G, Nachmany A, Shapiro-Feinberg M, Ophir D. Anatomical basis of sleep-related breathing abnormalities in children with nasal obstruction. Arch Otolaryngol Head Neck Surg. 2000; 126(5): 593–600.
41. Kawashima S, Niikuni N, Chia-hung L, Takahasi Y, Kohno M, Nakajima I, Akasaka M, Sakata H, Akashi S. Cephalometric comparisons of craniofacial and upper airway structures in young children with obstructive sleep apnea syndrome. Ear Nose Throat J. 2000; 79(7).
42. Zettergren-Wijk L, Forsberg CM, Linder-Aronson S. Changes in dentofacial morphology after adeno-/tonsillectomy in young children with obstructive sleep apnoea – a 5-year follow-up study. Eur J Orthod. 2006; 28(4): 319–326.
43. Raffaele IR. Habitual snoring, OSA and craniofacial modification. Orthodontic clinical and diagnostic aspects in a case control study. Minerva Stomatol. 1999; 48(4): 125–137.
44. Kim JH, Guilleminault C. The nasomaxillary complex, the mandible, and sleep-disordered breathing. Sleep Breath. 2011; 15(2): 185–193.
45. Pirilä-Parkkinen K, Löppönen H, Nieminen P, Tolonen U, Pirttiniemi P. Cephalometric evaluation of children with nocturnal sleep-disordered breathing. Eur J Orthod. 2010; 32(6): 662–671.
46. El Aouame A, Daoui A, El Quars F. Nasal breathing and the vertical dimension: a cephalometric study. Int Orthod. 2016; 14(4): 491–502.
47. Hultcrantz E, Tideström BL. The development of sleep disordered breathing from 4 to 12 years and dental arch morphology. Int J Pediatr Otorhinolaryngol. 2009; 73(9): 1234–1241.
48. Isono S, Shimada A, Utsugi M, Konno A, Nishino T. Comparison of static mechanical properties of the passive pharynx between normal children and children with sleep-disordered breathing. Am J Respir Crit Care Med. 1998; 157(4): 1204–1212.
49. Fregosi RF, Quan SF, Kaemingk KL, Morgan WJ, Goodwin JL, Cabrera R, Gmitro A. Sleep disordered breathing, pharyngeal size and soft tissue anatomy in children. J Appl Physiol. 2003; 95(5): 2030–2038.
50. Ikävalko T, Närhi M, Eloranta AM, Lintu N, Myllykangas R, Vierola A, Tuomilehto H, Lakka T, Pahkala R. Predictors of sleep disordered breathing in children: the PANIC study. Eur J Orthod. 2017; 40(3): 268–272.
51. Min GU, McGrath CP, Wong RW, Hägg U, Yang Y. Cephalometric norms for the upper airway of 12-year-old Chinese children. Head Face Med. 2014; 10(1): 38.
52. Dibelka F. The Pierre Robin syndrome. Čes Stomatol. 1978; 78(3): 199–202.
53. Česká společnost pro výzkum spánku a spánkovou medicínu. Doporučený postup u dětských pacientů s poruchami dýchání ve spánku. [cit. 28. 8. 2019]. Dostupné z: http://www.sleep-society.cz/images/odborne/doporucene-postupy/Guidelines-2016-def-deti-13.9..pdf
54. Garetz SL, Mitchell RB, Parker PD, Moore RH, Rosen CL, Giordani B, Muzumdar H, Paruthi S, Elden L, Willging P, Beebe DW. Quality of life and obstructive sleep apnea symptoms after pediatric adenotonsillectomy. Pediatrics. 2015; 135(2): e477–486.
55. Chervin RD, Hedger K, Dillon JE, Pituch KJ. Pediatric sleep questionnaire (PSQ): validity and reliability of scales for sleep-disordered breathing, snoring, sleepiness, and behavioral problems. Sleep med. 2000; 1(1): 21–32.
56. Sohn H, Rosenfeld RM. Evaluation of sleep-disordered breathing in children. Otolaryngol Head Neck Surg. 2003; 128(3): 344–352.
57. De Serres LM, Derkay C, Astley S, Deyo RA, Rosenfeld RM, Gates GA. Measuring quality of life in children with obstructive sleep disorders. Arch Otolaryngol Head Neck Surg. 2000; 126(12): 1423–1429.
58. Constantin E, Tewfik TL, Brouillette RT. Can the OSA-18 quality-of-life questionnaire detect obstructive sleep apnea in children? Pediatrics. 2010; 125(1): e162–168.
59. Isaiah A, Kiss E, Olomu P, Koral K, Mitchell RB. Characterization of upper airway obstruction using cine MRI in children with residual obstructive sleep apnea after adenotonsillectomy. Sleep Med. 2018; 50 : 79–86.
60. Hatcher DC. Cone beam computed tomography: craniofacial and airway analysis. Dent Clin North Am. 2012; 56(2): 343–357.
61. Shi H, Scarfe WC, Farman AG. Upper airway segmentation and dimensions estimation from cone-beam CT image datasets. I Int J Comput Assist Radiol Surg. 2006; 1(3): 177–186.
62. Ogawa T, Enciso R, Shintaku WH, Clark GT. Evaluation of cross-section airway configuration of obstructive sleep apnea. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007; 103(1): 102–108.
63. McCrillis JM, Haskell J, Haskell BS, Brammer M, Chenin D, Scarfe WC, Farman AG. Obstructive sleep apnea and the use of cone beam computed tomography in airway imaging: a review. Semin Orthod. 2009; 15(1): 63–69.
64. Aboudara CA, Hatcher D, Nielsen IL, Miller A. A three–dimensional evaluation of the upper airway in adolescents. Orthod Craniofac Res. 2003; 1 : 173–175.
65. Michaelson PG, Allan P, Chaney J, Mair EA. Validations of a portable home sleep study with twelve-lead polysomnography: comparisons and insights into a variable gold standard. Ann Otol Rhinol Laryngol. 2006; 115(11): 802–809.
66. Kirk V, Baughn J, D'Andrea L, Friedman N, Galion A, Garetz S, Hassan F, Wrede J, Harrod CG, Malhotra RK. American Academy of Sleep Medicine position paper for the use of a home sleep apnea test for the diagnosis of OSA in children. J Clin Sleep Med. 2017; 13(10): 1199–1203.
67. Loughlin GM, Brouillette RT, Brooke LJ, Carroll JL, Chipps BE, England SJ, Ferber P, Ferraro NF, Gaultier C, Givan DC, Haddad GG. Standards and indications for cardiopulmonary sleep studies in children. Am J Respir Crit Care Med. 1996; 153(2): 866–878.
68. Huang YS, Guilleminault C, Lee LA, Lin CH, Hwang FM. Treatment outcomes of adenotonsillectomy for children with obstructive sleep apnea: a prospective longitudinal study. Sleep. 2014; 37(1): 71–76.
69. Bhattacharjee R, Kheirandish-Gozal L,Spruyt K, Mitchell RB, Promchiarak J, Simakajornboon N, Kaditis AG, Splaingard D, Splaingard M, Brooks LJ, Marcus CL. Adenotonsillectomy outcomes in treatment of obstructive sleep apnea in children: a multicenter retrospective study. Am J Respir Crit Care Med. 2010; 182(5): 676–683.
70. Guilleminault C, Huang YS, Quo S, Monteyrol PJ, Lin CH. Teenage sleep-disordered breathing: recurrence of syndrome. Sleep Med. 2013; 14(1): 37–44.
71. Saito M, Yamaguchi T, Mikami S, Watanabe K, Gotouda A, Okada K, Hishikawa R, Shibuya E, Lavigne G. Temporal association between sleep apnea-hypopnea and sleep bruxism events. J Sleep Res. 2014; 23(2): 196–203.
72. Villa MP, Castaldo R, Miano S, Paolino MC, Vitelli O, Tabarrini A, Mazzotta AR, Cecili M, Barreto M. Adenotonsillectomy and orthodontic therapy in pediatric obstructive sleep apnea. Sleep Breath. 2014; 18(3): 533–539.
73. Pirelli P, Saponara M, Guilleminault C. Rapid maxillary expansion in children with obstructive sleep apnea syndrome. Sleep. 2004; 27(4): 761–766.
74. Pirelli P, Saponara M, Guilleminault C. Rapid maxillary expansion (RME) for pediatric obstructive sleep apnea: a 12-year follow-up. Sleep Med. 2015; 16(8): 933–935.
75. Rogers AP. Exercises for the development of the muscles of the face, with a view to increasing their functional activity. Dental Cosmos LX. 1918; 59(857): e76.
76. Guilleminault C. Pediatric obstructive sleep apnea and the critical role of oral-facial growth: evidences. Front Neurol. 2013; 3 : 184.
77. Guilleminault C, Akhtar F. Pediatric sleep-disordered breathing: New evidence on its development. Sleep Med Rev. 2015; 24 : 46–56.
78. Camacho M, Certal V, Abdullatif J, Zaghi S, Ruoff CM, Capasso R, Kushida CA. Myofunctional therapy to treat obstructive sleep apnea: a systematic review and meta-analysis. Sleep. 2015; 38(5): 669–675.
79. Chuang LC, Lian YC, Hervy-Auboiron M,Guilleminault C, Huang YS. Passive myofunctional therapy applied on children with obstructive sleep apnea: a 6-month follow-up. J Formos Med Assoc. 2017; 116(7): 536–541.
80. Timms DJ. The dawn of rapid maxillary expansion. Angle Orthod. 1999; 69(3): 247–250.
81. Haas AJ. The treatment of maxillary deficiency by opening the midpalatal suture. Angle Orthod. 1965; 35(3): 200–217.
82. Biederman W. A hygienic appliance for rapid expansion. JPO J Pract Orthod. 1968; 2(2): 67–70.
83. Schmidt-Nowara W, Lowe A, Wiegand L, Cartwright R, Perez-Guerra F, Menn S. Oral appliances for the treatment of snoring and obstructive sleep apnea: a review. Sleep. 1995; 18(6): 501–510.
84. Hershey HG, Stewart BL, Warren DW. Changes in nasal airway resistance associated with rapid maxillary expansion. Am J Orthod. 1976; 69(3): 274–284.
85. Hartgerink DV, Vig PS, Orth D, Abbott DW. The effect of rapid maxillary expansion on nasal airway resistance. Am J Orthod Dentofacial Orthop. 1987; 92(5): 381–389.
86. Hilberg OA, Jackson AC, Swift DL, Pedersen OF. Acoustic rhinometry: evaluation of nasal cavity geometry by acoustic reflection. J Appl Physiol. 1989; 66(1): 295–303.
87. Enoki C, Valera FC, Lessa FC, Elias AM, Matsumoto MA, Anselmo-Lima WT. Effect of rapid maxillary expansion on the dimension of the nasal cavity and on nasal air resistance. Int J Pediatr Otorhinolaryngol. 2006; 70(7): 1225–1230.
88. Bicakci AA, Agar U, Sökücü O, Babacan H, Doruk C. Nasal airway changes due to rapid maxillary expansion timing. Angle Orthod. 2005; 75(1): 1–6.
89. Compadretti GC, Tasca I, Bonetti GA. Nasal airway measurements in children treated by rapid maxillary expansion. Am J Rhinol. 2006; 20(4): 385–393.
90. Katyal V, Pamula Y, Martin AJ, Daynes CN, Kennedy JD, Sampson WJ. Craniofacial and upper airway morphology in pediatric sleep-disordered breathing: systematic review and meta-analysis. Am J Orthod Dentofacial Orthop. 2013; 143(1): 20–30.
91. Basciftci FA, Mutlu N, Karaman AI, Malkoc S, Küçükkolbasi H. Does the timing and method of rapid maxillary expansion have an effect on the changes in nasal dimensions? Angle Orthod. 2002; 72(2): 118–123.
92. Cistulli PA, Palmisano RG, Poole MD. Treatment of obstructive sleep apnea syndrome by rapid maxillary expansion. Sleep. 1998; 21(8): 831–835.
93. Iwasaki T, Saitoh I, Takemoto Y, Inada E, Kakuno E, Kanomi R, Hayasaki H, Yamasaki Y. Tongue posture improvement and pharyngeal airway enlargement as secondary effects of rapid maxillary expansion: a cone-beam computed tomography study. Am J Orthod Dentofacial Orthop. 2013; 143(2): 235–245.
94. Pirelli P, Saponara M, Attanasio G. Obstructive Sleep Apnoea Syndrome (OSAS) and rhino-tubaric disfunction in children: therapeutic effects of RME therapy. Prog Orthod. 2005; 6(1): 48–61.
95. Izuka EN, Feres MF, Pignatari SS. Immediate impact of rapid maxillary expansion on upper airway dimensions and on the quality of life of mouth breathers. Dental Press J Orthod. 2015; 20(3): 43–49.
96. Villa MP, Rizzoli A, Miano S, Malagola C. Efficacy of rapid maxillary expansion in children with obstructive sleep apnea syndrome: 36 months of follow-up. Sleep Breath. 2011; 15(2): 179–184.
97. Villa MP, Malagola C, Pagani J, Montesano M, Rizzoli A, Guilleminault C, Ronchetti R. Rapid maxillary expansion in children with obstructive sleep apnea syndrome: 12-month follow-up. Sleep med. 2007; 8(2): 128–134.
98. Júnior AJ, Zancanella E, Crespo AN. Rapid maxillary expansion and obstructive sleep apnea: A review and meta-analysis. Med Oral Patol Oral Cir Bucal. 2016; 21(4): e465–e469.
99. Ghodke S, Utreja AK, Singh SP, Jena AK. Effects of twin-block appliance on the anatomy of pharyngeal airway passage (PAP) in class II malocclusion subjects. Prog Orthod. 2014; 15(1): 68.
100. Zhang C, He H, Ngan P. Effects of twin block appliance on obstructive sleep apnea in children: a preliminary study. Sleep Breath. 2013; 17(4): 1309–1314.
101. Iwasaki T, Takemoto Y, Inada E, Sato H, Saitoh I, Kakuno E, Kanomi R, Yamasaki Y. Three-dimensional cone-beam computed tomography analysis of enlargement of the pharyngeal airway by the Herbst appliance. Am J Orthod Dentofacial Orthop. 2014; 146(6): 776–785.
102. Schütz TC, Dominguez GC, Hallinan MP, Cunha TC, Tufik S. Class II correction improves nocturnal breathing in adolescents. Angle Orthod. 2011; 81(2): 222–228.
103. Ow AT, Cheung LK. Meta-analysis of mandibular distraction osteogenesis: clinical applications and functional outcomes. Plast Reconstr Surg. 2008; 121(3): 54e–69e.
104. Noller MW, Guilleminault C, Gouveia CJ, Mack D, Neighbors CL, Zaghi S, Camacho M. Mandibular advancement for pediatric obstructive sleep apnea: a systematic review and meta-analysis. J Craniomaxillofac Surg. 2018; 46(8): 1296–1302.
105. Schütz TC, Dominguez GC, Hallinan MP, Cunha TC, Tufik S. Class II correction improves nocturnal breathing in adolescents. Angle Orthod. 2011; 81(2): 222–228.
106. Galeotti A, Festa P, Pavone M, De Vincentiis GC. Effects of simultaneous palatal expansion and mandibular advancement in a child suffering from OSA. Acta Otorhinolaryngol Ital. 2016; 36(4): 328.
Štítky
Chirurgie maxilofaciální Ortodoncie Stomatologie
Článek vyšel v časopiseČeská stomatologie / Praktické zubní lékařství
Nejčtenější tento týden
2020 Číslo 1- Horní limit denní dávky vitaminu D: Jaké množství je ještě bezpečné?
- Orální lichen planus v kostce: Jak v praxi na toto multifaktoriální onemocnění s různorodými symptomy?
- Význam ústní sprchy pro čištění mezizubních prostor
- Diagnostika alergie na bílkoviny kravského mléka − aktuální postupy a jejich vypovídací hodnota
- Benzydamin v léčbě zánětů v dutině ústní
Nejčtenější v tomto čísle- The impact of neglect and failed treatment of early childhood caries
- Universal adhesives – a new direction in the development of adhesive systems
- Obstructive sleep apnea in relation to orthodontic treatment in children
- Profesor Zdeněk Broukal zemřel
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání




